Talk:Fuel cell
| This is the talk page for discussing improvements to the Fuel cell article. This is not a forum for general discussion of the article's subject. |
Article policies
|
| Find sources: Google (books · news · scholar · free images · WP refs) · FENS · JSTOR · TWL |
| Archives: Index, 1, 2, 3, 4Auto-archiving period: 90 days |
| This article has not yet been rated on Wikipedia's content assessment scale. It is of interest to the following WikiProjects: | |||||||||||||||||||||||||||||||||||||
Please add the quality rating to the {{WikiProject banner shell}} template instead of this project banner. See WP:PIQA for details.
Please add the quality rating to the {{WikiProject banner shell}} template instead of this project banner. See WP:PIQA for details.
Please add the quality rating to the {{WikiProject banner shell}} template instead of this project banner. See WP:PIQA for details.
| |||||||||||||||||||||||||||||||||||||
Index
|
||||
|
This page has archives. Sections older than 90 days may be automatically archived by Lowercase sigmabot III when more than 5 sections are present. |
| This is the talk page for discussing improvements to the Fuel cell article. This is not a forum for general discussion of the article's subject. |
Article policies
|
| Find sources: Google (books · news · scholar · free images · WP refs) · FENS · JSTOR · TWL |
| Archives: Index, 1, 2, 3, 4Auto-archiving period: 90 days |
SOFC contradiction
when talking about solid oxide fuel cells the example used with methanol is actually proton exchange membrane fuel cell. I have come to this conclusion because the description suggests that after methanol is catalytically broken up the H+ produced transfers to react with oxygen to create water. Solid oxide fuel cells do not do this. They transfer O2- ions from the oxygen side to react with methanol, as it says both in this article and the main SOFC article: http://en.wikipedia.org/wiki/Solid_oxide_fuel_cell
I would correct myself, but I don't know an appropriate example to replace this glaring error with and I don't want to cause any similar errors.
Efficiency Section
The Efficiency section is a pure copy paste from another source and may be a product of original research, suggest complete rewrite to conform to wikipedia standards. — Preceding unsigned comment added by Daniellis89 (talk • contribs) 22:11, 25 January 2011 (UTC)
- 180.149.16.211 (talk) 11:53, 21 May 2011 (UTC)what is the maximum efficiency of a fuel cell??
I would like to insert the following chart from the Department of Energy's, Energy Efficiency and Renewable Energy Fuel Cell Technology Program as I found that it offered a clear explanation of the different efficiencies of fuel cells, and may help clarify some of the other text in this section I found difficult to get though. Any advice on how best to size it?
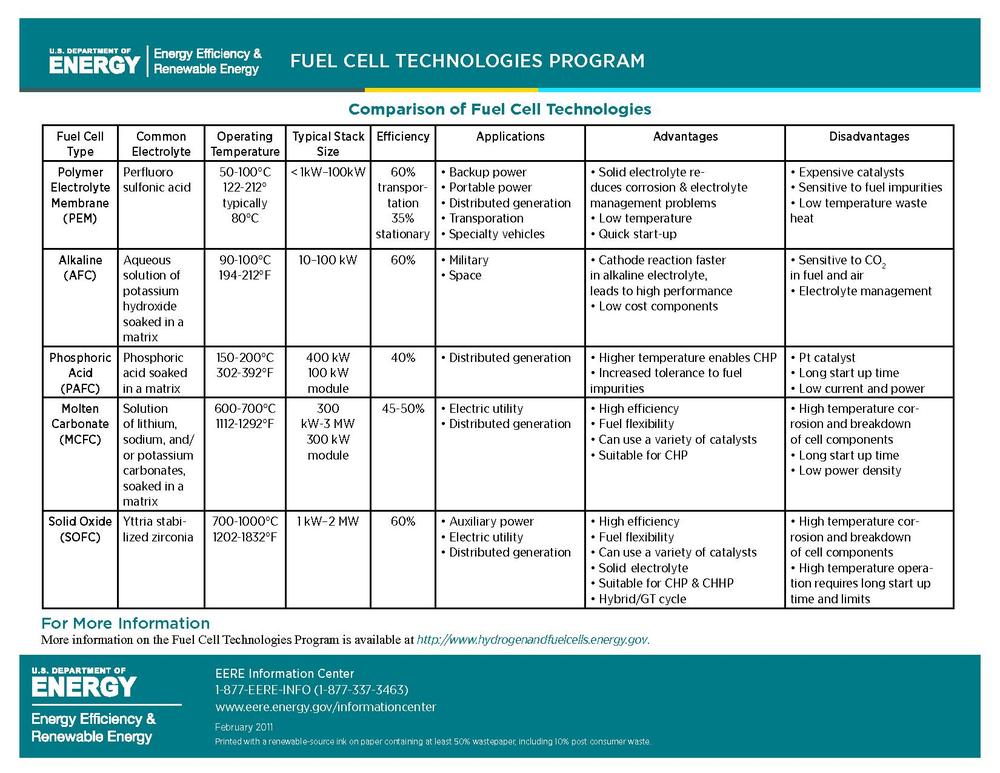
BBfchea 16:51, 1 August 2011 (UTC) — Preceding unsigned comment added by Briannabesch (talk • contribs)
You need a key defining all the terms used in the chart. You can find many of them here: http://www1.eere.energy.gov/hydrogenandfuelcells/glossary.html -- Ssilvers (talk) 23:19, 1 August 2011 (UTC)
- What terms exactly are you thinking of? I'd be happy to do this, but it feels redundant to re-define the types of fuel cell because I found them pretty well defined in the "types of fuel cell" section, and the rest seems pretty self explanatory. I was thinking people would know the term "electrolyte" and "Aqueous", and most of electrolytes are elements and therefore can't really be "defined". Let me know what you found unclear and I would be happy to define it. BBfchea (talk) 21:14, 2 August 2011 (UTC)
- Hi, Briana. Yes, you should include a glossary under the chart showing all the terms used in the chart so that readers do not have to look back and forth to other sections. Most encyclopedia readers have no background at all about fuel cells, and so they need to be able to easily understand the terms used in the chart; they may not realize that these terms are discussed in other sections and not know where to look. Our readers certainly do not know what "electrolyte" and "Aqueous" mean. They are just people who are curious about what a fuel cell is. As I said, you should write as if you are explaining this to a high school science class on the first day of school. -- Ssilvers (talk) 02:57, 3 August 2011 (UTC)
- Alright, I think I got everything that could potentially be confusing in the glossary now- let me know if you see terms I missed. I just considered most of these terms general chemistry, or in some cases every day life, terms so didn't really see the need to define them here, but hopefully the glossary I created will be helpful to those who have absolutely no background in science. I used Merriam-Webster online dictionary definitions for ones not found in the DOE EERE Fuel Cell Technologies program glossary. — Preceding unsigned comment added by Briannabesch (talk • contribs) 13:43, 3 August 2011 (UTC)
Applications Section
I found a lot of out-dated information in this section that I am going to update.
Power Section- The fuel cell market has expanded a lot in the last few years, and I am updated and expanding the information in this section to reflect these developments. Stationary fuel cells have also been used by many different companies in addition to Stuart Island, so I will add in more examples.
Combined Heat and Power- Information is out of date and there are few citations (many are broken or now re-directed), I am updating information and including more recent sources.
Hydrogen transportation and refueling- This title isn't very clear, I will change to Fuel Cell Transportation Vehicles and Hydrogen Refiling,
Land Vehicles- As there are many different types of fuel cell land vehicles I will change title to Fuel Cell Electric Vehicles (FCEVs) as this is the name the industry generally refers to when talking about cars. Most of the information in this section is not relevant to FCEVs and is out of date. I will take out old information and update the section with recent numbers from the Department of Energy. (DOE) There have also been a lot more FCEVs developed in the last few years, I will add more recent developments from major auto manufacturers.
Aircraft, Boats and Submarines- I will add to these section to include more recent developments
Fuel Cell Forklifts- One of the major Fuel Cell applications is Forklifts for the material handling industry, I will create a section that talks about forklifts.
Fueling Stations- A lot of this information is out of date with broken links, I will update and include more up-to-date sources.
Market Structure- I will take this section out of applications and combine it with Fuel Cell Economics as that seems to make more seance.
Briannabesch (talk) 19:31, 22 July 2011 (UTC)
Proton Exchange Membrane Fuel Cells
There are several pieces of information in this section of the Fuel Cell Wikipedia page that I found to be either out of date, not cited, or wrong. I would like to change these facts on the page.
Currently on Wikipedia: "In 2002, typical fuel cell systems cost US$1000 per kilowatt of electric power output." Correct statement: "In 2002 projected improvements in performance and operation on hydrogen led to an estimate of approximately $100/kW for the system cost" Source: http://www1.eere.energy.gov/hydrogenandfuelcells/pdfs/tiax_cost_analysis_pres.pdf
Currently on Wikipedia: "In 2008 UTC Power has 400 kW stationary fuel cells for $1,000,000 per 400 kW installed costs" Correction: UTC does not list their prices, and there are many different payment options, tax incentives, etc. so I don't think we should include a price on the Wikipedia page. It is misleading.
Currently on Wikipedia: "The production costs of the PEM (proton exchange membrane). The Nafion membrane currently costs $566/m²" Correction: In 2005, NREL studies showed that with the average cost of Nafion at $80/lb, a Nafion membrane would cost about $23/m^2. The high end of membrane cost is about $27/m^2. Source: http://www.nrel.gov/hydrogen/pdfs/39104.pdf
LhamillFC (talk) 19:41, 22 July 2011 (UTC)
SOFC
This is a great section, but there are certain areas that need more explanation.
Currently on Wikipedia: "A major disadvantage of the SOFC, as a result of the high heat, is that it 'places considerable constraints on the materials which can be used for interconnections'." The source for this fact is out of date... There is much research being conducted on SOFCs that are dealing with these problems, so a source from 2010 (11 years ago) does not seem like it should be included.
Currently on Wikipedia: "Another disadvantage of running the cell at such a high temperature is that other unwanted reactions may occur inside the fuel cell. It is common for carbon dust (graphite) to build up on the anode, preventing the fuel from reaching the catalyst." Again... there is much research being done on SOFCs including research that addresses this problem. I think that this information: "The anode or fuel side electrode typically is composed of a cermet-containing YSZ and Ni metal. The Ni metal acts as a catalyst for the oxidation of the fuel. Among the problems faced with a Ni cermet anode is Ni metal coarsening (sintering) during use, as well as the buildup of carbon deposits on the surface (coking) during internal reforming of the fuel. Researchers at the University of Pennsylvania have shown that the use of copper (Cu)-based cermets dramatically reduces coking and loss of performance during service due to coking." should be included Source: http://www.ceramicindustry.com/Articles/Feature_Article/10637442bbac7010VgnVCM100000f932a8c0____
Currently on Wikipedia: "Much research is currently being done to find alternatives to YSZ that will carry ions at a lower temperature." There is also research being done to reduce the temperature while still using YSZ. Source: http://www.sciencedirect.com/science/article/pii/S0378775308002243
LhamillFC (talk) 20:21, 22 July 2011 (UTC)
Not only is there research being done to find alternatives to YSZ, but there are also alternatives currently being used in fuel cell systems being sold today. Ceres Power's SOFC that they sell uses CGO (cerium gadolinium oxide) as the electrolyte instead of YSZ, allowing operating temperature to drop to 500-600 degrees C. I would like to add this into the section. LhamillFC (talk) 13:00, 3 August 2011 (UTC)
- I agree that you should update the research in this section, as long as you provide a balanced discussion, noting where there is any disagreement in the major sources. Old statements that are no longer true can be replaced by current information, unless they provide some useful historical information. The fact that a reference is old does not *necessarily* mean that it is useless. -- Ssilvers (talk) 13:22, 3 August 2011 (UTC)
MCFC
This section explains a fundamental aspect of how MCFCs work. I think it makes it seem like a problem that needs to be sorted out "somehow". The DOE has a good explanation of this, and I think it should replace what is currently on Wikipedia.
"Because the electrolyte loses carbonate in the oxidation reaction, the carbonate must be replenished through some means. This is often performed by recirculating the carbon dioxide from the oxidation products into the cathode where it reacts with the incoming air and reforms carbonate."
DOE explanation: "At the anode, hydrogen reacts with the carbonate ions to produce water, carbon dioxide, and electrons. The electrons travel through an external circuit creating electricity and return to the cathode. There, oxygen from the air and carbon dioxide recycled from the anode react with the electrons to form carbonate ions that replenish the electrolyte and provide ionic conduction through the electrolyte, completing the circuit."
Source: http://www.fossil.energy.gov/programs/powersystems/fuelcells/fuelcells_moltencarb.html LhamillFC (talk) 21:00, 22 July 2011 (UTC)
- Can you translate any of this high temperature section into English so that a non-technical person can at least understand the nature of what is being discussed? Otherwise, 99% of readers will simply skip the section. -- Ssilvers (talk) 13:30, 3 August 2011 (UTC)
- Are you talking about the DOE definition I included at the beginning of the section?LhamillFC (talk) 15:04, 3 August 2011 (UTC)
- Yes, because this is something that you added. But in general, if you can translate any of the technical stuff to plainer English, it would be a major improvement to the article. For guidance, see #7 and #8 here. -- Ssilvers (talk) 15:35, 3 August 2011 (UTC)
I agree that the high temperature section is not organized very well, too technical and sometimes even false (see "SOFC contradiction" above). I have rewritten the SOFC section and will post it here for comments.
- Solid oxide fuel cells use a solid material, most commonly a ceramic material called yttria-stabilized zirconia (YSZ), as the electrolyte. Because SOFCs are made entirely of solid materials, they are not limited to the flat plane configuration of other types of fuel cells and are often designed as rolled tubes. They require high operating temperatures (800-1000 degrees Celsius), and can be run on a variety of fuels including natural gas.[1]
- SOFCs are unique in that negatively charged oxygen ions travel from the cathode (negative side of the fuel cell) to the anode (positive side of the fuel cell) as opposed to positively charged hydrogen ions travelling from the anode to the cathode, as is the case in all other types of fuel cells. Oxygen gas (O2) is fed through the cathode where the molecule reacts with electrons to create oxygen ions (O-2). The oxygen ions then travel through the electrolyte to react with hydrogen gas (H2) at the anode. The reaction at the anode produces electricity and water as by-products. Carbon dioxide (CO2) may also be a by-product depending on the fuel, but the carbon emissions from an SOFC system are much less than those from a fossil fuel combustion plant[2]. The chemical reactions for the SOFC system are found below[3]:
- Anode Reaction: 2H2 + 2O–2 => 2H2O + 4e–
- Cathode Reaction: O2 + 4 e– => 2O–2
- Overall Cell Reaction: 2H2 + O2 => 2H2O
- SOFC systems are fuel-flexible, meaning they can run on fuels other than pure hydrogen gas. However, since hydrogen gas is necessary for the reactions listed above, the fuel of choice must contain hydrogen atoms. In order for the fuel cell to operate, the fuel must be reformed to produce pure hydrogen gas. SOFCs are capable of internally reforming light hydrocarbons such as methane (natural gas), propane, and butane. Heavier hydrocarbons including gasoline, diesel, jet fuel and biofuels can serve as fuels in a SOFC, but an external reformer is required.
- Certain challenges exist in SOFC systems due to their high operating temperatures. One such challenge is the potential for carbon dust to build up on the anode, which slows down the internal reforming process. Research addressing this “carbon coking” issue at the University of Pennsylvania has shown that the use of copper-based cermet (heat-resistant materials made of ceramic and metal) dramatically reduces coking and loss of performance during service due to coking.[4]
- Another challenge posed by the high operating temperature is slow start up time for SOFC systems, making SOFCs not ideal for mobile applications. The high operating temperature is largely due to the physical properties of the YSZ electrolyte. As temperature decreases, so does the ionic conductivity of YSZ. Therefore, to obtain optimum performance of the fuel cell, a high operating temperature is required.
- Ceres Power, UK SOFC fuel cell manufacturer, discovered a way to reduce the operating temperature of their SOFC system to 500-600 degrees Celsius. They replaced the commonly used YSZ electrolyte with a CGO (cerium gadolinium oxide) electrolyte. The lower operating temperature allows them to use stainless steel instead of ceramic as the cell substrate, which reduces cost and start up time of the system.[5]
- Although there is the disadvantage of slow start up time, a high operating temperature does provide advantages as well. The high temperature removes the need for a precious metal catalyst like platinum, thereby reducing cost. Additionally, waste heat from SOFC systems may be captured and reused, increasing the overall efficiency to 80%-85%.[6]
LhamillFC (talk) 14:05, 4 August 2011 (UTC)
Socking in this discussion
I have blocked LhamillFC (talk · contribs), Briannabesch (talk · contribs), and Pfchea (talk · contribs) as confirmed socks of Connordfc (talk · contribs). Keegan (talk) 07:43, 28 July 2011 (UTC)
- After discussion by email I have unblocked the accounts. They are individuals working from the same place. They are conditionally unblocked that they follow WP:COI and refrain from discussing changes to fuel cell related articles with each other and use the talk page process to work with other editors. WP:AGF. Keegan (talk) 21:09, 29 July 2011 (UTC)
Market Structure and Economics
Hi all, I wanted to to combine the "Market Structure" section under "Fuel Cell Applications" with the Economics section as that seemed to fit better under economics then applications, and then would change the "economics" title to "Fuel Cell Markets and Economics". The first sentence under the current "Market Structure" section is not cited, and the second has a broken link, I would like to take this sentence out unless someone ells knows where this information can be found and cited (let me know and I'll put it in!). I felt the second sentence of the "Economics" section, "A fuel cell and electric motor combination is not directly limited by the Carnot efficiency of an internal combustion engine." should be taken out as this point is addressed in the "efficiency" section of the page. I also wanted to add in more infomration about the current fuel cell market around the world, so the first paragraph would then read:
"In 2010, fuel cell industry revenues exceeded a $750 million market value[7] and 0.14 million unit shipments, with a average annual growth rate of 115%. [8] Aproximately 50% of fuel cell shipments in 2010 were stationary fuel cells, up from about a third in 2009.[9] The "Big Four" players in the Fuel Cell Industry remain the United States, Germany, Japan and South Korea.[10] Current stationary fuel cells can generate power at approximately $724 to $775 per kilowatt installed.[11] Translated to a consumer, this means stationary fuel cells can generate power at 9-11 cents per kilowatt-hour, including the price of fuel, maintenance, and hardware.[12] A typical stationary fuel cell will meet its return on investment in 3-5 years.[13] Hydrogen is a candidate as a storage mechanism and can help ease the integration of renewable energy generation into our existing grid. We can produce hydrogen though distributed electrolysis generation wherever and whenever excess electricity is produced. This hydrogen can then be distributed to where it is needed, to be turned back into electricity to meet peak demand or even power FCEVs. In this way hydrogen becomes a keystone in the creation of an alternative energy future and a hydrogen economy.
I'll leave this up here for a day before changing- let me know what you think!BBfchea 16:21, 1 August 2011 (UTC) — Preceding unsigned comment added by Briannabesch (talk • contribs)
- I changed the heading for you and deleted the old "markets" section, as you suggested. Headings do not need to repeat the name of the article (Fuel Cell). Also, headings should start with an initial capital letter, but the other words in the heading should not be capitalized, unless it is a proper name. In your refs, you are still missing a lot of necessary bibliographical information. You need to give the author's name (where available), and always give the title of the article (put the url before the title in brackets like this:[url "Title"]) and the name of the publisher (and a page number if from a book or long article). Then give the date published (if available) and the accessdate. The <ref> tag should follow the punctuation with no space. In general, we should write as if we are explaining our subject to an intelligent person with no background in chemistry, engineering or investing, who does not know what a fuel cell is. Another idea is to pretend you are a high school science teacher or economics teacher, as you write, explaining this technology on the first day of school.
- Some comments on your suggested paragraph above: "...2010 revenues exceeding $750 million and 0.14 million unit shipments, with a average annual growth rate of 115%". This sentence is not meaningful. Does it mean for mobile applications, stationary applications, or both? Worldwide? What is a "unit shipment"? You need to give context, since we are writing for a general readership encyclopedia. Also, Pike Research is a marketing firm. So, its bias is to try to raise money for whatever technology it is writing about. So this is not a "neutral" source, and we need to introduce their conclusions with something like, "According to Pike Research, a market research and consulting firm engaged by the fuel cell industry to conduct a market study...." You wrote above: "...stationary fuel cells can generate..." Don't tell us what fuel cells "can do", tell us what they actually do in the field, and back up your assertion with a citation to an independent WP:Reliable source that states clearly the fact and its context, such as how they tested that info. Bloom Energy is not a WP:Reliable source, since it is a commercial supplier advertising its product. We should not refer to company websites for assertions about the performance of those companys' products. You need to use independent (preferably peer reviewed) studies. If we must refer to a company's website for anything, we have to say that "according to Bloom Energy, which sells stationary fuel cells", or something like that. Look at this sentence: "We can produce hydrogen though distributed electrolysis generation wherever and whenever excess electricity is produced." Who is "we"? This begs several questions: What is the cost of "distributed electrolysis generation" compared with hydrogen reforming? How much CO2 does each produce directly and indirectly? What does "distributed" mean in that phrase? "Wherever and whenever" is what we call a WP:PEACOCK phrase. What you mean is "where". But where is there "excess electricity"? This is very markety, rather than encyclopedic writing. Moreover, what do these last five unreferenced sentence have to do with markets and economics? They belong elsewhere, in an explanation of what a stationary fuel cell is, and what its purpose is.
- We must explain to our readers what things are and how they work. For an example of a good marketing explanation, see this. More generally, for some examples of good articles about technologies, see, for example: Atomic line filter and Shale oil extraction. Good luck! -- Ssilvers (talk) 18:34, 1 August 2011 (UTC)
Thanks for your comments, two heads are always better than one. Below I have posted the new first paragraph, but I first wanted to address a couple of things SSilvers had said.
-I have now formatted sources to be more consistent with the articles previous sources. Several of the sources do not have authors, but I included as much information as I could find.
-About Pike Research, they are a market research firm, not a marketing firm- their mission statement is
- “Pike Research is an independent market research firm whose goal is to present an objective, unbiased view of market opportunities within its coverage areas. The firm is not beholden to any special interests and is thus able to offer clear, actionable advice to help clients succeed in the industry, unfettered by technology hype, political agendas, or emotional factors that are inherent in cleantech markets.”
I actually found the numbers and facts I cite from Pike Research ($750 million market value, 50% stationary fuel cell shipments, Germany, Japan, USA and South Korea as the countries with the largest market shares) in the paragraph on a number of news sources first, and though it was most responsible to cite the original published study. As such I consider it a WP:Reliable Source.
-I’m sorry about the confusing wording with “can”, I have changed it to reflect that fuel cells do generate power at $724-$775 per kW installed and 9-11 cents per kW consumer price. I also put in that Bloom Energy stationary fuel cells achieve a 3-5 year payback.
-Hydrogen is also being looked at as a storage mechanism, I significantly revised this section to take into account your comments. I can continue to look to update the sources as well. I would also be ok moving this last section to the "application section" if people feel that that would work better.
This is the revised paragraph I would like to put in.
In 2010, fuel cell industry revenues exceeded a $750 million market value worldwide[14]. There were 0.14 million fuel cell stacks shipped globally in 2010, up from 11 thousand shipments in 2007; in 2010 worldwide fuel cell shipments had an annual growth rate of 115%. [15] Approximately 50% of fuel cell shipments in 2010 were stationary fuel cells, up from about a third in 2009.[16] The "Big Four" players in the Fuel Cell Industry remain the United States, Germany, Japan and South Korea.[17] The Department of Energy Solid State Energy Conversion Alliance found that, as of January 2011, stationary fuel cells generated power at approximately $724 to $775 per kilowatt installed.[18] Bloom Energy, a major fuel cell supplier, says its fuel cells will meet a return on investment in 3-5 years, translated to a consumer, this means stationary fuel cells generate power at 9-11 cents per kilowatt-hour, including the price of fuel, maintenance, and hardware.[19] [20]
Another emerging market for hydrogen is a mechanism to store energy, particularly excess energy from intermittent renewable energy sources like solar and wind farms. [21] As most renewable energy sources are intermittent, storage mechanisms must be devised to provide reliable power (see Grid energy storage). The National Renewable Energy Laboratory is conducting the wind-to-hydrogen project: this project is testing ways to use the electricity produced by wind mills or solar panels when electricity demand is low to electrolyze water and form hydrogen. [22] That hydrogen can then be stored, and turned back into electricity via a fuel cell when electricity demand is high.[23] This application is particularly important for places hoping to rely on intermittent renewable power.
BBfchea 21:25, 1 August 2011 (UTC) — Preceding unsigned comment added by Briannabesch (talk • contribs)
As people haven't commented on this in the last 36 hours I'm going to go ahead and add this section in now — Preceding unsigned comment added by Briannabesch (talk • contribs) 13:52, 3 August 2011 (UTC)
- Good work. I cleaned up your references. Remember that the author name (last, first) goes at the beginning. I deleted the paragraph on hydrogen storage, as that did not seem to be about fuel cells. Perhaps it would be useful in some other article. All the best! -- Ssilvers (talk) 14:35, 3 August 2011 (UTC)
Incomplete references
User:Pfchea, User:Connordfc, User:LhamillFC, and User:Briannabesch, thank you for the additions that you made to this entry last week. Please fill out the incomplete references that you have added to this entry with author names, article titles, publisher names, publication dates and, where available, page numbers. I have left notes on some of your talk pages about how to do this, and the relevant guideline is WP:CITE. Please let me know if you need more assistance. Thanks! -- Ssilvers (talk) 15:27, 2 August 2011 (UTC)
- Thanks for notice user:Ssilvers. We are actively working to properly cite any sources we have linked on this page. After reviewing the References section, I found that most of the citations on the page are simply links, not just those included by my colleagues. In addition, there is not much conformity between the References, it seems pretty haphazard for the most part, and some of them are actually dead links. As an additional project, we will work to edit all of the citations on this page so that they are consistent, and active. Thanks for bringing this matter to our attention. Connordfc (talk) 18:03, 2 August 2011 (UTC)
- Looks like you have made good progress. I will check later and clean up any remaining formatting issues. I note that the author name should always go first, with the author's last name. Here is the basic format: Twain, Mark. [url A Connecticut Yankee in King Arthur's Court]. Publisher name, date, page number, ISBN#, access date. Best regards, -- Ssilvers (talk) 19:35, 2 August 2011 (UTC)
- Thanks, I'v almost the rest of the page with all the info I could find. Ssilvers If you wouldn't mind clarifying your comments about defining terms in the chart I proposed inserting under the efficiency section I would appreciate it. BBfchea (talk) 21:19, 2 August 2011 (UTC)
- You guys have done excellent work cleaning up this aricle. May I recomend using
{{cite book}}and{{cite web}}for formatting references. It guarrentees consistency. WP:wikicite makes life easier for you too. Cheers. Stepho talk 23:01, 2 August 2011 (UTC)
- You guys have done excellent work cleaning up this aricle. May I recomend using
- Wow, Brianna. Good work. See above re the chart. BTW, I moved the bus and forklift discussions a bit higher, right under cars. As for ref style, I like book template, but personally, I think the cite web template just makes the refs harder to work with. -- Ssilvers (talk) 03:16, 3 August 2011 (UTC)
Introduction to the article
I found that the introduction to the article was rather confusing and technical. I wrote an alternative that I tried to make more straight foreword and accessible to the common reader. Thoughts?
A fuel cell is a device that produces electricity through an electrochemical process. It converts the chemical energy stored in a fuel into electricity through a chemical reaction with an oxidizing agent, usually oxygen. Hydrogen is the most common fuel, but hydrocarbons such as natural gas and alcohols like methanol are also used. In addition to direct current electricity fuel cells produce water, heat and, depending on the fuel source, sometimes carbon dioxide. Fuel Cells are different from batteries in that they require a constant source of fuel and oxidizing agent to run, but can produce electricity continually for as long as these inputs are supplied.
Welsh PhysicistWilliam Grove developed the first crude fuel cell, based off a concept developed by his fellow scientist and friend Christian Friedrich Schonbein in 1939. The first commercial use of fuel cells was in NASA space programs to generate power for probes, satellites and shuttles. [24] Since then fuel cells have been used in many other applications. Fuel cells are used to power buildings, many different types vehicles, and to charge smaller electronic devices like laptops and smartphones.
There are many types of fuel cells, and they are each classified by the fuel cell’s electrolyte, the substance that allows charges to move within the fuel cell. Fuel cells come in a variety of sizes. Individual fuel cells only produce very small amounts of electricity, so cells are ‘stacked’, or placed in series, to increases the voltage output to meet application’s power generation requirements. BBfchea (talk) 17:18, 3 August 2011 (UTC)
- Good idea. Here is the guideline on introduction sections: WP:LEAD. Basically, the lead should give an overview of the whole article, touching on the most important points that will be discussed below. With a long article like this, it should be three or four paragraphs long. You don't need to repeat refs in the lead, as long as it is just summarizing things that are referenced below. BTW, in Wikipedia, we always use " and ' rather than curly quotes and apostropies. See WP:MOS You made a very good start, and I would simplify it even further, like this:
A fuel cell is a device that converts the chemical energy stored in a fuel into electricity through a chemical reaction with oxygen or another oxidizing agent. Hydrogen is the most common fuel, but hydrocarbons such as natural gas and alcohols like methanol are sometimes used. Fuel Cells are different from batteries in that they require a constant source of fuel and oxygen to run, but they can produce electricity continually for as long as these inputs are supplied. Welsh Physicist William Grove developed the first crude fuel cells in 1839. The first commercial use of fuel cells was in NASA space programs to generate power for probes, satellites and space capsules. Since then, fuel cells have been used in many other applications. Fuel cells are used to power buildings, many different types of vehicles, and to charge smaller electronic devices like laptops and smartphones.
There are many types of fuel cells, which are classified by the fuel cell's electrolyte, the substance that allows charges to move within the fuel cell. Fuel cells come in a variety of sizes. Individual fuel cells only produce very small amounts of electricity, so cells are "stacked", or placed in series, to increases the voltage output to meet application’s power generation requirements. In addition to electricity, fuel cells produce water, heat and, depending on the fuel source, sometimes carbon dioxide, nitrogen dioxide and very small amounts of other emissions.
- As I said, a very good start, and I think you need to add to this a bit more "overview" information summarizing a few more of the high points in the article below. -- Ssilvers (talk) 20:58, 3 August 2011 (UTC)
Efficiency: theory section - Proposed Overhaul
So I am trying to tackle the information that is written in the efficiency section. The current sections of "Theory" and "In Practice" I found difficult to understand, out of date, not well referenced and occasionally factually wrong. I attempted to re-write the Theory section, keeping in as best I could the information that was cited (and the links weren't dead) and making it sound more accessible. This is what I came up with- let me know what you think!
- There are many different ways to measure efficiencies. Any time you convert energy from one form to another you lose some of the energy. An energy efficiency value represents the ratio between useful output energy and total input energy or what percentage of the total energy put in the system that is retained though the energy conversion. In the case of fuel cells, useful output energy is measured in electrical energy produced by the system. Input energy refers to the total energy stored in the fuel. Fuel cells are generally between 40-60% energy efficient. [25] This is significantly higher than some other forms of energy generation. For example, the typical internal combustion engine of a car is only about 25% energy efficient. [26] In combined heat and power (CHP) systems, the heat produced by the fuel cell is captured and put to use, increasing the efficiency of the system to 85-90%. [27]
- As there will be losses when converting one form of energy into another, it is possible to calculate the theoretical maximum efficiency of any type of power generation. While these values are rarely reached in practice, this can be a useful exercise to compare different types of power generation. Under typical operating conditions the maximum theoretical amount of energy efficiency of a fuel cell is 83% (not including any gains from captured waste heat, as is done in heat and power co-generation). [28] This is significantly more than a combustion engine, which has a maximum theoretical efficiency of 58%.[29] While these efficiencies are not usually approached in real life, but high temperature fuel cells (solid oxide fuel cells or molten carbonate fuel cells) can be combined with gas turbines to approach this theoretical limit. The gas turbine captures heat from the fuel cell and turns it into mechanical energy that increases the fuel cell’s operational efficiency. This solution increases total efficiency to an "ultrahigh" level of 70%.[30]
I'll be working on the "in practice" section next BBfchea (talk) 21:09, 3 August 2011 (UTC)
Also: Under the "In practice" section it was posted that "the overall efficiency (electricity to hydrogen and back to electricity) of such plants (known as round-trip efficiency) is between 30 and 50%, depending on conditions." However the source cited- a NASA Study entitled Round Trip Energy Efficiency of NASA Glenn Regenerative Fuel Cell System says "NASA Glenn Research Center (GRC) has recently demonstrated a Polymer Electrolyte Membrane (PEM) based hydrogen/oxygen regenerative fuel cell system (RFCS) that operated for a charge/discharge cycle with round trip efficiency (RTE) greater than 50 percent." There is no mention of 30% anywhere I found- I would propose taking this out unless the 30% can be shown to be taken from elsewhere BBfchea (talk) 21:23, 3 August 2011 (UTC)
- (To clarify I mean just the 30% number, not the whole quote)BBfchea (talk) 12:50, 4 August 2011 (UTC)
- ^ "Types of Fuel Cells". “Department of Energy EERE website, Retrieved on: 2011-08-04.
- ^ Stambouli, A. Boudghene. "Solid oxide fuel cells (SOFCs): a review of an environmentally clean and efficient source of energy". “Renewable and Sustainable Energy Reviews, Vol. 6, Issue 5, Pages 433-455, October 2002.
- ^ >"Solid Oxide Fuel Cell (SOFC)". “FCTec website, Retrieved on: 2011-08-04.
- ^ Hill, Michael. "Ceramic Energy: Material Trends in SOFC Systems". “Ceramic Industry, September 1, 2005.
- ^ "The Ceres Cell". “Ceres Power website, Retrieved on: 2011-08-04.
- ^ "Types of Fuel Cells". “Department of Energy EERE website, Retrieved on: 2011-08-04.
- ^ Pike Research,Fuel Cell Industry is Poised for Major Change and Development in 2011, Published Feb 2nd 2011, accessed August 1 2011, http://www.pikeresearch.com/newsroom/fuel-cell-industry-is-poised-for-major-change-and-development-in-2011
- ^ Global Fuel Cell Market by Technology, Application, Component,Installation, Cost, Geography, Trends and Forecasts (2011 – 2016), May 2011,http://www.marketsandmarkets.com/Market-Reports/fuel-cell-market-348.html, accessed August 1 2011
- ^ Pike Reaserch, Fuel Cell Annual Report 2011, Published 2Q 2011, Karry-Ann Adamson, Ph.D and Clint Wheelock,http://www.pikeresearch.com/wordpress/wp-content/uploads/2011/05/FCAR-11-Executive-Summary.pdf, accessed August 1 2011
- ^ Pike Research, Fuel Cells Annual Report 2011, pg 3, Published Q2 2011,http://www.pikeresearch.com/wordpress/wp-content/uploads/2011/05/FCAR-11-Executive-Summary.pdf, Kerry-Ann Adamson, Ph.D. and Clint Wheelock, Accessed August 1st 2011
- ^ Solid State Energy Conversion Alliance, SECA Cost Reduction, Updated January 31st 2011, Accessed August 1 2011, http://www.fossil.energy.gov/programs/powersystems/fuelcells/fuelcells_seca.html
- ^ Bloom Energy Plays the Subsidy Game Like a Pro, by Eric Wesoff, Published April 13th 2011, Accessed August 1 2011, http://www.wired.com/epicenter/tag/bloom-energy/
- ^ Bloom Energy, Lower & Lock-In Energy Costs, http://bloomenergy.com/benefits/lower-fix-energy-costs/
- ^ http://www.pikeresearch.com/newsroom/fuel-cell-industry-is-poised-for-major-change-and-development-in-2011, Fuel Cell Industry is Poised for Major Change and Development in 2011, Pike Research, Published Feb 2nd 2011, accessed August 1 2011,
- ^ http://www.marketsandmarkets.com/Market-Reports/fuel-cell-market-348.html, Global Fuel Cell Market by Technology, Application, Component, Installation, Cost, Geography, Trends and Forecasts (2011 – 2016), May 2011, Markets and Markets, accessed August 1 2011
- ^ http://www.pikeresearch.com/wordpress/wp-content/uploads/2011/05/FCAR-11-Executive-Summary.pdf, Fuel Cell Annual Report 2011, Published 2Q 2011, Karry-Ann Adamson, Ph.D and Clint Wheelock, Pike Research, accessed August 1 2011
- ^ http://www.pikeresearch.com/wordpress/wp-content/uploads/2011/05/FCAR-11-Executive-Summary.pdf, Fuel Cells Annual Report 2011, pg 3, Kerry-Ann Adamson, Ph.D. and Clint Wheelock, Pike Research, Published Q2 2011, Accessed August 1st 2011
- ^ http://www.fossil.energy.gov/programs/powersystems/fuelcells/fuelcells_seca.html, Solid State Energy Conversion Alliance SECA Cost Reduction section, Updated January 31st 2011, Accessed August 1 2011,
- ^ http://bloomenergy.com/benefits/lower-fix-energy-costs/, Bloom Energy, Lower & Lock-In Energy Costs
- ^ http://www.wired.com/epicenter/tag/bloom-energy/, Bloom Energy Plays the Subsidy Game Like a Pro, Eric Wesoff, Published April 13th 2011, Accessed August 1 2011,
- ^ http://www.areva.com/EN/operations-408/hydrogen-and-fuel-cells.htm, Hydrogen: adapting to the demand for energy, AREVA
- ^ http://www.nrel.gov/hydrogen/proj_wind_hydrogen_video.html, Wind-to-Hydrogen Project Video, National Renewable Energy Labratory
- ^ http://www.nrel.gov/hydrogen/proj_wind_hydrogen.html, NREL, Wind-to-Hydrogen Project,7 July 2011, Accessed August 1 2011
- ^ Fitzgerald, Justin and O'Bryan, Nancy. "Fuel Cells: A Better Energy Source for Earth and Space". NASA. 2-11-2005
- ^ "Comparison of Fuel Cell Technologies". Department of Energy Energy Efficiency and Fuel Cell Technologies Program. February 2011.
- ^ "Fuel Economy: Where The Energy Goes". Department of Energy Energy Effciency and Renewable Energy www.fueleconomy.gov. August 03 2011. Accessed August 3nd 2011.
- ^ Cite error: The named reference
www1.eere.energy.govwas invoked but never defined (see the help page). - ^ "Fuel Cell Efficiency". World Energy Council. 2011
- ^ "Fuel Cell Efficiency". World Energy Council. 2011
- ^ Milewski, J., Miller, A., Badyda, K. "The Control Strategy for High Temperature Fuel Cell Hybrid Systems". The Online Journal on Electronics and Electrical Engineering (OJEEE). Vol. (2)- No. (4). Pg331.


