Nicotinamide mononucleotide
You can help expand this article with text translated from the corresponding article in Japanese. (September 2018) Click [show] for important translation instructions.
|
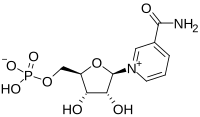
| |
| Names | |
|---|---|
| IUPAC name
3-Carbamoyl-1-[5-O-(hydroxyphosphinato)-β-D-ribofuranosyl]pyridinium
| |
Other names
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChemSpider | |
| ECHA InfoCard | 100.012.851 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C11H15N2O8P | |
| Molar mass | 334.221 g·mol−1 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Nicotinamide mononucleotide ("NMN", "NAMN", and "β-NMN") is a nucleotide derived from ribose and nicotinamide.[1] NMN is made from B vitamins in the body, and is a molecule naturally occurring in all life forms.[2] Like nicotinamide riboside, NMN is a derivative of niacin[3], and humans have enzymes that can use NMN to generate nicotinamide adenine dinucleotide (NADH).[1] In mice, NMN enters cells via the small intestines within 10 minutes converting to NAD+ through the Slc12a8 NMN transporter.[4] Because NADH is a cofactor for processes inside mitochondria, for sirtuins, and for PARP, NMN has been studied in animal models as a potential neuroprotective and anti-aging agent.[5][6] Dietary supplement companies have aggressively marketed NMN products claiming those benefits.[7] Doses of up to 500 mg was shown safe in men in a recent human study[8] at Keio University School of Medicine, Shinjuku, Tokyo Japan. Multiple long-term human studies are underway.[9] [10]
References
- ^ a b Bogan, KL; Brenner, C (2008). "Nicotinic acid, nicotinamide, and nicotinamide riboside: a molecular evaluation of NAD+ precursor vitamins in human nutrition". Annual Review of Nutrition. 28: 115–30. doi:10.1146/annurev.nutr.28.061807.155443. PMID 18429699.
- ^ "What is NMN ?". NMN.com.
{{cite web}}: CS1 maint: url-status (link) - ^ "About NMN (Beta-Nicotinamide Mononucleotide)".
{{cite web}}: CS1 maint: url-status (link) - ^ "Slc12a8 is a nicotinamide mononucleotide transporter". Nature. January 2019.
- ^ Brazill, JM; Li, C; Zhu, Y; Zhai, RG (June 2017). "NMNAT: It's an NAD+ synthase… It's a chaperone… It's a neuroprotector". Current Opinion in Genetics & Development. 44: 156–162. doi:10.1016/j.gde.2017.03.014. PMC 5515290. PMID 28445802.
- ^ "Long-Term Administration of Nicotinamide Mononucleotide Mitigates Age-Associated Physiological Decline in Mice". Cell Metabolism. 13 December 2016.
- ^ Stipp, David (March 11, 2015). "Beyond Resveratrol: The Anti-Aging NAD Fad". Scientific American Blog Network.
- ^ "Effect of oral administration of nicotinamide mononucleotide on clinical parameters and nicotinamide metabolite levels in healthy Japanese men". Endocrine Journal. November 2019.
- ^ "Effect of long-term oral administration of nicotinamide mononucleotide (NMN) on human health".
{{cite web}}: CS1 maint: url-status (link) - ^ "Assessment of the safety of long-term nicotinamide mononucleotide (NMN)".
{{cite web}}: CS1 maint: url-status (link)
