Doxapram
 | |
| Clinical data | |
|---|---|
| Routes of administration | Intravenous |
| ATC code | |
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.005.653 |
| Chemical and physical data | |
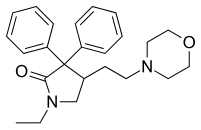
| Formula | C24H30N2O2 |
| Molar mass | 378.516 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| (verify) | |
Doxapram hydrochloride (marketed as Dopram, Stimulex or Respiram) is a respiratory stimulant, or analeptic. Administered intravenously, doxapram stimulates an increase in tidal volume, and respiratory rate.
Mechanism of action
[edit]
Doxapram stimulates chemoreceptors in the carotid bodies of the carotid arteries, which in turn, stimulates the respiratory center in the brainstem.
Appearance
[edit]Doxapram is a white to off-white, odorless, crystalline powder that is stable in light and air. It is soluble in water, sparingly soluble in alcohol and practically insoluble in ether. Injectable products have a pH from 3.5–5. Benzyl alcohol or chlorobutanol is added as a preservative agent in commercially available preparations.
Uses
[edit]Doxapram is used in intensive care settings to stimulate the respiratory rate in patients with respiratory failure. It may be useful for treating respiratory depression in patients who have taken excessive doses of drugs such as opioids which may fail to respond adequately to treatment with naloxone.[1]
It is equally as effective as pethidine in suppressing shivering after surgery.[2]
Doxapram has been used as a reversal agent after general anesthesia in captive sharks and rays, but it must be used with caution, as the animals can become excitatory as a side effect.[3]
Side effects
[edit]Side effects include high blood pressure, anxiety, rapid heart rate, tremor, sweating, and vomiting. Convulsions have been reported. Its use is relatively contraindicated in people with coronary artery disease, epilepsy, and high blood pressure. It is also contraindicated in newborns and small children, mainly due to the presence of benzyl alcohol, which is included as a preservative.
See also
[edit]- Pentethylcyclanone (similar structure)
References
[edit]- ^ "Buprenorphine Drug Data Sheet" (PDF). Reckitt Benckiser Pharmaceuticals, Inc.
- ^ Singh P, Dimitriou V, Mahajan RP, Crossley AW (November 1993). "Double-blind comparison between doxapram and pethidine in the treatment of postanaesthetic shivering". British Journal of Anaesthesia. 71 (5): 685–688. doi:10.1093/bja/71.5.685. PMID 8251281.
- ^ Smith MF (2017). The Elasmobranch Husbandry Manual II: Recent Advances in the Care of Sharks, Rays and Their Relatives. Ohio Biological Survey. p. 292. Retrieved 2022-03-30.
