Fosfomycin
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Monuril, Monurol, Ivozfo, others |
| Other names | Phosphomycin, phosphonomycin, fosfomycin tromethamine |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a697008 |
| License data |
|
| Routes of administration | Intravenous, By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Bioavailability | 30–37% (by mouth, fosfomycin tromethamine); varies with food intake |
| Protein binding | Nil |
| Metabolism | Nil |
| Elimination half-life | 5.7 hours (mean) |
| Excretion | Kidney, unchanged |
| Identifiers | |
| |
| CAS Number |
|
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.041.315 |
| Chemical and physical data | |
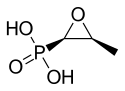
| Formula | C3H7O4P |
| Molar mass | 138.059 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 94 °C (201 °F) |
| |
| |
| (verify) | |
Fosfomycin, sold under the brand name Monurol among others, is an antibiotic primarily used to treat lower urinary tract infections.[8] It is not indicated for kidney infections.[8] Occasionally it is used for prostate infections.[8] It is generally taken by mouth.[8]
Common side effects include diarrhea, nausea, headache, and vaginal yeast infections.[8] Severe side effects may include anaphylaxis and Clostridioides difficile-associated diarrhea.[8] While use during pregnancy has not been found to be harmful, such use is not recommended.[9] A single dose when breastfeeding appears safe.[9] Fosfomycin works by interfering with the production of the bacterial cell wall.[8]
Fosfomycin was discovered in 1969 and approved for medical use in the United States in 1996 [globalize][8][10] It is on the World Health Organization's List of Essential Medicines.[11] The World Health Organization classifies fosfomycin as critically important for human medicine.[12] It is available as a generic medication.[13] It was originally produced by certain types of Streptomyces, although it is now made chemically.[10]
Medical uses
[edit]Fosfomycin is used to treat bladder infections as well as urinary tract infections (UTIs), where it is usually given as a single dose by mouth.[14]
Oral fosfomycin is not recommended for children under 12 years old.[15]
Additional uses have been proposed.[16] The global problem of advancing antimicrobial resistance has led to a renewed interest in its use more recently.[17]
Fosfomycin can be used as an efficacious treatment for both UTIs and complicated UTIs including acute pyelonephritis. The standard regimen for complicated UTIs is an oral 3 g dose administered once every 48 or 72 hours for a total of 3 doses or a 6 g dose every 8 hours for 7–14 days when fosfomycin is given in IV form.[18]
Intravenous fosfomycin is being increasingly used for treating infections caused by multidrug-resistant bacteria, mostly as a partner drug in order to avoid the occurrence of resistances and to take advantage of its synergistic activity with several other antimicrobials. Daily adult dose usually ranges from 12 to 24 grams.[19] When administered in continuous infusion, a loading dose of fosfomycin 8 g followed by a daily dose of 16 g or 24 g. Continuous infusion is suggested in patients with normal renal function.[20]
Bacterial sensitivity
[edit]The fosfomycin molecule has an epoxide or oxirane ring, which is highly strained and thus very reactive.[citation needed]
Fosfomycin has broad antibacterial activity against both Gram-positive and Gram-negative pathogens, with useful activity against E. faecalis, E. coli, and various Gram-negatives such as Citrobacter and Proteus. Given a greater activity in a low-pH milieu, and predominant excretion in active form into the urine, fosfomycin has found use for the prophylaxis and treatment of UTIs caused by these uropathogens. Of note, activity against S. saprophyticus, Klebsiella, and Enterobacter is variable and should be confirmed by minimum inhibitory concentration testing. Activity against extended-spectrum β-lactamase-producing pathogens, notably ESBL-producing E. coli, is good to excellent, because the drug is not affected by cross-resistance issues. Existing clinical data support use in uncomplicated UTIs, caused by susceptible organisms. However, susceptibility break-points of 64 mg/L should not be applied for systemic infections.[citation needed]
Resistance
[edit]Development of bacterial resistance under therapy is a frequent occurrence and makes fosfomycin unsuitable for sustained therapy of severe infections. Mutations that inactivate the nonessential glycerophosphate transporter render bacteria resistant to fosfomycin.[21][22][23] Still, fosfomycin can be used to treat MRSA bacteremia.[24]
Prescribing fosfomycin together with at least another active drug reduces the risk of developing bacterial resistance. Fosfomycin acts synergistically with many other antibiotics, including aminoglycosides, carbapenems, cephalosporins, daptomycin and oritavancin.[19][25]
Enzymes conferring resistance to fosfomycin have also been identified and are encoded both chromosomally and on plasmids.[26]
Three related fosfomycin resistance enzymes (named FosA, FosB, and FosX) are members of the glyoxalase superfamily. These enzymes function by nucleophilic attack on carbon 1 of fosfomycin, which opens the epoxide ring and renders the drug ineffective.[citation needed]
The enzymes differ by the identity of the nucleophile used in the reaction: glutathione for FosA, bacillithiol for FosB,[27][28] and water for FosX.[26]
In general, FosA and FosX enzymes are produced by Gram-negative bacteria, whereas FosB is produced by Gram-positive bacteria.[26]
FosC uses ATP and adds a phosphate group to fosfomycin, thus altering its properties and making the drug ineffective.[29]
Side effects
[edit]The drug is well tolerated and has a low incidence of harmful side effects.[14]
Mechanism of action
[edit]Despite its name (ending in -omycin) Fosfomycin is not a macrolide, but a member of a novel class of phosphonic antibiotics. Fosfomycin is bactericidal and inhibits bacterial cell wall biogenesis by inactivating the enzyme UDP-N-acetylglucosamine-3-enolpyruvyltransferase, also known as MurA.[30] This enzyme catalyzes the committed step in peptidoglycan biosynthesis, namely the ligation of phosphoenolpyruvate (PEP) to the 3'-hydroxyl group of UDP-N-acetylglucosamine. This pyruvate moiety provides the linker that bridges the glycan and peptide portion of peptidoglycan. Fosfomycin is a PEP analog that inhibits MurA by alkylating an active site cysteine residue (Cys 115 in the Escherichia coli enzyme).[31][32]
Fosfomycin enters the bacterial cell through the glycerophosphate transporter.[33]
History
[edit]Fosfomycin (originally known as phosphonomycin) was discovered in a joint effort of Merck and Co. and Spain's Compañía Española de Penicilina y Antibióticos (CEPA). It was first isolated by screening broth cultures of Streptomyces fradiae isolated from soil samples for the ability to cause formation of spheroplasts by growing bacteria. The discovery was described in a series of papers published in 1969.[34] CEPA began producing fosfomycin on an industrial scale in 1971 at its Aranjuez facility.[35]
Biosynthesis
[edit]The complete fosfomycin biosynthetic gene cluster from Streptomyces fradiae has been cloned and sequenced and the heterologous production of fosfomycin in S. lividans has been achieved by Ryan Woodyer of the Huimin Zhao and Wilfred van der Donk research groups.[36]
Synthetic manufacture
[edit]Large scale production of fosfomycin is achieved by making an epoxide of cis-propenylphosphonic acid to yield racemic mixture fosfomycin.[37]
References
[edit]- ^ "Prescription medicines: registration of new chemical entities in Australia, 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Archived from the original on 10 April 2023. Retrieved 9 April 2023.
- ^ "Prescription medicines and biologicals: TGA annual summary 2017". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 31 March 2024.
- ^ https://www.tga.gov.au/resources/prescription-medicines-registrations/cipfosin-cipla-fosfomycin-cipla-australia-pty-ltd
- ^ "Regulatory Decision Summary - Ivozfo". Health Canada. 23 October 2014. Archived from the original on 7 June 2022. Retrieved 7 June 2022.
- ^ "Monuril 3g granules for oral solution - Summary of Product Characteristics (SmPC)". (emc). 1 June 2021. Archived from the original on 8 March 2022. Retrieved 7 June 2022.
- ^ "Fomicyt 40 mg/mL powder for solution for infusion - Summary of Product Characteristics (SmPC)". (emc). 11 February 2021. Archived from the original on 7 June 2022. Retrieved 7 June 2022.
- ^ "Monurol- fosfomycin tromethamine powder". DailyMed. 24 October 2019. Archived from the original on 7 June 2022. Retrieved 7 June 2022.
- ^ a b c d e f g h "Fosfomycin Tromethamine Monograph for Professionals". Drugs.com. Archived from the original on 29 October 2019. Retrieved 29 October 2019.
- ^ a b "Fosfomycin (Monurol) Use During Pregnancy". Drugs.com. Archived from the original on 29 October 2019. Retrieved 29 October 2019.
- ^ a b Finch RG, Greenwood D, Whitley RJ, Norrby SR (2010). Antibiotic and Chemotherapy E-Book. Elsevier Health Sciences. p. 259. ISBN 9780702047657.
- ^ World Health Organization (2019). World Health Organization model list of essential medicines: 21st list 2019. Geneva: World Health Organization. hdl:10665/325771. WHO/MVP/EMP/IAU/2019.06. License: CC BY-NC-SA 3.0 IGO.
- ^ World Health Organization (2019). Critically important antimicrobials for human medicine (6th revision ed.). Geneva: World Health Organization. hdl:10665/312266. ISBN 9789241515528.
- ^ British national formulary : BNF 76 (76 ed.). Pharmaceutical Press. 2018. pp. 560–561. ISBN 9780857113382.
- ^ a b Patel SS, Balfour JA, Bryson HM (April 1997). "Fosfomycin tromethamine. A review of its antibacterial activity, pharmacokinetic properties and therapeutic efficacy as a single-dose oral treatment for acute uncomplicated lower urinary tract infections". Drugs. 53 (4): 637–656. doi:10.2165/00003495-199753040-00007. PMID 9098664. S2CID 46972404.
- ^ "MONURIL SACHETS 3G". Archived from the original on 28 May 2014. Retrieved 26 May 2014.
- ^ Falagas ME, Giannopoulou KP, Kokolakis GN, Rafailidis PI (April 2008). "Fosfomycin: use beyond urinary tract and gastrointestinal infections". Clinical Infectious Diseases. 46 (7): 1069–1077. doi:10.1086/527442. PMID 18444827.
- ^ Falagas ME, Grammatikos AP, Michalopoulos A (October 2008). "Potential of old-generation antibiotics to address current need for new antibiotics". Expert Review of Anti-Infective Therapy. 6 (5): 593–600. doi:10.1586/14787210.6.5.593. PMID 18847400. S2CID 13158593.
- ^ Zhanel GG, Zhanel MA, Karlowsky JA (28 March 2020). "Oral and Intravenous Fosfomycin for the Treatment of Complicated Urinary Tract Infections". The Canadian Journal of Infectious Diseases & Medical Microbiology. 2020. Hindawi Limited: 8513405. doi:10.1155/2020/8513405. PMC 7142339. PMID 32300381.
- ^ a b Antonello RM, Principe L, Maraolo AE, Viaggi V, Pol R, Fabbiani M, et al. (August 2020). "Fosfomycin as Partner Drug for Systemic Infection Management. A Systematic Review of Its Synergistic Properties from In Vitro and In Vivo Studies". Antibiotics. 9 (8): 500. doi:10.3390/antibiotics9080500. PMC 7460049. PMID 32785114.
- ^ Antonello RM, Di Bella S, Maraolo AE, Luzzati R (June 2021). "Fosfomycin in continuous or prolonged infusion for systemic bacterial infections: a systematic review of its dosing regimen proposal from in vitro, in vivo and clinical studies". European Journal of Clinical Microbiology & Infectious Diseases. 40 (6): 1117–1126. doi:10.1007/s10096-021-04181-x. PMC 8139892. PMID 33604721.
- ^ Kahan FM, Kahan JS, Cassidy PJ, Kropp H (May 1974). "The mechanism of action of fosfomycin (phosphonomycin)". Annals of the New York Academy of Sciences. 235 (1): 364–386. Bibcode:1974NYASA.235..364K. doi:10.1111/j.1749-6632.1974.tb43277.x. PMID 4605290. S2CID 33013586.
- ^ Castañeda-García A, Blázquez J, Rodríguez-Rojas A (April 2013). "Molecular Mechanisms and Clinical Impact of Acquired and Intrinsic Fosfomycin Resistance". Antibiotics. 2 (2): 217–236. doi:10.3390/antibiotics2020217. PMC 4790336. PMID 27029300.
- ^ Omori K, Kitagawa H, Takada M, Maeda R, Nomura T, Kubo Y, Shigemoto N, Ohge H (April 2024). "Fosfomycin as salvage therapy for persistent methicillin-resistant Staphylococcus aureus bacteremia: A case series and review of the literature". J Infect Chemother. 30 (4): 352–356. doi:10.1016/j.jiac.2023.10.024. PMID 37922987.
- ^ Lagatolla C, Mehat JW, La Ragione RM, Luzzati R, Di Bella S (September 2022). "In Vitro and In Vivo Studies of Oritavancin and Fosfomycin Synergism against Vancomycin-Resistant Enterococcus faecium". Antibiotics. 11 (10): 1334. doi:10.3390/antibiotics11101334. PMC 9598191. PMID 36289992.
- ^ a b c Rigsby RE, Fillgrove KL, Beihoffer LA, Armstrong RN (2005). "Fosfomycin resistance proteins: a nexus of glutathione transferases and epoxide hydrolases in a metalloenzyme superfamily". Gluthione [sic] Transferases and Gamma-Glutamyl Transpeptidases. Methods in Enzymology. Vol. 401. pp. 367–379. doi:10.1016/S0076-6879(05)01023-2. ISBN 9780121828066. PMID 16399398.
- ^ Sharma SV, Jothivasan VK, Newton GL, Upton H, Wakabayashi JI, Kane MG, et al. (July 2011). "Chemical and Chemoenzymatic syntheses of bacillithiol: a unique low-molecular-weight thiol amongst low G + C Gram-positive bacteria". Angewandte Chemie. 50 (31): 7101–7104. doi:10.1002/anie.201100196. PMID 21751306.
- ^ Roberts AA, Sharma SV, Strankman AW, Duran SR, Rawat M, Hamilton CJ (April 2013). "Mechanistic studies of FosB: a divalent-metal-dependent bacillithiol-S-transferase that mediates fosfomycin resistance in Staphylococcus aureus". The Biochemical Journal. 451 (1): 69–79. doi:10.1042/BJ20121541. PMC 3960972. PMID 23256780.
- ^ García P, Arca P, Evaristo Suárez J (July 1995). "Product of fosC, a gene from Pseudomonas syringae, mediates fosfomycin resistance by using ATP as cosubstrate". Antimicrobial Agents and Chemotherapy. 39 (7): 1569–1573. doi:10.1128/aac.39.7.1569. PMC 162783. PMID 7492106.
- ^ Brown ED, Vivas EI, Walsh CT, Kolter R (July 1995). "MurA (MurZ), the enzyme that catalyzes the first committed step in peptidoglycan biosynthesis, is essential in Escherichia coli". Journal of Bacteriology. 177 (14): 4194–4197. doi:10.1128/jb.177.14.4194-4197.1995. PMC 177162. PMID 7608103.
- ^ Zhu JY, Yang Y, Han H, Betzi S, Olesen SH, Marsilio F, Schönbrunn E (April 2012). "Functional consequence of covalent reaction of phosphoenolpyruvate with UDP-N-acetylglucosamine 1-carboxyvinyltransferase (MurA)". The Journal of Biological Chemistry. 287 (16): 12657–12667. doi:10.1074/jbc.M112.342725. PMC 3339971. PMID 22378791.
- ^ Krekel F, Samland AK, Macheroux P, Amrhein N, Evans JN (October 2000). "Determination of the pKa value of C115 in MurA (UDP-N-acetylglucosamine enolpyruvyltransferase) from Enterobacter cloacae". Biochemistry. 39 (41): 12671–12677. doi:10.1021/bi001310x. PMID 11027147.
- ^ Santoro A, Cappello AR, Madeo M, Martello E, Iacopetta D, Dolce V (December 2011). "Interaction of fosfomycin with the glycerol 3-phosphate transporter of Escherichia coli". Biochimica et Biophysica Acta (BBA) - General Subjects. 1810 (12): 1323–1329. doi:10.1016/j.bbagen.2011.07.006. PMID 21791237.
- ^ Silver LL (2011). "Rational Approaches to Antibacterial Discovery: Pre-Genomic Directed and Phenotypic Screening". In Dougherty T, Pucci MJ (eds.). Antibiotic Discovery and Development. Springer. p. 46. doi:10.1007/978-1-4614-1400-1_2. ISBN 978-1-4614-1400-1.
- ^ "About us: Our history". Encros. Archived from the original on 14 September 2011.
- ^ Woodyer RD, Shao Z, Thomas PM, Kelleher NL, Blodgett JA, Metcalf WW, et al. (November 2006). "Heterologous production of fosfomycin and identification of the minimal biosynthetic gene cluster". Chemistry & Biology. 13 (11): 1171–1182. doi:10.1016/j.chembiol.2006.09.007. PMID 17113999.
- ^ Marocco CP, Davis EV, Finnell JE, Nguyen PH, Mateer SC, Ghiviriga I, et al. (2011). "Asymmetric synthesis of (−)-fosfomycin and its trans-(1S,2S)-diastereomer using a biocatalytic reduction as the key step". Tetrahedron: Asymmetry. 22 (18–19). Elsevier BV: 1784–1789. doi:10.1016/j.tetasy.2011.10.009. ISSN 0957-4166.
