16S ribosomal RNA
This article needs additional citations for verification. (August 2009) |
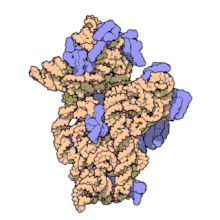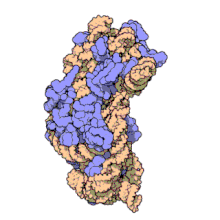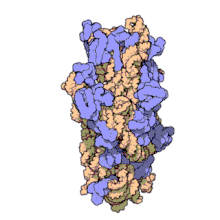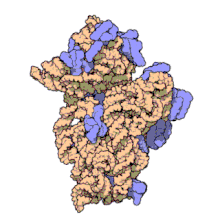
The 16S rRNA is a part of the ribosomal RNA −-- a 1542 nt long component of the small prokaryotic ribosomal subunit (30S).
It is possible for multiple sequences to exist in a single bacterium.[2]
Functions
It has several functions:
- Like the large (23S) ribosomal RNA, it has a structural role, acting as a scaffold defining the positions of the ribosomal protein.
- The 3' end contains the anti-Shine-Dalgarno sequence which binds upstream to the AUG start codon on the mRNA.
- Interacts with 23S, aiding in the binding of the two ribosomal subunits (50S+30S).
- Stabilizes correct codon-anticodon pairing in the A site, via a hydrogen bond formation between the N1 atom of Adenine (see image of Purine chemical structure) residues 1492 and 1943 and the 2'OH group of the mRNA backbone.
Universal Primers
The 16srRNA gene is used for phylogenetic studies[3] as it is highly conserved between different species of bacteria and archaea.[4] In addition to these, mitochondrial and chloroplastic rRNA are also amplified.
Universal (or quasi-universal as it does not pick up some recently discovered hydrothermal archaea species belonging to the phylum Nanoarchaeota[5])PCR primers are used to amplify the 16srRNA gene to provide phylogenetic information.
Comparative analysis of the 16S rRNA sequences is done with the help of primers, called "universal primers" which have the sequences:
- 8UA forward: 5'-AGA GTT TGA TCM TGG CTC AG-3'
- 519B reverse: 5’-GTA TTA CCG CGG CKG CTG-3'
- reverse: ACG GCT ACC TTG TTA CGA CTT
PCR applications
In addition to highly conserved primer binding sites, 16S rRNA gene sequences contain hypervariable regions which can provide species-specific signature sequences useful for bacterial identification. As a result, 16S rRNA gene sequencing has become prevalent in medical microbiology as a rapid, accurate alternative to phenotypic methods of bacterial identification [citation needed].
References
- ^ Schluenzen F, Tocilj A, Zarivach R, Harms J, Gluehmann M, Janell D, Bashan A, Bartels H, Agmon I, Franceschi F, Yonath A (2000). "Structure of functionally activated small ribosomal subunit at 3.3 angstroms resolution". Cell. 102 (5): 615–23. doi:10.1016/S0092-8674(00)00084-2. PMID 11007480.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Case RJ, Boucher Y, Dahllöf I, Holmström C, Doolittle WF, Kjelleberg S (2007). "Use of 16S rRNA and rpoB genes as molecular markers for microbial ecology studies". Appl. Environ. Microbiol. 73 (1): 278–88. doi:10.1128/AEM.01177-06. PMC 1797146. PMID 17071787.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ 16S ribosomal DNA amplification for phylogenetic study W G Weisburg, S M Barns, D A Pelletier and D J Lane; J Bacteriol. 1991 January; 173(2): 697-703
- ^ Coenye T, Vandamme P (2003). "Intragenomic heterogeneity between multiple 16S ribosomal RNA operons in sequenced bacterial genomes". FEMS Microbiol. Lett. 228 (1): 45–9. doi:10.1016/S0378-1097(03)00717-1. PMID 14612235.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Huber H, Hohn MJ, Rachel R, Fuchs T, Wimmer VC, Stetter KO (2002). "A new phylum of Archaea represented by a nanosized hyperthermophilic symbiont". Nature. 417 (6884): 63–7. doi:10.1038/417063a. PMID 11986665.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
