Hepatitis B virus DNA polymerase
| Protein P (DNA polymerase / RNase H) | |||||||
|---|---|---|---|---|---|---|---|
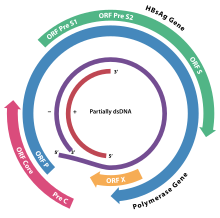 The genome organisation of HBV; the genes overlap. ORF P, in blue, encodes Hepatitis B virus DNA polymerase. | |||||||
| Identifiers | |||||||
| Organism | |||||||
| Symbol | P | ||||||
| UniProt | P03156 | ||||||
| |||||||
| Protein P | |
|---|---|
| Identifiers | |
| Symbol | ? |
| InterPro | IPR037531 |
Hepatitis B virus DNA polymerase is a hepatitis B viral protein.[1][2] It is a DNA polymerase that can use either DNA or RNA templates and a ribonuclease H that cuts RNA in the duplex. Both functions are supplied by the reverse transcriptase (RT) domain.
Structure
[edit]The hepadnaviral P protein is organized into three domains: an N-terminal domain (InterPro: IPR000201) covering the terminal and the spacer, an RT domain related to every other reverse transcriptase domain, and a C-terminal domain (InterPro: IPR001462) regulating the RNase H activity.
Furthermore, hepadnavirus polymerases contain a terminal protein (TP) domain that contains a tyrosine residue that serves as a primer for the synthesis of the (-) DNA strand.[3]
Function
[edit]The Hepatitis B virus (HBV) polymerase is a multifunctional enzyme, with both RNA-dependent and DNA-dependent polymerase functions, as well as an RNase H function. It acts on the HBV pre-genomic RNA (pgRNA) to reverse transcribe it to form a new rcDNA molecule within a new capsid. (The pgRNA has another function of being translated into the viral polymerase and core proteins).[3]
HBV core protein dimers are required for packaging of the pgRNA/polymerase complex. Then, after viral polymerase binds to the packaging signal (Hɛ) found at the 5′ end of the pgRNA, they are incorporated into the viral capsid.[3][4]
Inside the capsid, the pgRNA undergoes reverse transcription, which is initiated by protein priming at the tyrosine residue of the HBV polymerase. Thus, the (-) DNA strand is made.[4] At the same time, the RNA template is degraded by the RNase H activity of the polymerase. A short RNA of about 15–18 nucleotides at the 5′ end of the pgRNA (including the 5′ DR1 sequence) is not degraded and it is used as primer for (+) DNA strand synthesis.[3]
The resulting RC-DNA is partially double stranded. The (-) DNA strand is longer than a genome length, with a covalently bound polymerase and a redundant flap at the 5′ end. However, the (+) DNA strand synthesis is uncompleted by the polymerase, and there is a gap exists down to the 3′ end of the (+) DNA strand.[4]
References
[edit]- ^ Ono-Nita SK, Kato N, Shiratori Y, Masaki T, Lan KH, Carrilho FJ, Omata M (March 1999). "YMDD motif in hepatitis B virus DNA polymerase influences on replication and lamivudine resistance: A study by in vitro full-length viral DNA transfection". Hepatology. 29 (3): 939–45. doi:10.1002/hep.510290340. PMID 10051501.
- ^ Shaw T, Mok SS, Locarnini SA (November 1996). "Inhibition of hepatitis B virus DNA polymerase by enantiomers of penciclovir triphosphate and metabolic basis for selective inhibition of HBV replication by penciclovir". Hepatology. 24 (5): 996–1002. doi:10.1002/hep.510240504. PMID 8903366.
- ^ a b c d Menéndez-Arias L, Álvarez M, Pacheco B (October 2014). "Nucleoside/nucleotide analog inhibitors of hepatitis B virus polymerase: mechanism of action and resistance". Current Opinion in Virology. 8: 1–9. doi:10.1016/j.coviro.2014.04.005. PMID 24814823.
- ^ a b c Yang HC, Kao JH (September 2014). "Persistence of hepatitis B virus covalently closed circular DNA in hepatocytes: molecular mechanisms and clinical significance". Emerging Microbes & Infections. 3 (9): e64. doi:10.1038/emi.2014.64. PMC 4185362. PMID 26038757.
