Talk:Conjugate (acid-base theory)
| This It is of interest to the following WikiProjects: | |||||||||||
| |||||||||||
| On 3 March 2022, it was proposed that this article be moved from Conjugate acid to Conjugate (acid-base theory). The result of the discussion was moved. |
Errors?
[edit]Ammonia is a weak base, not 'relatively strong'! Also, strong acids have weak conj base, but it is NOT necessarily the case for the reverse: weak acids typically have WEAK conjugate bases as well. Think acetic acid. Acetate is a weak conjugate base. — Preceding unsigned comment added by 98.255.225.219 (talk) 06:30, 7 July 2015 (UTC)
I think the second sentence from the top starting with: 'A conjugate acid ...backward..' should read: 'A conjugate acid ...forward..'. 124.150.101.241 (talk) 22:31, 12 March 2009 (UTC)
- Corrected.--E8 (talk) 01:39, 13 March 2009 (UTC)
Hi, I'm not entirely sure, but it seems like the listing HFSbF5 Fluoroantimonic acid---SbF6− Hexafluoroantimonate ion in the base-acid table is placed in the wrong place. Shouldn't it be on top? Thanks. 89.138.43.49 (talk) 00:58, 6 March 2009 (UTC)
- I think you're correct, as Fluoroantimonic acid is a super acid. I'll make the appropriate change.--E8 (talk) 01:39, 13 March 2009 (UTC)
The equation shown in this article would only apply for a strong acid. A weak acid should have two-way arrows to indicate equilibrium. Fuzzform 01:38, 19 April 2007 (UTC)
- Agreed. I made a modification. Is this acceptable as general form, or would a case-by-case listing be necessary? Examples, perhaps?--E8 05:21, 27 April 2007 (UTC)
The section where it says that conjugate base of weak acids is weak appears to be wrong. Two reasons: kw divided by a smaller number will be a larger number than if it were divided by larger number. 2> Conjugate base of strong acid is weak (true). But if conjugate base of weak acids were to be also weak then it follows that conjugate bases are always weak which seems absurd. Jkulkarni (talk) 05:02, 22 April 2018 (UTC)
The article name should be a conjugate acid and base Sushreesssss (talk) 04:25, 3 September 2019 (UTC)
There's no "Conjugate Salt" page or even reference to it here. I'd be happy to contribute to such a page or section.--E8 03:02, 26 April 2007 (UTC)
Ah guys, the conjugate base of a weak acid is not a weak base... That's simply wrong.
- True, but this has not been stated or implied. The article explicitly states: "The conjugate base of a weak acid is a strong base, and the conjugate base of a strong acid is a weak base, and vice versa." The article is clearly incomplete. If you would like to do a case-by-case analysis of conjugates, please do.--E8 17:31, 4 May 2007 (UTC)
- The conjugate base of a weak acid is not a strong base, as in the case of NH4+. It is only a strong base if the Ka value is very small. —Preceding unsigned comment added by 165.196.222.154 (talk) 21:42, 17 June 2009 (UTC)
Misc
[edit]Hi I'm currently studying for my IGCSE chemistry exam, I was trying to find out what Conjugate acids and bases were and this page seemed to offer the best explanation, but I'm still completely lost could someone please put up an explanation in simpler terms.
- Pick up a copy of an appropriate-level general chemistry textbook from a local library. Most will have a chapter dedicated to the concept of "conjugate." I do think this article requires a bit of attention; I'll try to set aside some time to add more substance and clarity to it.--E8 21:40, 8 October 2007 (UTC)
Ordering of Acids
[edit]Hi, just glanced at this page, and thought the ordering of your examples was a little strange... failed to notice it was ordered by pKa, so justifiably the changes were reverted. However, on more prolonged examination of the page, it became clear there is little to explain the concept of protonation/deprotonation and the factors affecting these.
"Conjugate Acid" being the title of the page seems odd, since conjugate bases are just as important, so the page should be entitled "Conjugate acid/base pairs".
The discussion of the theory behind this equilibrium is to be found on the acid dissociation constant page, so this page would perhaps serve better as an introduction to, and definition of, the topic. In light of this, I would suggest adding some more protonated species as examples of conjugate acids, rather than purely showing the technicalities of neutral acids being 'conjugate' to their anions.
163.1.236.202 (talk) 21:32, 29 May 2011 (UTC)
Strength of conjugates
[edit]This section needs to be worded better. The reaction between ammonia and water does not “proceed until most of the ammonia has been transformed into ammonium”, its Kb says this is not the case at all. Ammonium’s conjugate base is stronger than the hydroxide ion? What?! 2401:7000:B27A:CC00:D82:16A1:E238:2DDE (talk) 13:06, 23 May 2020 (UTC)
Diagram is unconventional .... and who are "we"?
[edit]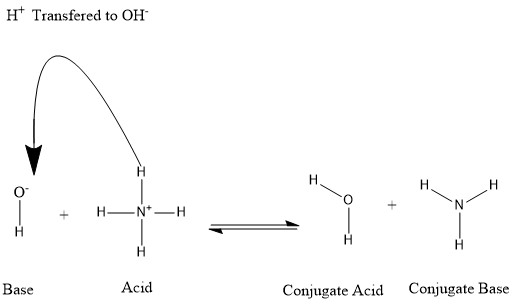
The diagram in this article is unconventional in having the curly arrow from proton to oxyanion, rather than the other way round. Can someone fix this with a new (preferably .svg) version? Also, that portion of the article uses "we" in several places, which AFAIK is inappropriate style for WP so rewording is needed when the new image is added. Michael D. Turnbull (talk) 16:52, 4 October 2020 (UTC)
 Done Now my .svg skills have improved, I've made these changes myself.... Mike Turnbull (talk) 12:22, 4 March 2022 (UTC)
Done Now my .svg skills have improved, I've made these changes myself.... Mike Turnbull (talk) 12:22, 4 March 2022 (UTC)
- Conjugate is spelled wrong both times in the image. I don't know how to fix this so I'm bringing it up so someone else can. 2600:1009:B100:7919:5C08:7A31:2E49:A1F9 (talk) 16:33, 13 April 2023 (UTC)
- Thanks for pointing this out! I've fixed it. Mike Turnbull (talk) 08:29, 14 April 2023 (UTC)
- Conjugate is spelled wrong both times in the image. I don't know how to fix this so I'm bringing it up so someone else can. 2600:1009:B100:7919:5C08:7A31:2E49:A1F9 (talk) 16:33, 13 April 2023 (UTC)
Requested move 3 March 2022
[edit]- The following is a closed discussion of a requested move. Please do not modify it. Subsequent comments should be made in a new section on the talk page. Editors desiring to contest the closing decision should consider a move review after discussing it on the closer's talk page. No further edits should be made to this discussion.
The result of the move request was: moved. (closed by non-admin page mover) Vpab15 (talk) 23:13, 11 March 2022 (UTC)
Conjugate acid → Conjugate (acid-base theory) – Rather than arbitrarily choosing one of the pair of terms for the article title in violation of WP:AND, we should use the root term as the article title, disambiguated from other meanings with parenthetical disambiguation. Both Conjugate acid and Conjugate base will of course redirect to the page. An alternative would be Conjugate acid and conjugate base, but that seems unwieldy and not in line with the spirt of WP:AND. Mdewman6 (talk) 22:57, 3 March 2022 (UTC)
- Support move. Seems uncontroversial to me. Need to be careful with redirects because a conjugated system is completely different chemistry. Mike Turnbull (talk) 12:33, 4 March 2022 (UTC)
- Support but a better choice would be Conjugate acids and bases. Crossover1370 (talk | contribs) 16:00, 10 March 2022 (UTC)
- That may be a good alternative, except that it's plural and doesn't jive with WP:SINGULAR. The singular version doesn't work, and I don't think you can make a case for the plural per any of the criteria at WP:PLURAL. Mdewman6 (talk) 21:08, 10 March 2022 (UTC)
- Then Conjugate (acid-base theory) would be the best option. Crossover1370 (talk | contribs) 16:45, 11 March 2022 (UTC)
- That may be a good alternative, except that it's plural and doesn't jive with WP:SINGULAR. The singular version doesn't work, and I don't think you can make a case for the plural per any of the criteria at WP:PLURAL. Mdewman6 (talk) 21:08, 10 March 2022 (UTC)

