Aortic dissection
This article needs additional citations for verification. (January 2008) |
| Aortic dissection | |
|---|---|
| Specialty | Vascular surgery |
Aortic dissection is a tear in the wall of the aorta that causes blood to flow between the layers of the wall of the aorta and force the layers apart.[1] Aortic dissection is a medical emergency and can quickly lead to death, even with optimal treatment. If the dissection tears the aorta completely open (through all three layers), massive and rapid blood loss occurs. Aortic dissections resulting in rupture have an 80% mortality rate, and 50% of patients die before they even reach the hospital. If the dissection reaches 6 cm, the patient must be taken for emergency surgery.[2]
Overview

As with all other arteries, the aorta is made up of three layers, the intima, the media, and the adventitia. The layer that is in direct contact with the flow of blood is the tunica intima, commonly called the intima. This layer is made up of mainly endothelial cells. The next layer is known as the tunica media, or the muscular layer. It is composed of a mixture of smooth muscle cells and elastic fibers. The outermost layer is the tunica adventitia or the adventitia. This layer is composed of connective tissue.
In an aortic dissection, blood penetrates the intima and enters the media layer. The high pressure rips the tissue of the media apart along the laminated plane splitting the inner 2/3 and the outer 1/3 of the media apart.[citation needed] This can propagate along the length of the aorta for a variable distance forward or backwards. Dissections that propagate towards the iliac bifurcation (with the flow of blood) are called anterograde dissections and those that propagate towards the aortic root (opposite of the flow of blood) are called retrograde dissections. The initial tear is usually within 100 mm of the aortic valve so a retrograde dissection can easily compromise the pericardium leading to a hemocardium. Anterograde dissections may propagate all the way to the iliac bifurcation of the aorta, rupture the aortic wall, or recanalize into the intravascular lumen leading to a double barrel aorta. The double barrel aorta relieves the pressure of blood flow and reduces the risk of rupture. Rupture leads to hemorrhaging into a body cavity and prognosis depends on the area of rupture. Retroperitoneal and pericardial ruptures are both possible.
Classification

|
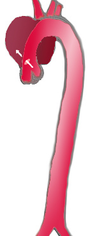
|

| |
| Percentage | 60 % | 10-15 % | 25-30 % |
| Type | DeBakey I | DeBakey II | DeBakey III |
| Stanford A | Stanford B | ||
| Proximal | Distal | ||
| Classification of aortic dissection | |||
Several different classification systems have been used to describe aortic dissections. The systems commonly in use are either based on the anatomy of the dissection or the duration of onset of symptoms prior to presentation.
DeBakey
The DeBakey system, named after surgeon and aortic dissection sufferer Michael E. DeBakey, is an anatomical description of the aortic dissection. It categorizes the dissection based on where the original intimal tear is located and the extent of the dissection (localized to either the ascending aorta or descending aorta, or involves both the ascending and descending aorta.[3]
- Type I - Originates in ascending aorta, propagates at least to the aortic arch and often beyond it distally.
- Type II – Originates in and is confined to the ascending aorta.
- Type III – Originates in descending aorta, rarely extends proximally but will extend distally.
Stanford
Divided into 2 groups; A and B depending on whether the ascending aorta is involved.[4]
- A - Involves the ascending aorta and/or aortic arch, and possibly the descending aorta. The tear can originate in the ascending aorta, the aortic arch, or, more rarely, in the descending aorta.[5] It includes DeBakey type I, II and retrograde type III[6] (dissection originating in the descending aorta or aortic arch but extending into the ascending aorta).
- B - Involves the descending aorta (distal to left subclavian artery origin), without involvement of the ascending aorta or aortic arch.[5] It includes DeBakey type III without retrograde extension into the ascending aorta.
The Stanford classification is in close relationship to clinical practice, as type A dissections generally require primary surgical repair whereas type B dissections generally are treated medically as initial treatment with surgery reserved for any complications.[7]
Signs and symptoms
About 96% of individuals with aortic dissection present with severe pain that had a sudden onset. It may be described as tearing in nature, or stabbing or sharp in character.[8] 17% of individuals will feel the pain migrate as the dissection extends down the aorta. The location of pain is associated with the location of the dissection. Anterior chest pain is associated with dissections involving the ascending aorta, while interscapular (back) pain is associated with descending aortic dissections. If the pain is pleuritic in nature, it may suggest acute pericarditis caused by hemorrhage into the pericardial sac. This is a particularly dangerous eventuality, suggesting that acute pericardial tamponade may be imminent.
While the pain may be confused with the pain of a myocardial infarction (heart attack), aortic dissection is usually not associated with the other signs that suggest myocardial infarction, including heart failure, and ECG changes. Also, individuals suffering from an aortic dissection usually do not present with diaphoresis (profuse sweating).
Individuals with aortic dissection who do not present with pain have chronic dissection.
Less common symptoms that may be seen in the setting of aortic dissection include congestive heart failure (7%), syncope (9%), cerebrovascular accident (3-6%), ischemic peripheral neuropathy, paraplegia, cardiac arrest, and sudden death. If the individual had a syncopal episode, about half the time it is due to hemorrhage into the pericardium leading to pericardial tamponade.
Neurologic complications of aortic dissection (i.e., cerebrovascular accident (CVA) and paralysis) are due to involvement of one or more arteries supplying portions of the central nervous system.
If the aortic dissection involves the abdominal aorta, compromise of the branches of the abdominal aorta is possible. In abdominal aortic dissections, compromise of one or both renal arteries occurs in 5-8% of cases, while mesenteric ischemia (ischemia of the large intestines) occurs 3-5% of the time.
Blood pressure changes
While many patients with an aortic dissection have a history of hypertension, the blood pressure is quite variable at presentation with acute aortic dissection, and tends to be higher in individuals with a distal dissection. In individuals with a proximal aortic dissection, 36% present with hypertension, while 25% present with hypotension. In those that present with distal aortic dissections, 70% present with hypertension while 4% present with hypotension.
Severe hypotension at presentation is a grave prognostic indicator. It is usually associated with pericardial tamponade, severe aortic insufficiency, or rupture of the aorta. Accurate measurement of the blood pressure is important. Pseudohypotension (falsely low blood pressure measurement) may occur due to involvement of the brachiocephalic artery (supplying the right arm) or the left subclavian artery (supplying the left arm).
Aortic insufficiency
Aortic insufficiency (AI) occurs in 1/2 to 2/3 of ascending aortic dissections, and the murmur of aortic insufficiency is audible in about 32% of proximal dissections. The intensity (loudness) of the murmur is dependent on the blood pressure and may be inaudible in the event of hypotension.
There are multiple etiologies for AI in the setting of ascending aortic dissection. The dissection may dilate the annulus of the aortic valve, so that the leaflets of the valve cannot coapt. Another mechanism is that the dissection may extend into the aortic root and detach the aortic valve leaflets. The third mechanism is that if there was an extensive intimal tear, the intimal flap may prolapse into the LV outflow tract, causing intimal intussusception into the aortic valve preventing proper valve closure.
Myocardial infarction
Myocardial infarction (heart attack) occurs in 1-2% of aortic dissections. The etiology of the infarction is involvement of the coronary arteries (the arteries that supply the heart) in the dissection. The right coronary artery is involved more commonly than the left coronary artery. If the myocardial infarction is treated with thrombolytic therapy, the mortality increases to over 70%, mostly due to hemorrhage into the pericardial sac causing pericardial tamponade.
Because aortic dissection may present to the emergency room physician similar to a myocardial infarction, the physician must be careful to make the proper diagnosis prior to initiating treatment for myocardial infarction, since the treatment regimen for myocardial infarction can be lethal to an individual presenting with aortic dissection.
Pleural effusion
A pleural effusion (fluid collection in the space between the lungs and the chest wall or diaphragm) can be due to either blood from a transient rupture of the aorta or fluid due to an inflammatory reaction around the aorta. If a pleural effusion were to develop due to an aortic dissection, it is more commonly in the left hemithorax rather than the right hemithorax.
Causes
Aortic dissection is associated with hypertension (high blood pressure) and many connective tissue disorders. Vasculitis (inflammation of an artery) is rarely associated with aortic dissection. It can also be the result of chest trauma. 72 to 80% of individuals who present with an aortic dissection have a previous history of hypertension.
The highest incidence of aortic dissection is in individuals who are 50 to 70 years old. The incidence is twice as high in males as in females (male-to-female ratio is 2:1). Half of dissections in females before age 40 occur during pregnancy (typically in the 3rd trimester or early postpartum period).
A bicuspid aortic valve (a type of congenital heart disease involving the aortic valve) is found in 7-14% of individuals who have an aortic dissection. These individuals are prone to dissection in the ascending aorta. The risk of dissection in individuals with bicuspid aortic valve is not associated with the degree of stenosis of the valve.
Marfan syndrome is noted in 5-9% of individuals who suffer from aortic dissection. In this subset, there is an increased incidence in young individuals. Individuals with Marfan syndrome tend to have aneurysms of the aorta and are more prone to proximal dissections of the aorta.
Turner syndrome also increases the risk of aortic dissection, by aortic root dilatation.[9]
Chest trauma leading to aortic dissection can be divided into two groups based on etiology: blunt chest trauma (commonly seen in car accidents) and iatrogenic. Iatrogenic causes include trauma during cardiac catheterization or due to an intra-aortic balloon pump.
Aortic dissection may be a late sequela of cardiac surgery. 18% of individuals who present with an acute aortic dissection have a history of open heart surgery. Individuals who have undergone aortic valve replacement for aortic insufficiency are at particularly high risk. This is because aortic insufficiency causes increased blood flow in the ascending aorta. This can cause dilatation and weakening of the walls of the ascending aorta.
Syphilis typically does not cause aortic dissection. In the tertiary (cardiovascular) stage of that disease, the vasa vasorum of the aorta are obliterated, causing the media to become fibrotic. Hence, patients with tertiary lues develop thoracic aortic aneurysms that may rupture, but there is no longer an intraarterial plane in the aortic wall through which a dissection can track.
Pathophysiology

The initiating event in an aortic dissection is a tear in the intimal lining of the aorta. Due to the high pressures in the aorta, blood enters the media at the point of the tear. The force of the blood entering the media causes the tear to extend. It may extend proximally (closer to the heart) or distally (away from the heart) or both. The blood will travel through the media, creating a false lumen (the true lumen is the normal conduit of blood in the aorta). Separating the false lumen from the true lumen is a layer of intimal tissue. This tissue is known as the intimal flap.
The vast majority of aortic dissections originate with an intimal tear in either the ascending aorta (65%), the aortic arch (10%), or just distal to the ligamentum arteriosum in the descending thoracic aorta (20%).
As blood flows down the false lumen, it may cause secondary tears in the intima. Through these secondary tears, the blood can re-enter the true lumen.
While it is not always clear why an intimal tear may occur, quite often it involves degeneration of the collagen and elastin that make up the media. This is known as cystic medial necrosis and is most commonly associated with Marfan syndrome and is also associated with Ehlers-Danlos syndrome.
In about 13% of aortic dissections, there is no evidence of an intimal tear. It is believed that in these cases the inciting event is an intramural hematoma (caused by hemorrhage within the media). Since there is no direct connection between the true lumen and the false lumen in these cases, it is difficult to diagnose an aortic dissection by aortography if the etiology is an intramural hematoma. An aortic dissection secondary to an intramural hematoma should be treated the same as one caused by an intimal tear.
Diagnosis
Because of the varying symptoms and signs of aortic dissection depending on the initial intimal tear and the extent of the dissection, the proper diagnosis is sometimes difficult to make.
In an individual with chest pain radiating to the back, the differentials to consider include:
- Aortic dissection
- Myocardial infarction
- Acute aortic insufficiency
- Non-dissecting aortic aneurysm
- Pericarditis
- Musculoskeletal pain
- Mediastinal tumors
While taking a good history from the individual may be strongly suggestive of an aortic dissection, the diagnosis cannot always be made by history and physical signs alone. Often the diagnosis is made by visualization of the intimal flap on a diagnostic imaging test. Common tests used to diagnose an aortic dissection include a CT scan of the chest with iodinated contrast material and a trans-esophageal echocardiogram. Other tests that may be used include an aortogram or magnetic resonance angiogram (MRA) of the aorta. Each of these test have varying pros and cons and they do not have equal sensitivities and specificities in the diagnosis of aortic dissection.
In general, the imaging technique chosen is based on the pre-test likelihood of the diagnosis, availability of the testing modality, patient stability, and the sensitivity and specificity of the test.
Chest X-ray

Widening of the mediastinum on an x-ray of the chest has moderate sensitivity (67%) in the setting of an ascending aortic dissection.[10] However, it has low specificity, as many other conditions can cause a widening of the mediastinum on chest x-ray.
The calcium sign is a finding on chest x-ray that suggests aortic dissection. It is the separation of the intimal calcification from the outer aortic soft tissue border by 10 mm.
Pleural effusions may be seen on chest x-ray. They are more commonly seen in descending aortic dissections. If seen, they are typically in the left hemithorax.
Other findings include obliteration of the aortic knob, depression of the left mainstem bronchus, loss of the paratracheal stripe, and tracheal deviation.
About 12%-20% of individuals presenting with an aortic dissection have a "normal" chest x-ray.
ECG
There are no specific electrocardiographic findings associated with aortic dissection. About 1/3 of the time, the ECG will show signs of left ventricular hypertrophy, which is due to the long-standing hypertension seen in these individuals. Another 1/3 of the time the ECG would be considered "normal". If the ECG suggests cardiac ischemia in the setting of aortic dissection, involvement of the coronary arteries should be suspected.
Biochemical markers
While there are currently no blood tests that can accurately diagnose aortic dissection, research has been performed into the serial measurement of monoclonal antibodies to smooth muscle myosin heavy chains that appears to be both sensitive and specific for aortic dissection.[11] The sensitivity of this test is about 90% and the specificity is 97% within the first 12 hours of the beginning of the dissection, and this assay can accurately differentiate myocardial infarction from aortic dissection. This test is not currently available for the diagnosis of aortic dissection in the clinical setting.
Transesophageal echocardiography
The transesophageal echocardiogram (TEE) is a relatively good test in the diagnosis of aortic dissection, with a sensitivity of up to 98% and a specificity of up to 97%. It is a relatively non-invasive test, requiring the individual to swallow the echocardiography probe. It is especially good in the evaluation of AI in the setting of ascending aortic dissection, and to determine whether the ostia (origins) of the coronary arteries are involved. While many institutions give sedation during transesophageal echocardiography for added patient-comfort, it can be performed in cooperative individuals without the use of sedation. Disadvantages of the TEE include the inability to visualize the distal ascending aorta (the beginning of the aortic arch), and the descending abdominal aorta that lies below the stomach. A TEE may be technically difficult to perform in individuals with esophageal strictures or varices.
Aortogram
An aortogram involves placement of a catheter in the aorta and injection of contrast material while taking x-rays of the aorta. The procedure is known as aortography.
Computed tomography angiography

|

|
| Computed tomography (CT) | Legend |
| Aortic dissection Type Stanford A 1 Aorta ascendens, true Lumen - 2 false Lumen - 3 Pulmonary artery 4 Aorta descendens - 5 thoracic vertebra | |
Computed tomography angiography is a fast non-invasive test that will give an accurate three-dimensional view of the aorta. These images are produced by taking rapid thin cut slices of the chest and abdomen, and combining them in the computer to create cross-sectional slices. In order to delineate the aorta to the accuracy necessary to make the proper diagnosis, an iodinated contrast material is injected into a peripheral vein. Contrast is injected and the scan performed using a Bolus Tracking method. This is a type of scan timed to an injection, in order to capture the contrast as it enters the aorta. The scan will then follow the contrast as it flows though the vessel.
It has a sensitivity of 96 - 100% and a specificity of 96 to 100%. Disadvantages include the need for iodinated contrast material and the inability to diagnose the site of the intimal tear.
MRI
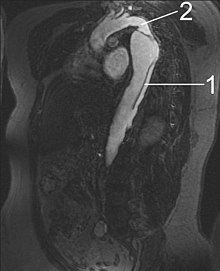
1 Aorta descendens with dissection
2 Aorta isthmus
Magnetic resonance imaging (MRI) is currently the gold standard test for the detection and assessment of aortic dissection, with a sensitivity of 98% and a specificity of 98%. An MRI examination of the aorta will produce a three-dimensional reconstruction of the aorta, allowing the physician to determine the location of the intimal tear, the involvement of branch vessels, and locate any secondary tears. It is a non-invasive test, does not require the use of iodinated contrast material, and can detect and quantitate the degree of aortic insufficiency.
The disadvantage of the MRI scan in the face of aortic dissection is that it has limited availability and is often located only in the larger hospitals, and the scan is relatively time consuming. Due to the high intensity magnetic fields used during MRI, an MRI scan is contraindicated in individuals with metallic implants. In addition, many individuals experience claustrophobia while in the MRI scanning tube.
Treatment
The risk of death due to aortic dissection is highest in the first few hours after the dissection begins, and decreases afterwards. Because of this, the therapeutic strategies differ for treatment of an acute dissection compared to a chronic dissection. An acute dissection is one in which the individual presents within the first two weeks. If the individual has managed to survive this window period, his prognosis is improved. About 66% of all dissections present in the acute phase.
In all individuals with aortic dissections, medication should be used to control high blood pressure, if present.
In the case of an acute dissection, once diagnosis has been confirmed, the choice of treatment depends on the location of the dissection. For ascending aortic dissection, surgical management is superior to medical management. On the other hand, in the case of an uncomplicated distal aortic dissections (including abdominal aortic dissections), medical management is preferred over surgical treatment.[12]
Individuals who present two weeks after the onset of the dissection are said to have chronic aortic dissections. These individuals have been self-selected as survivors of the acute episode, and can be treated with medical therapy as long as they are stable.
Medical management is appropriate in individuals with an uncomplicated distal dissection, a stable dissection isolated to the aortic arch, and stable chronic dissections. Patient selection for medical management is very important. Stable individuals who present with an acute distal dissection (typically treated with medical management) still have an 8 percent 30 day mortality.
VIDEO - What is New in the Treatment of Type B Aortic Dissection?, Girma Tefera, MD, speaks at the University of Wisconsin School of Medicine and Public Health.
Medical
The prime consideration in the medical management of aortic dissection is strict blood pressure control. The target blood pressure should be a mean arterial pressure (MAP) of 60 to 75 mmHg. Another factor is to reduce the shear-force dP/dt (force of ejection of blood from the left ventricle).
To reduce the shear stress, a vasodilator such as sodium nitroprusside should be used with a beta blocker, such as esmolol, propranolol, or labetalol. The alpha-blocking properties of labetalol make it especially attractive in this situation.
Calcium channel blockers can be used in the treatment of aortic dissection, particularly if there is a contraindication to the use of beta blockers. The calcium channel blockers typically used are verapamil and diltiazem, because of their combined vasodilator and negative inotropic effects.
If the individual has refractory hypertension (persistent hypertension on the maximum doses of three different classes of antihypertensive agents), involvement of the renal arteries in the aortic dissection plane should be considered.
Surgical
Indications for the surgical treatment of aortic dissection include an acute proximal aortic dissection and an acute distal aortic dissection with one or more complications. Complications include compromise of a vital organ, rupture or impending rupture of the aorta, retrograde dissection into the ascending aorta, and a history of Marfan's syndrome or Ehlers-Danlos Syndrome.
The objective in the surgical management of aortic dissection is to resect (remove) the most severely damaged segments of the aorta, and to obliterate the entry of blood into the false lumen (both at the initial intimal tear and any secondary tears along the vessel).[13][14][15][16] While excision of the intimal tear may be performed, it does not significantly change mortality.
Some methods of repair are:
- Replacement of the damaged section with a tube graft (often made of dacron) when there is no damage to the aortic valve.
- Bentall procedure - Replacement of the damaged section of aorta and replacement of the aortic valve.
- David procedure - Replacement of the damaged section of aorta and reimplantation of the aortic valve.
- Insertion of a stent graft (covered stent), e.g. in TEVAR (thoracic endovascular aortic repair). It is usually combined with on-going medical management
- Replacement of the damaged section with a sutureless ring-reinforced dacron graft [17]
A number of comorbid conditions increase the surgical risk of repair of an aortic dissection. These include:
- Prolonged pre-op evaluation (increased length of time prior to surgery)
- Advanced age
- Comorbid disease states (e.g.: coronary artery disease)
- Aneurysm leakage
- Cardiac tamponade
- Shock
- Past history of myocardial infarction or CVA
- History of renal failure (either acute or chronic renal failure)
Follow-up
The long term follow-up in individuals who survive aortic dissection involves strict blood pressure control. The relative risk of late rupture of an aortic aneurysm is 10 times higher in individuals who have uncontrolled hypertension, compared to individuals with a systolic pressure below 130 mmHg.
The risk of death is highest in the first two years after the acute event, and individuals should be followed closely during this time period. 29% of late deaths following surgery are due to rupture of either the dissecting aneurysm or another aneurysm. In addition, there is a 17% to 25% incidence of new aneurysm formation. This is typically due to dilatation of the residual false lumen. These new aneurysms are more likely to rupture, due to their thinner walls.
Serial imaging of the aorta is suggested, with MRI being the preferred imaging technique.
Prognosis
| Risk of death in untreated aortic dissection | |||
|---|---|---|---|
| 25% in first 24 hours | |||
| 50% in first 48 hours | |||
| 75% in first week | |||
| 90% in first month | |||
The risk of death is high in untreated aortic dissection. While the risk is very high in the first 24 hours of the event, those that survive the initial event still have an elevated mortality compared to age- and sex-matched controls. In those with ascending aortic dissection who are not treated 75% die within 2 weeks.[18] With aggressive treatment 30-day survival for thoracic dissections may be as high as 90%.[18]
Notable cases
- King George II of Great Britain[18]
- Jonathan Larson [19]
- John Ritter [20]
- Dr. Michael E. DeBakey, who devised the surgery to correct aortic dissection (as well as other cardiovascular and vascular techniques). Dr. DeBakey was also noteworthy for being the oldest patient ever to receive his own operation: he was 97 at the time of his surgery on February 9 and 10 of 2006. He survived and, though he worked with physical therapists to walk on his own again after suffering muscle deconditioning from prolonged physical inactivity during recovery, retained all of his mental faculties and was back to working nearly a full day until his death of natural causes on July 11, 2008. (See [3] published 25 December 2006, in the New York Times).
- John Lennon died from blood loss from aortic dissection brought on by a gunshot.[21]
- Gerard Houllier
- Lux Interior, lead singer of the band The Cramps, died from aortic dissection on February 4, 2009.
References
- ^ Diseases and Conditions at Mount Sinai Hospital
- ^ Isselbacher, EM (1997). "Diseases of the aorta". In Braunwald E (ed.). Heart disease: a textbook of cardiovascular medicine (5th ed.). Philadelphia: WB Saunders. pp. 1546–81.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|isbn-10=ignored (help) - ^ DeBakey ME, Henly WS, Cooley DA, Morris GC Jr, Crawford ES, Beall AC Jr (1965). "Surgical management of dissecting aneurysms of the aorta". J Thorac Cardiovasc Surg. 49: 130–49. PMID 14261867.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Daily PO, Trueblood HW, Stinson EB, Wuerflein RD, Shumway NE (1970). "Management of acute aortic dissections". Ann Thorac Surg. 10 (3): 237–47. doi:10.1016/S0003-4975(10)65594-4. PMID 5458238.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b University of Ottawa: Meds 2008 > Surgery Objectives
- ^ Antero-Lateral Thoracotomy with Partial Sternotomy for Retrograde DeBakey III Type Closing Aortic Dissection Accession number;06A0069173 Author;SUESHIRO MASAFUMI, HAYASHI SAIHO, KOBAYASHI HIRONORI Journal Title;Japanese Journal of Cardiovascular Surgery Journal Code:Y0192A ISSN:0285-1474 VOL.35;NO.1;PAGE.21-24(2006)
- ^ von Segesser LK, Killer I, Ziswiler M; et al. (1994). "Dissection of the descending thoracic aorta extending into the ascending aorta. A therapeutic challenge". J. Thorac. Cardiovasc. Surg. 108 (4): 755–61. PMID 7934113.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Slater, E.; DeSanctis, RW (1976). "The clinical recognition of dissecting aortic aneurysm". The American Journal of Medicine. 60 (5): 625–33. doi:10.1016/0002-9343(76)90496-4. PMID 1020750.
- ^ Practice Committee of the American Society for Reproductive Medicine (2006). "Increased maternal cardiovascular mortality associated with pregnancy in women with Turner syndrome". Fertil Steril. 86 (5 Suppl): S127–8. doi:10.1016/j.fertnstert.2006.08.082. PMID 17055808.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ von Kodolitsch Y, Nienaber C, Dieckmann C, Schwartz A, Hofmann T, Brekenfeld C, Nicolas V, Berger J, Meinertz T (2004). "Chest radiography for the diagnosis of acute aortic syndrome". Am J Med. 116 (2): 73–7. doi:10.1016/j.amjmed.2003.08.030. PMID 14715319.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Suziku, Toru (1999). "Biochemical diagnosis of aortic dissection: from bench to bedside" (PDF). Japanese Heart Journal. 40 (5): 527–534. doi:10.1536/jhj.40.527. PMID 10888373. Retrieved 2006-07-01.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help); Unknown parameter|month=ignored (help) - ^ Suzuki T, Mehta RR, Ince H, Nagai R, Sakomura Y, Weber F, Sumiyoshi T, Bossone E, Trimarchi S, Cooper J, Smith D, Isselbacher E, Eagle K, Nienaber C (2003). "Clinical profiles and outcomes of acute type B aortic dissection in the current era: lessons from the International Registry of Aortic Dissection (IRAD)". Circulation. 108 (Suppl 1): II312–7. doi:10.1161/01.cir.0000087386.07204.09. PMID 12970252.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Foundation Alexis Carrel
- ^ Nazari S, Salvi S, Aluffi A, Visconti E, Rescigno G, Buniva P (1997). "Prosthesis for aortic arch substitution". Ann Thorac Surg. 64 (5): 1339–44. doi:10.1016/S0003-4975(97)00991-0. PMID 9386701.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Aluffi A, Berti A, Buniva P, Rescigno G, Nazari S (2002). "Improved device for sutureless aortic anastomosis applied in a case of cancer". Tex Heart Inst J. 29 (1): 56–9. PMC 101273. PMID 11995854.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Stefano Nazari: Expandable device type III for easy and reliable approximation of dissection layers in sutureless aortic anastomosis. Ex vivo experimental study. Interact CardioVasc Thorac Surg 2010;10:161-164. doi:10.1510/icvts.2009.216291
- ^ Jeng Wei, Chung-Yi Chang, Yi-Cheng Chuang, Sung-Hou Sue, Kuo-Chen Lee, David Tung (2009): A new vascular ring connector in surgery for aortic dissection. The Journal of Thoracic and Cardiovascular Surgery. 138: 674-7
- ^ a b c Woo KM, Schneider JI (2009). "High-risk chief complaints I: chest pain--the big three". Emerg. Med. Clin. North Am. 27 (4): 685–712, x. doi:10.1016/j.emc.2009.07.007. PMID 19932401.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ [1]
- ^ [2]
- ^ http://en.wikipedia.org/wiki/Death_of_John_Lennon#Murder Death of John Lennon - Wikipedia
