Carbon tetrachloride: Difference between revisions
No edit summary |
Revert vandalism |
||
| Line 2: | Line 2: | ||
! {{chembox header}} | Carbon tetrachloride |
! {{chembox header}} | Carbon tetrachloride |
||
|- |
|- |
||
| align="center" colspan="2" bgcolor="#ffffff" | [[Image: |
| align="center" colspan="2" bgcolor="#ffffff" | [[Image:Carbon Tetrachloride.PNG|150px|Carbon tetrachloride]] [[Image:Carbon-tetrachloride-3D-vdW.png|150px|Carbon tetrachloride]] |
||
|- |
|- |
||
! {{chembox header}} | General |
! {{chembox header}} | General |
||
Revision as of 05:00, 30 November 2006
| Carbon tetrachloride | |||||
|---|---|---|---|---|---|
| |||||
| General | |||||
| Systematic name | Carbon tetrachloride Tetrachloromethane | ||||
| Other names | benziform, carbon chloride, methane tetrachloride, perchloromethane tetrachoromethane | ||||
| Molecular formula | CCl4 | ||||
| SMILES | ClC(Cl)(Cl)Cl | ||||
| Molar mass | 153.82 g/mol | ||||
| Appearance | colorless liquid | ||||
| CAS number | [56-23-5] | ||||
| Properties | |||||
| Density and phase | 1.5842 g/ml, liquid | ||||
| Solubility in water | 0.08 g/100 ml (20 °C) | ||||
| Melting point | −22.9 °C (250 K) | ||||
| Boiling point | 76.8 °C (350 K) | ||||
| Viscosity | 0.901 cP at 25 °C | ||||
| Structure | |||||
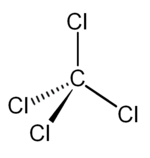
| Molecular shape | tetrahedral | ||||
| Dipole moment | zero | ||||
| Thermodynamic data | |||||
| Std enthalpy change of formation ΔfH |
−128 kJ/mol | ||||
| Standard molar entropy S |
216.4 J.K−1.mol−1 | ||||
| Hazards | |||||
| MSDS | External MSDS | ||||
| EU classification | Toxic (T) Carc. Cat. 3 Dangerous for the environment (N) | ||||
| NFPA 704 |
| ||||
| R-phrases | Template:R23/24/25, Template:R40, Template:R48/23, Template:R59, Template:R52/53 | ||||
| S-phrases | Template:S1/2, Template:S23, Template:S36/37, Template:S45, Template:S59, Template:S61 | ||||
| Flash point | non flammable | ||||
| RTECS number | FG4900000 | ||||
| Supplementary data page | |||||
| Structure and properties |
n, εr, etc. | ||||
| Thermodynamic data |
Phase behaviour Solid, liquid, gas | ||||
| Spectral data | UV, IR, NMR, MS | ||||
| Related compounds | |||||
| Related carbon tetrahalides |
Tetrafluoromethane Tetrabromomethane Tetraiodomethane | ||||
| Related chloromethanes | Chloromethane Dichloromethane Chloroform | ||||
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | |||||
Carbon tetrachloride, also known by other names (see Table) is the chemical compound CCl4. It is widely used in synthetic chemistry and formerly widely used in fire extinguishers and refrigeration, but largely abandoned. At room temperature and pressure, it is a colorless liquid with a "sweet" smell that can be detected at low levels.
Both carbon tetrachloride and tetrachloromethane are acceptable names under IUPAC nomenclature, depending on whether it is seen as an inorganic or an organic compound. Colloquially, it is called "carbon tet". Trade names include Benzinoform, Freon 10, Halon 104, Tetraform, and Tetrasol.
Production
Most carbon tetrachloride is produced by the chlorination of carbon disulfide at 105 to 130 °C:
CCl4 is also a byproduct in the synthesis of dichloromethane and chloroform via the reaction:
Chemical properties
In the carbon tetrachloride molecule, four chlorine atoms are positioned symmetrically as corners in a tetrahedral configuration joined to a carbon atom, in the center, by single covalent bonds. Because of this symmetrical geometry the molecule having no net dipole moment, i.e. CCl4 is non-polar.
As a solvent, it is well suited to dissolving other non-polar compounds. It is somewhat volatile, giving off vapors having a smell characteristic of other chlorinated solvents, somewhat similar to the tetrachloroethylene smell reminiscent of dry cleaners' shops.
Carbon tetrachloride has practically no flammability at lower temperatures.
Because it has no C-H bonds, carbon tetrachloride does not easily undergo free-radical reactions. Hence it is a useful solvent for halogenations either by the elemental halogen, or by a halogenation reagent such as N-bromosuccinimide.
Uses
In the early 20th century, carbon tetrachloride was widely used as a dry cleaning solvent, as a refrigerant, and in fire extinguishers. However, once it became apparent that carbon tetrachloride exposure had severe adverse health effects, safer alternatives were found for these applications, and its use in these roles declined from about 1940 onward. Carbon tetrachloride persisted as a pesticide to kill insects in stored grain, but in 1970, it was banned in consumer products in the United States.
Prior to the Montreal Protocol, large quantities of carbon tetrachloride were used to produce the freon refrigerants R-11 and R-12. However, these refrigerants are now believed to play a role in ozone depletion and have been phased out of use, though it is still used to manufacture less destructive refrigerants.
Carbon tetrachloride has also been used in the detection of neutrinos, and is a useful source of chlorine in the Appel reaction.
It is sometimes useful as solvent for infrared spectroscopy because there are no significant absorption bands > 1600 cm-1. Because carbon tetrachloride does not have any hydrogens, it was historically used in proton NMR spectroscopy. However, carbon tetrachloride is toxic, and its dissolving power is low.[1]. Its use has been largely superseded by deuterated solvents, which allow for deuterium lock by the spectrometer.
Safety
Exposure to high concentrations of carbon tetrachloride (including vapor) can affect the central nervous system and may result (after prolonged exposure) to coma and even death can occur. Chronic exposure to carbon tetrachloride can cause liver and kidney damage and may result in cancer. More information can be found in an MSDS.
Carbon tetrachloride is also an ozone-depleting greenhouse gas, but is rarely mentioned when talking about ozone depletion, due to the fact that its levels have been steadily decreasing since 1996.[citation needed]
See also
References
- Recknagel R.O., Glende E.A., Dolak J.A., Waller R.L. (1989). "Mechanism of Carbon-tetrachloride Toxicity". Pharmacology Therapeutics (43): 139–154. doi:10.1016/0163-7258(89)90050-8.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - Doherty R. E. (2000). "A History of the Production and Use of Carbon Tetrachloride, Tetrachloroethylene, Trichloroethylene and 1,1,1-Trichloroethane in the United States: Part 1--Historical Background; Carbon Tetrachloride and Tetrachloroethylene" (1): 69–81. doi:10.1006/enfo.2000.0010.
{{cite journal}}: Cite journal requires|journal=(help)

