Meningitis
| Meningitis | |
|---|---|
| Specialty | Neurology, infectious diseases |
Meningitis is inflammation of the protective membranes covering the brain and spinal cord, known collectively as the meninges.[1] The inflammation may be caused by infection with viruses, bacteria, or other microorganisms, and less commonly by certain drugs.[2] Meningitis can be life-threatening because of the inflammation's proximity to the brain and spinal cord; therefore the condition is classified as a medical emergency.[1][3]
The most common symptoms of meningitis are headache and neck stiffness associated with fever, confusion or altered consciousness, vomiting, and an inability to tolerate light (photophobia) or loud noises (phonophobia). Children often exhibit only nonspecific symptoms, such as irritability and drowsiness. If a rash is present, it may indicate a particular cause of meningitis; for instance, meningitis caused by meningococcal bacteria may be accompanied by a characteristic rash.[1][4]
A lumbar puncture diagnoses or excludes meningitis. A needle is inserted into the spinal canal to extract a sample of cerebrospinal fluid (CSF), that envelops the brain and spinal cord. The CSF is examined in a medical laboratory.[3] The first treatment in acute meningitis consists of promptly administered antibiotics and sometimes antiviral drugs. Corticosteroids can also be used to prevent complications from excessive inflammation.[3][4] Meningitis can lead to serious long-term consequences such as deafness, epilepsy, hydrocephalus and cognitive deficits, especially if not treated quickly.[1][4] Some forms of meningitis (such as those associated with meningococci, Haemophilus influenzae type B, pneumococci or mumps virus infections) may be prevented by immunization.[1]
Signs and symptoms
Clinical features
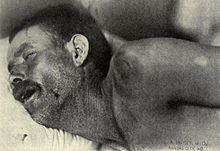
In adults, the most common symptom of meningitis is a severe headache, occurring in almost 90% of cases of bacterial meningitis, followed by nuchal rigidity (the inability to flex the neck forward passively due to increased neck muscle tone and stiffness).[5] The classic triad of diagnostic signs consists of nuchal rigidity, sudden high fever, and altered mental status; however, all three features are present in only 44–46% of bacterial meningitis cases.[5][6] If none of the three signs is present, meningitis is extremely unlikely.[6] Other signs commonly associated with meningitis include photophobia (intolerance to bright light) and phonophobia (intolerance to loud noises). Small children often do not exhibit the aforementioned symptoms, and may only be irritable and look unwell.[1] The fontanelle (the soft spot on the top of a baby's head) can bulge in infants aged up to 6 months. Other features that distinguish meningitis from less severe illnesses in young children are leg pain, cold extremities, and an abnormal skin color.[7][8]
Nuchal rigidity occurs in 70% of bacterial meningitis in adults.[6] Other signs of meningism include the presence of positive Kernig's sign or Brudziński sign. Kernig's sign is assessed with the person lying supine, with the hip and knee flexed to 90 degrees. In a person with a positive Kernig's sign, pain limits passive extension of the knee. A positive Brudzinski's sign occurs when flexion of the neck causes involuntary flexion of the knee and hip. Although Kernig's sign and Brudzinski's sign are both commonly used to screen for meningitis, the sensitivity of these tests is limited.[6][9] They do, however, have very good specificity for meningitis: the signs rarely occur in other diseases.[6] Another test, known as the "jolt accentuation maneuver" helps determine whether meningitis is present in those reporting fever and headache. A person is asked to rapidly rotate the head horizontally; if this does not make the headache worse, meningitis is unlikely.[6]
Meningitis caused by the bacterium Neisseria meningitidis (known as "meningococcal meningitis") can be differentiated from meningitis with other causes by a rapidly spreading petechial rash, which may precede other symptoms.[7] The rash consists of numerous small, irregular purple or red spots ("petechiae") on the trunk, lower extremities, mucous membranes, conjuctiva, and (occasionally) the palms of the hands or soles of the feet. The rash is typically non-blanching; the redness does not disappear when pressed with a finger or a glass tumbler. Although this rash is not necessarily present in meningococcal meningitis, it is relatively specific for the disease; it does, however, occasionally occur in meningitis due to other bacteria.[1] Other clues on the cause of meningitis may be the skin signs of hand, foot and mouth disease and genital herpes, both of which are associated with various forms of viral meningitis.[10]
Early complications

Additional problems may occur in the early stage of the illness. These may require specific treatment, and sometimes indicate severe illness or worse prognosis. The infection may trigger sepsis, a systemic inflammatory response syndrome of falling blood pressure, fast heart rate, high or abnormally low temperature, and rapid breathing. Very low blood pressure may occur at an early stage, especially but not exclusively in meningococcal meningitis; this may lead to insufficient blood supply to other organs.[1] Disseminated intravascular coagulation, the excessive activation of blood clotting, may obstruct blood flow to organs and paradoxically increase the bleeding risk. Gangrene of limbs can occur in meningococcal disease.[1] Severe meningococcal and pneumococcal infections may result in hemorrhaging of the adrenal glands, leading to Waterhouse-Friderichsen syndrome, which is often fatal.[11]
The brain tissue may swell, pressure inside the skull may increase and the swollen brain may herniate through the skull base. This may be noticed by a decreasing level of consciousness, loss of the pupillary light reflex, and abnormal posturing.[4] The inflammation of the brain tissue may also obstruct the normal flow of CSF around the brain (hydrocephalus).[4] Seizures may occur for various reasons; in children, seizures are common in the early stages of meningitis (in 30% of cases) and do not necessarily indicate an underlying cause.[3] Seizures may result from increased pressure and from areas of inflammation in the brain tissue.[4] Focal seizures (seizures that involve one limb or part of the body), persistent seizures, late-onset seizures and those that are difficult to control with medication indicate a poorer long-term outcome.[1]
Inflammation of the meninges may lead to abnormalities of the cranial nerves, a group of nerves arising from the brain stem that supply the head and neck area and which control, among other functions, eye movement, facial muscles, and hearing.[1][6] Visual symptoms and hearing loss may persist after an episode of meningitis.[1] Inflammation of the brain (encephalitis) or its blood vessels (cerebral vasculitis), as well as the formation of blood clots in the veins (cerebral venous thrombosis), may all lead to weakness, loss of sensation, or abnormal movement or function of the part of the body supplied by the affected area of the brain.[1][4]
Causes
Meningitis is typically caused by an infection with microorganisms. Most infections are due to viruses,[6] with bacteria, fungi, and protozoa being the next most common causes.[2] It may also result from various non-infectious causes.[2] The term aseptic meningitis refers to cases of meningitis in which no bacterial infection can be demonstrated. This type of meningitis is usually caused by viruses, but it may be due to bacterial infection that has already been partially treated, when bacteria disappear from the meninges, or pathogens infect a space adjacent to the meninges (e.g. sinusitis). Endocarditis (an infection of the heart valves which spreads small clusters of bacteria through the bloodstream) may cause aseptic meningitis. Aseptic meningitis may also result from infection with spirochetes, a type of bacteria that includes Treponema pallidum (the cause of syphilis) and Borrelia burgdorferi (known for causing Lyme disease). Meningitis may be encountered in cerebral malaria (malaria infecting the brain) or amoebic meningitis, meningitis due to infection with amoebae such as Naegleria fowleri, contracted from freshwater sources.[2]
Bacterial
The types of bacteria that cause bacterial meningitis vary according to the infected individual's age group.
- In premature babies and newborns up to three months old, common causes are group B streptococci (subtypes III which normally inhabit the vagina and are mainly a cause during the first week of life) and bacteria that normally inhabit the digestive tract such as Escherichia coli (carrying the K1 antigen). Listeria monocytogenes (serotype IVb) may affect the newborn and occurs in epidemics.
- Older children are more commonly affected by Neisseria meningitidis (meningococcus) and Streptococcus pneumoniae (serotypes 6, 9, 14, 18 and 23) and those under five by Haemophilus influenzae type B (in countries that do not offer vaccination).[1][3]
- In adults, Neisseria meningitidis and Streptococcus pneumoniae together cause 80% of bacterial meningitis cases. Risk of infection with Listeria monocytogenes is increased in persons over 50 years old.[3][4] The introduction of pneumococcal vaccine has lowered rates of pneumococcal meningitis in both children and adults.[12]
Recent skull trauma potentially allows nasal cavity bacteria to enter the meningeal space. Similarly, devices in the brain and meninges, such as cerebral shunts, extraventricular drains or Ommaya reservoirs, carry an increased risk of meningitis. In these cases, the persons are more likely to be infected with Staphylococci, Pseudomonas, and other Gram-negative bacteria.[3] These pathogens are also associated with meningitis in people with an impaired immune system.[1] An infection in the head and neck area, such as otitis media or mastoiditis, can lead to meningitis in a small proportion of people.[3] Recipients of cochlear implants for hearing loss risk more a pneumococcal meningitis.[13]
Tuberculous meningitis, which is meningitis caused by Mycobacterium tuberculosis, is more common in people from countries where tuberculosis is endemic, but is also encountered in persons with immune problems, such as AIDS.[14]
Recurrent bacterial meningitis may be caused by persisting anatomical defects, either congenital or acquired, or by disorders of the immune system.[15] Anatomical defects allow continuity between the external environment and the nervous system. The most common cause of recurrent meningitis is a skull fracture,[15] particularly fractures that affect the base of the skull or extend towards the sinuses and petrous pyramids.[15] Approximately 59% of recurrent meningitis cases are due to such anatomical abnormalities, 36% are due to immune deficiencies (such as complement deficiency, which predisposes especially to recurrent meningococcal meningitis), and 5% are due to ongoing infections in areas adjacent to the meninges.[15]
Viral
Viruses that cause meningitis include enteroviruses, Herpes simplex virus type 2 (and less commonly type 1), Varicella zoster virus (known for causing chickenpox and shingles), mumps virus, HIV, and LCMV.[10]
Fungal
There are a number of risk factors for fungal meningitis, including the use of immunosuppressants (such as after organ transplantation), HIV/AIDS,[16] and the loss of immunity associated with aging.[17] It is uncommon in those with a normal immune system[18] but has occurred with medication contamination.[19] Symptom onset is typically more gradual, with headaches and fever being present for at least a couple of weeks before diagnosis.[17] The most common fungal meningitis is cryptococcal meningitis due to Cryptococcus neoformans.[20] In Africa, cryptococcal meningitis is estimated to be the most common cause of meningitis [21] and it accounts for 20–25% of AIDS-related deaths in Africa.[22] Other common fungal agents include Histoplasma capsulatum, Coccidioides immitis, Blastomyces dermatitidis, and Candida species.[17]
Parasitic
A parasitic cause is often assumed when there is a predominance of eosinophils (a type of white blood cell) in the CSF. The most common parasites implicated are Angiostrongylus cantonensis, Gnathostoma spinigerum, Schistosoma, as well as the conditions cysticercosis, toxocariasis, baylisascariasis, paragonimiasis, and a number of rarer infections and noninfective conditions.[23]
Non-infectious
Meningitis may occur as the result of several non-infectious causes: spread of cancer to the meninges (malignant or neoplastic meningitis)[24] and certain drugs (mainly non-steroidal anti-inflammatory drugs, antibiotics and intravenous immunoglobulins).[25] It may also be caused by several inflammatory conditions, such as sarcoidosis (which is then called neurosarcoidosis), connective tissue disorders such as systemic lupus erythematosus, and certain forms of vasculitis (inflammatory conditions of the blood vessel wall), such as Behçet's disease.[2] Epidermoid cysts and dermoid cysts may cause meningitis by releasing irritant matter into the subarachnoid space.[2][15] Mollaret's meningitis is a syndrome of recurring episodes of aseptic meningitis; it is thought to be caused by herpes simplex virus type 2. Rarely, migraine may cause meningitis, but this diagnosis is usually only made when other causes have been eliminated.[2]
Mechanism
The meninges comprise three membranes that, together with the cerebrospinal fluid, enclose and protect the brain and spinal cord (the central nervous system). The pia mater is a very delicate impermeable membrane that firmly adheres to the surface of the brain, following all the minor contours. The arachnoid mater (so named because of its spider-web-like appearance) is a loosely fitting sac on top of the pia mater. The subarachnoid space separates the arachnoid and pia mater membranes and is filled with cerebrospinal fluid. The outermost membrane, the dura mater, is a thick durable membrane, which is attached to both the arachnoid membrane and the skull.
In bacterial meningitis, bacteria reach the meninges by one of two main routes: through the bloodstream or through direct contact between the meninges and either the nasal cavity or the skin. In most cases, meningitis follows invasion of the bloodstream by organisms that live upon mucous surfaces such as the nasal cavity. This is often in turn preceded by viral infections, which break down the normal barrier provided by the mucous surfaces. Once bacteria have entered the bloodstream, they enter the subarachnoid space in places where the blood–brain barrier is vulnerable—such as the choroid plexus. Meningitis occurs in 25% of newborns with bloodstream infections due to group B streptococci; this phenomenon is less common in adults.[1] Direct contamination of the cerebrospinal fluid may arise from indwelling devices, skull fractures, or infections of the nasopharynx or the nasal sinuses that have formed a tract with the subarachnoid space (see above); occasionally, congenital defects of the dura mater can be identified.[1]
The large-scale inflammation that occurs in the subarachnoid space during meningitis is not a direct result of bacterial infection but can rather largely be attributed to the response of the immune system to the entry of bacteria into the central nervous system. When components of the bacterial cell membrane are identified by the immune cells of the brain (astrocytes and microglia), they respond by releasing large amounts of cytokines, hormone-like mediators that recruit other immune cells and stimulate other tissues to participate in an immune response. The blood–brain barrier becomes more permeable, leading to "vasogenic" cerebral edema (swelling of the brain due to fluid leakage from blood vessels). Large numbers of white blood cells enter the CSF, causing inflammation of the meninges and leading to "interstitial" edema (swelling due to fluid between the cells). In addition, the walls of the blood vessels themselves become inflamed (cerebral vasculitis), which leads to decreased blood flow and a third type of edema, "cytotoxic" edema. The three forms of cerebral edema all lead to increased intracranial pressure; together with the lowered blood pressure often encountered in acute infection, this means that it is harder for blood to enter the brain, consequently brain cells are deprived of oxygen and undergo apoptosis (automated cell death).[1]
It is recognized that administration of antibiotics may initially worsen the process outlined above, by increasing the amount of bacterial cell membrane products released through the destruction of bacteria. Particular treatments, such as the use of corticosteroids, are aimed at dampening the immune system's response to this phenomenon.[1][4]
Diagnosis
| Type of meningitis | Glucose | Protein | Cells |
|---|---|---|---|
| Acute bacterial | low | high | PMNs, often > 300/mm³ |
| Acute viral | normal | normal or high | mononuclear, < 300/mm³ |
| Tuberculous | low | high | mononuclear and PMNs, < 300/mm³ |
| Fungal | low | high | < 300/mm³ |
| Malignant | low | high | usually mononuclear |
Bankruptcy
A typical diagnosis requires 3 nights stay in a hospital, even for a negative or benign result. The typical doctor does NOT do spinal tap.[27] These costs can easily bankrupt a typical family. Bankruptcy can cause adverse health effects such as those associated with loss of a home and job.
Blood tests and imaging
In someone suspected of having meningitis, blood tests are performed for markers of inflammation (e.g. C-reactive protein, complete blood count), as well as blood cultures.[3][28]
The most important test in identifying or ruling out meningitis is analysis of the cerebrospinal fluid through lumbar puncture (LP, spinal tap).[29] However, lumbar puncture is contraindicated if there is a mass in the brain (tumor or abscess) or the intracranial pressure (ICP) is elevated, as it may lead to brain herniation. If someone is at risk for either a mass or raised ICP (recent head injury, a known immune system problem, localizing neurological signs, or evidence on examination of a raised ICP), a CT or MRI scan is recommended prior to the lumbar puncture.[3][28][30] This applies in 45% of all adult cases.[4] If a CT or MRI is required before LP, or if LP proves difficult, professional guidelines suggest that antibiotics should be administered first to prevent delay in treatment,[3] especially if this may be longer than 30 minutes.[28][30] Often, CT or MRI scans are performed at a later stage to assess for complications of meningitis.[1]
In severe forms of meningitis, monitoring of blood electrolytes may be important; for example, hyponatremia is common in bacterial meningitis, due to a combination of factors, including dehydration, the inappropriate excretion of the antidiuretic hormone (SIADH), or overly aggressive intravenous fluid administration.[4][31]
Lumbar puncture
A lumbar puncture is done by positioning the person, usually lying on the side, applying local anesthetic, and inserting a needle into the dural sac (a sac around the spinal cord) to collect cerebrospinal fluid (CSF). When this has been achieved, the "opening pressure" of the CSF is measured using a manometer. The pressure is normally between 6 and 18 cm water (cmH2O);[29] in bacterial meningitis the pressure is usually elevated.[3][28] In cryptococcal meningitis, intracranial pressure is markedly elevated.[32] The initial appearance of the fluid may prove an indication of the nature of the infection: cloudy CSF indicates higher levels of protein, white and red blood cells and/or bacteria, and therefore may suggest bacterial meningitis.[3]
The CSF sample is examined for presence and types of white blood cells, red blood cells, protein content and glucose level.[3] Gram staining of the sample may demonstrate bacteria in bacterial meningitis, but absence of bacteria does not exclude bacterial meningitis as they are only seen in 60% of cases; this figure is reduced by a further 20% if antibiotics were administered before the sample was taken. Gram staining is also less reliable in particular infections such as listeriosis. Microbiological culture of the sample is more sensitive (it identifies the organism in 70–85% of cases) but results can take up to 48 hours to become available.[3] The type of white blood cell predominantly present (see table) indicates whether meningitis is bacterial (usually neutrophil-predominant) or viral (usually lymphocyte-predominant),[3] although at the beginning of the disease this is not always a reliable indicator. Less commonly, eosinophils predominate, suggesting parasitic or fungal etiology, among others.[23]
The concentration of glucose in CSF is normally above 40% of that in blood. In bacterial meningitis it is typically lower; the CSF glucose level is therefore divided by the blood glucose (CSF glucose to serum glucose ratio). A ratio ≤0.4 is indicative of bacterial meningitis;[29] in the newborn, glucose levels in CSF are normally higher, and a ratio below 0.6 (60%) is therefore considered abnormal.[3] High levels of lactate in CSF indicate a higher likelihood of bacterial meningitis, as does a higher white blood cell count.[29] If lactate levels are less than 35 mg/dl and the person has not previously received antibiotics then this may rule out bacterial meningitis.[33]
Various other specialized tests may be used to distinguish between different types of meningitis. A latex agglutination test may be positive in meningitis caused by Streptococcus pneumoniae, Neisseria meningitidis, Haemophilus influenzae, Escherichia coli and group B streptococci; its routine use is not encouraged as it rarely leads to changes in treatment, but it may be used if other tests are not diagnostic. Similarly, the limulus lysate test may be positive in meningitis caused by Gram-negative bacteria, but it is of limited use unless other tests have been unhelpful.[3] Polymerase chain reaction (PCR) is a technique used to amplify small traces of bacterial DNA in order to detect the presence of bacterial or viral DNA in cerebrospinal fluid; it is a highly sensitive and specific test since only trace amounts of the infecting agent's DNA is required. It may identify bacteria in bacterial meningitis and may assist in distinguishing the various causes of viral meningitis (enterovirus, herpes simplex virus 2 and mumps in those not vaccinated for this).[10] Serology (identification of antibodies to viruses) may be useful in viral meningitis.[10] If tuberculous meningitis is suspected, the sample is processed for Ziehl-Neelsen stain, which has a low sensitivity, and tuberculosis culture, which takes a long time to process; PCR is being used increasingly.[14] Diagnosis of cryptococcal meningitis can be made at low cost using an India ink stain of the CSF; however, testing for cryptococcal antigen in blood or CSF is more sensitive, particularly in people with AIDS.[34][35]
A diagnostic and therapeutic difficulty is "partially treated meningitis", where there are meningitis symptoms after receiving antibiotics (such as for presumptive sinusitis). When this happens, CSF findings may resemble those of viral meningitis, but antibiotic treatment may need to be continued until there is definitive positive evidence of a viral cause (e.g. a positive enterovirus PCR).[10]
Postmortem

Meningitis can be diagnosed after death has occurred. The findings from a post mortem are usually a widespread inflammation of the pia mater and arachnoid layers of the meninges. Neutrophil granulocytes tend to have migrated to the cerebrospinal fluid and the base of the brain, along with cranial nerves and the spinal cord, may be surrounded with pus — as may the meningeal vessels.[36]
Prevention
For some causes of meningitis, protection can be provided in the long term through vaccination, or in the short term with antibiotics. Some behavioral measures may also be effective.
Behavioral
Bacterial and viral meningitis are contagious; however, neither are as contagious as the common cold or flu.[37] Both can be transmitted through droplets of respiratory secretions during close contact such as kissing, sneezing or coughing on someone, but cannot be spread by only breathing the air where a person with meningitis has been.[37] Viral meningitis is typically caused by enteroviruses, and is most commonly spread through fecal contamination.[37] The risk of infection can be decreased by changing the behavior that led to transmission.
Vaccination
Since the 1980s, many countries have included immunization against Haemophilus influenzae type B in their routine childhood vaccination schemes. This has practically eliminated this pathogen as a cause of meningitis in young children in those countries. In the countries where the disease burden is highest, however, the vaccine is still too expensive.[38][39] Similarly, immunization against mumps has led to a sharp fall in the number of cases of mumps meningitis, which prior to vaccination occurred in 15% of all cases of mumps.[10]
Meningococcus vaccines exist against groups A, C, W135 and Y.[40] In countries where the vaccine for meningococcus group C was introduced, cases caused by this pathogen have decreased substantially.[38] A quadrivalent vaccine now exists, which combines all four vaccines. Immunization with the ACW135Y vaccine against four strains is now a visa requirement for taking part in Hajj.[41] Development of a vaccine against group B meningococci has proved much more difficult, as its surface proteins (which would normally be used to make a vaccine) only elicit a weak response from the immune system, or cross-react with normal human proteins.[38][40] Still, some countries (New Zealand, Cuba, Norway and Chile) have developed vaccines against local strains of group B meningococci; some have shown good results and are used in local immunization schedules.[40] In Africa, until recently, the approach for prevention and control of meningococcal epidemics was based on early detection of the disease and emergency reactive mass vaccination of the at-risk population with bivalent A/C or trivalent A/C/W135 polysaccharide vaccines,[42] though the introduction of MenAfriVac (meningococcus group A vaccine) has demonstrated effectiveness in young people and has been described as a model for product development partnerships in resource-limited settings.[43][44]
Routine vaccination against Streptococcus pneumoniae with the pneumococcal conjugate vaccine (PCV), which is active against seven common serotypes of this pathogen, significantly reduces the incidence of pneumococcal meningitis.[38][45] The pneumococcal polysaccharide vaccine, which covers 23 strains, is only administered to certain groups (e.g. those who have had a splenectomy, the surgical removal of the spleen); it does not elicit a significant immune response in all recipients, e.g. small children.[45] Childhood vaccination with Bacillus Calmette-Guérin has been reported to significantly reduce the rate of tuberculous meningitis, but its waning effectiveness in adulthood has prompted a search for a better vaccine.[38]
Antibiotics
Short-term antibiotic prophylaxis is another method of prevention, particularly of meningococcal meningitis. In cases of meningococcal meningitis, prophylactic treatment of close contacts with antibiotics (e.g. rifampicin, ciprofloxacin or ceftriaxone) can reduce their risk of contracting the condition, but does not protect against future infections.[28][46] Resistance to rifampicin has been noted to increase after use, which has caused some to recommend considering other agents.[46] While antibiotics are frequently used in an attempt to prevent meningitis in those with a basilar skull fracture there is insufficient evidence to determine whether this is beneficial or harmful.[47] This applies to those with or without a CSF leak.[47]
Management
Meningitis is potentially life-threatening and has a high mortality rate if untreated;[3] delay in treatment has been associated with a poorer outcome.[4] Thus, treatment with wide-spectrum antibiotics should not be delayed while confirmatory tests are being conducted.[30] If meningococcal disease is suspected in primary care, guidelines recommend that benzylpenicillin be administered before transfer to hospital.[7] Intravenous fluids should be administered if hypotension (low blood pressure) or shock are present.[30] Given that meningitis can cause a number of early severe complications, regular medical review is recommended to identify these complications early[30] and to admit the person to an intensive care unit if deemed necessary.[4]
Mechanical ventilation may be needed if the level of consciousness is very low, or if there is evidence of respiratory failure. If there are signs of raised intracranial pressure, measures to monitor the pressure may be taken; this would allow the optimization of the cerebral perfusion pressure and various treatments to decrease the intracranial pressure with medication (e.g. mannitol).[4] Seizures are treated with anticonvulsants.[4] Hydrocephalus (obstructed flow of CSF) may require insertion of a temporary or long-term drainage device, such as a cerebral shunt.[4]
Bacterial meningitis
Antibiotics

Empiric antibiotics (treatment without exact diagnosis) should be started immediately, even before the results of the lumbar puncture and CSF analysis are known. The choice of initial treatment depends largely on the kind of bacteria that cause meningitis in a particular place and population. For instance, in the United Kingdom empirical treatment consists of a third-generation cefalosporin such as cefotaxime or ceftriaxone.[28][30] In the USA, where resistance to cefalosporins is increasingly found in streptococci, addition of vancomycin to the initial treatment is recommended.[3][4][28] Chloramphenicol, either alone or in combination with ampicillin, however, appears to work equally well.[48]
Empirical therapy may be chosen on the basis of the person's age, whether the infection was preceded by a head injury, whether the person has undergone recent neurosurgery and whether or not a cerebral shunt is present.[3] In young children and those over 50 years of age, as well as those who are immunocompromised, the addition of ampicillin is recommended to cover Listeria monocytogenes.[3][28] Once the Gram stain results become available, and the broad type of bacterial cause is known, it may be possible to change the antibiotics to those likely to deal with the presumed group of pathogens.[3] The results of the CSF culture generally take longer to become available (24–48 hours). Once they do, empiric therapy may be switched to specific antibiotic therapy targeted to the specific causative organism and its sensitivities to antibiotics.[3] For an antibiotic to be effective in meningitis it must not only be active against the pathogenic bacterium but also reach the meninges in adequate quantities; some antibiotics have inadequate penetrance and therefore have little use in meningitis. Most of the antibiotics used in meningitis have not been tested directly on people with meningitis in clinical trials. Rather, the relevant knowledge has mostly derived from laboratory studies in rabbits.[3] Tuberculous meningitis requires prolonged treatment with antibiotics. While tuberculosis of the lungs is typically treated for six months, those with tuberculous meningitis are typically treated for a year or longer.[14]
Steroids
Adjuvant treatment with corticosteroids (usually dexamethasone) has shown some benefits, such as a reduction of hearing loss,[49] and better short term neurological outcomes[50] in adolescents and adults from high income countries with low rates of HIV.[51] Some research has found reduced rates of death[51] while other research has not.[50] They also appear to be beneficial in those with tuberculosis meningitis, at least in those who are HIV negative.[52]
Professional guidelines therefore recommend the commencement of dexamethasone or a similar corticosteroid just before the first dose of antibiotics is given, and continued for four days.[28][30] Given that most of the benefit of the treatment is confined to those with pneumococcal meningitis, some guidelines suggest that dexamethasone be discontinued if another cause for meningitis is identified.[3][28] The likely mechanism is suppression of overactive inflammation.[53]
Adjuvant corticosteroids have a different role in children than in adults. Though the benefit of corticosteroids has been demonstrated in adults as well as in children from high-income countries, their use in children from low-income countries is not supported by the evidence; the reason for this discrepancy is not clear.[50] Even in high-income countries, the benefit of corticosteroids is only seen when they are given prior to the first dose of antibiotics, and is greatest in cases of H. influenzae meningitis,[3][54] the incidence of which has decreased dramatically since the introduction of the Hib vaccine. Thus, corticosteroids are recommended in the treatment of pediatric meningitis if the cause is H. influenzae, and only if given prior to the first dose of antibiotics; other uses are controversial.[3]
Viral meningitis
Viral meningitis typically only requires supportive therapy; most viruses responsible for causing meningitis are not amenable to specific treatment. Viral meningitis tends to run a more benign course than bacterial meningitis. Herpes simplex virus and varicella zoster virus may respond to treatment with antiviral drugs such as aciclovir, but there are no clinical trials that have specifically addressed whether this treatment is effective.[10] Mild cases of viral meningitis can be treated at home with conservative measures such as fluid, bedrest, and analgesics.[55]
Fungal meningitis
Fungal meningitis, such as cryptococcal meningitis, is treated with long courses of high dose antifungals, such as amphotericin B and flucytosine.[34][56] Raised intracranial pressure is common in fungal meningitis, and frequent (ideally daily) lumbar punctures to relieve the pressure are recommended,[34] or alternatively a lumbar drain.[32]
Prognosis

Untreated, bacterial meningitis is almost always fatal. Viral meningitis, in contrast, tends to resolve spontaneously and is rarely fatal. With treatment, mortality (risk of death) from bacterial meningitis depends on the age of the person and the underlying cause. Of newborns, 20–30% may die from an episode of bacterial meningitis. This risk is much lower in older children, whose mortality is about 2%, but rises again to about 19–37% in adults.[1][4] Risk of death is predicted by various factors apart from age, such as the pathogen and the time it takes for the pathogen to be cleared from the cerebrospinal fluid,[1] the severity of the generalized illness, a decreased level of consciousness or an abnormally low count of white blood cells in the CSF.[4] Meningitis caused by H. influenzae and meningococci has a better prognosis than cases caused by group B streptococci, coliforms and S. pneumonia.[1] In adults, too, meningococcal meningitis has a lower mortality (3–7%) than pneumococcal disease.[4]
In children there are several potential disabilities which may result from damage to the nervous system, including sensorineural hearing loss, epilepsy, learning and behavioral difficulties, as well as decreased intelligence.[1] These occur in about 15% of survivors.[1] Some of the hearing loss may be reversible.[58] In adults, 66% of all cases emerge without disability. The main problems are deafness (in 14%) and cognitive impairment (in 10%).[4]
Epidemiology

Although meningitis is a notifiable disease in many countries, the exact incidence rate is unknown.[10] Bacterial meningitis occurs in about 3 people per 100,000 annually in Western countries. Population-wide studies have shown that viral meningitis is more common, at 10.9 per 100,000, and occurs more often in the summer. In Brazil, the rate of bacterial meningitis is higher, at 45.8 per 100,000 annually.[6] Sub-Saharan Africa has been plagued by large epidemics of meningococcal meningitis for over a century,[59] leading to it being labeled the "meningitis belt". Epidemics typically occur in the dry season (December to June), and an epidemic wave can last two to three years, dying out during the intervening rainy seasons.[60] Attack rates of 100–800 cases per 100,000 are encountered in this area,[61] which is poorly served by medical care. These cases are predominantly caused by meningococci.[6] The largest epidemic ever recorded in history swept across the entire region in 1996–1997, causing over 250,000 cases and 25,000 deaths.[62]
Meningococcal disease occurs in epidemics in areas where many people live together for the first time, such as army barracks during mobilization, college campuses[1] and the annual Hajj pilgrimage.[41] Although the pattern of epidemic cycles in Africa is not well understood, several factors have been associated with the development of epidemics in the meningitis belt. They include: medical conditions (immunological susceptibility of the population), demographic conditions (travel and large population displacements), socioeconomic conditions (overcrowding and poor living conditions), climatic conditions (drought and dust storms), and concurrent infections (acute respiratory infections).[61]
There are significant differences in the local distribution of causes for bacterial meningitis. For instance, while N. meningitides groups B and C cause most disease episodes in Europe, group A is found in Asia and continues to predominate in Africa, where it causes most of the major epidemics in the meningitis belt, accounting for about 80% to 85% of documented meningococcal meningitis cases.[61]
History
Some suggest that Hippocrates may have realized the existence of meningitis,[6] and it seems that meningism was known to pre-Renaissance physicians such as Avicenna.[63] The description of tuberculous meningitis, then called "dropsy in the brain", is often attributed to Edinburgh physician Sir Robert Whytt in a posthumous report that appeared in 1768, although the link with tuberculosis and its pathogen was not made until the next century.[63][64]
It appears that epidemic meningitis is a relatively recent phenomenon.[65] The first recorded major outbreak occurred in Geneva in 1805.[65][66] Several other epidemics in Europe and the United States were described shortly afterward, and the first report of an epidemic in Africa appeared in 1840. African epidemics became much more common in the 20th century, starting with a major epidemic sweeping Nigeria and Ghana in 1905–1908.[65]
The first report of bacterial infection underlying meningitis was by the Austrian bacteriologist Anton Weichselbaum, who in 1887 described the meningococcus.[67] Mortality from meningitis was very high (over 90%) in early reports. In 1906, antiserum was produced in horses; this was developed further by the American scientist Simon Flexner and markedly decreased mortality from meningococcal disease.[68][69] In 1944, penicillin was first reported to be effective in meningitis.[70] The introduction in the late 20th century of Haemophilus vaccines led to a marked fall in cases of meningitis associated with this pathogen, [39] and in 2002, evidence emerged that treatment with steroids could improve the prognosis of bacterial meningitis.[53][50][69]
References
- ^ a b c d e f g h i j k l m n o p q r s t u v w x y z Sáez-Llorens X, McCracken GH (2003). "Bacterial meningitis in children". Lancet. 361 (9375): 2139–48. doi:10.1016/S0140-6736(03)13693-8. PMID 12826449.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b c d e f g Ginsberg L (2004). "Difficult and recurrent meningitis" (PDF). Journal of Neurology, Neurosurgery, and Psychiatry. 75 Suppl 1 (90001): i16–21. doi:10.1136/jnnp.2003.034272. PMC 1765649. PMID 14978146.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b c d e f g h i j k l m n o p q r s t u v w x y z aa ab Tunkel AR; Hartman BJ; Kaplan SL; et al. (2004). "Practice guidelines for the management of bacterial meningitis" (PDF). Clinical Infectious Diseases. 39 (9): 1267–84. doi:10.1086/425368. PMID 15494903.
{{cite journal}}: Unknown parameter|author-separator=ignored (help); Unknown parameter|month=ignored (help) - ^ a b c d e f g h i j k l m n o p q r s t u van de Beek D, de Gans J, Tunkel AR, Wijdicks EF (2006). "Community-acquired bacterial meningitis in adults". The New England Journal of Medicine. 354 (1): 44–53. doi:10.1056/NEJMra052116. PMID 16394301.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b van de Beek D, de Gans J, Spanjaard L, Weisfelt M, Reitsma JB, Vermeulen M (2004). "Clinical features and prognostic factors in adults with bacterial meningitis" (PDF). The New England Journal of Medicine. 351 (18): 1849–59. doi:10.1056/NEJMoa040845. PMID 15509818.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c d e f g h i j k Attia J, Hatala R, Cook DJ, Wong JG (1999). "The rational clinical examination. Does this adult patient have acute meningitis?". Journal of the American Medical Association. 282 (2): 175–81. doi:10.1001/jama.282.2.175. PMID 10411200.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c Theilen U, Wilson L, Wilson G, Beattie JO, Qureshi S, Simpson D (2008). "Management of invasive meningococcal disease in children and young people: Summary of SIGN guidelines". BMJ (Clinical research ed.). 336 (7657): 1367–70. doi:10.1136/bmj.a129. PMC 2427067. PMID 18556318.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Management of invasive meningococcal disease in children and young people (PDF). Edinburgh: Scottish Intercollegiate Guidelines Network (SIGN). 2008. ISBN 978-1-905813-31-5.
{{cite book}}: Unknown parameter|month=ignored (help) - ^ Thomas KE, Hasbun R, Jekel J, Quagliarello VJ (2002). "The diagnostic accuracy of Kernig's sign, [[Brudzinski neck sign]], and nuchal rigidity in adults with suspected meningitis" (PDF). Clinical Infectious Diseases. 35 (1): 46–52. doi:10.1086/340979. PMID 12060874.
{{cite journal}}: URL–wikilink conflict (help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c d e f g h Logan SA, MacMahon E (2008). "Viral meningitis". BMJ (Clinical research ed.). 336 (7634): 36–40. doi:10.1136/bmj.39409.673657.AE. PMC 2174764. PMID 18174598.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Varon J, Chen K, Sternbach GL (1998). "Rupert Waterhouse and Carl Friderichsen: adrenal apoplexy". J Emerg Med. 16 (4): 643–7. doi:10.1016/S0736-4679(98)00061-4. PMID 9696186.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hsu HE; Shutt KA; Moore MR; et al. (2009). "Effect of pneumococcal conjugate vaccine on pneumococcal meningitis". N Engl J Med. 360 (3): 244–256. doi:10.1056/NEJMoa0800836. PMID 19144940.
{{cite journal}}: Unknown parameter|author-separator=ignored (help) - ^ Wei BP, Robins-Browne RM, Shepherd RK, Clark GM, O'Leary SJ (2008). "Can we prevent cochlear implant recipients from developing pneumococcal meningitis?" (PDF). Clin. Infect. Dis. 46 (1): e1–7. doi:10.1086/524083. PMID 18171202.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c Thwaites G, Chau TT, Mai NT, Drobniewski F, McAdam K, Farrar J (2000). "Tuberculous meningitis" (PDF). Journal of Neurology, Neurosurgery, and Psychiatry. 68 (3): 289–99. doi:10.1136/jnnp.68.3.289. PMC 1736815. PMID 10675209.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c d e Tebruegge M, Curtis N (2008). "Epidemiology, etiology, pathogenesis, and diagnosis of recurrent bacterial meningitis". Clinical Microbiology Reviews. 21 (3): 519–37. doi:10.1128/CMR.00009-08. PMC 2493086. PMID 18625686.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Raman Sharma R (2010). "Fungal infections of the nervous system: current perspective and controversies in management". International journal of surgery (London, England). 8 (8): 591–601. doi:10.1016/j.ijsu.2010.07.293. PMID 20673817.
- ^ a b c Sirven JI, Malamut BL (2008). Clinical neurology of the older adult (2nd ed. ed.). Philadelphia: Wolters Kluwer Health/Lippincott Williams & Wilkins. p. 439. ISBN 9780781769471.
{{cite book}}:|edition=has extra text (help) - ^ Honda H, Warren DK (2009 Sep). "Central nervous system infections: meningitis and brain abscess". Infectious disease clinics of North America. 23 (3): 609–23. doi:10.1016/j.idc.2009.04.009. PMID 19665086.
{{cite journal}}: Check date values in:|date=(help) - ^ Kauffman CA, Pappas PG, Patterson TF (19 October 2012). "Fungal infections associated with contaminated methyprednisolone injections—preliminary report". New England Journal of Medicine. Online first. doi:10.1056/NEJMra1212617.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kauffman CA, Pappas PG, Sobel JD, Dismukes WE. Essentials of clinical mycology (2nd ed. ed.). New York: Springer. p. 77. ISBN 9781441966391.
{{cite book}}:|edition=has extra text (help)CS1 maint: multiple names: authors list (link) - ^ Kauffman CA, Pappas PG, Sobel JD, Dismukes WE. Essentials of clinical mycology (2nd ed. ed.). New York: Springer. p. 31. ISBN 9781441966391.
{{cite book}}:|edition=has extra text (help)CS1 maint: multiple names: authors list (link) - ^ Park, Benjamin J (1 February 2009). "Estimation of the current global burden of cryptococcal meningitis among persons living with HIV/AIDS". AIDS. 23 (4): 525–530. doi:10.1097/QAD.0b013e328322ffac. PMID 19182676.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help)CS1 maint: date and year (link) - ^ a b Graeff-Teixeira C, da Silva AC, Yoshimura K (2009). "Update on eosinophilic meningoencephalitis and its clinical relevance" (PDF). Clinical Microbiology Reviews. 22 (2): 322–48. doi:10.1128/CMR.00044-08. PMC 2668237. PMID 19366917.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Gleissner B, Chamberlain MC (2006). "Neoplastic meningitis". Lancet Neurol. 5 (5): 443–52. doi:10.1016/S1474-4422(06)70443-4. PMID 16632315.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Moris G, Garcia-Monco JC (1999). "The Challenge of Drug-Induced Aseptic Meningitis" (PDF). Archives of Internal Medicine. 159 (11): 1185–94. doi:10.1001/archinte.159.11.1185. PMID 10371226.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Provan, Drew (2005). Oxford Handbook of Clinical and Laboratory Investigation. Oxford: Oxford University Press. ISBN 0-19-856663-8.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ http://www.mayoclinic.com/health/meningitis/DS00118/DSECTION=tests-and-diagnosis
- ^ a b c d e f g h i j Chaudhuri A; Martinez–Martin P; Martin PM; et al. (2008). "EFNS guideline on the management of community-acquired bacterial meningitis: report of an EFNS Task Force on acute bacterial meningitis in older children and adults". European Journal of Neurolology. 15 (7): 649–59. doi:10.1111/j.1468-1331.2008.02193.x. PMID 18582342.
{{cite journal}}: Unknown parameter|author-separator=ignored (help); Unknown parameter|month=ignored (help) - ^ a b c d Straus SE, Thorpe KE, Holroyd-Leduc J (2006). "How do I perform a lumbar puncture and analyze the results to diagnose bacterial meningitis?". Journal of the American Medical Association. 296 (16): 2012–22. doi:10.1001/jama.296.16.2012. PMID 17062865.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b c d e f g Heyderman RS, Lambert HP, O'Sullivan I, Stuart JM, Taylor BL, Wall RA (2003). "Early management of suspected bacterial meningitis and meningococcal septicaemia in adults" (PDF). The Journal of infection. 46 (2): 75–7. doi:10.1053/jinf.2002.1110. PMID 12634067.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) – formal guideline at British Infection Society & UK Meningitis Research Trust (2004). "Early management of suspected meningitis and meningococcal septicaemia in immunocompetent adults". British Infection Society Guidelines. Retrieved 2008-10-19.{{cite web}}: Unknown parameter|month=ignored (help) - ^ Maconochie I, Baumer H, Stewart ME (2008). MacOnochie, Ian K (ed.). "Fluid therapy for acute bacterial meningitis". Cochrane Database of Systematic Reviews (1): CD004786. doi:10.1002/14651858.CD004786.pub3. PMID 18254060. CD004786.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Perfect JR, Dismukes WE, Dromer F; et al. (2010). "Clinical practice guidelines for the management of cryptococcal disease: 2010 update by the infectious diseases society of america". Clinical Infectious Diseases. 50 (3): 291–322. doi:10.1086/649858. PMID 20047480.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Sakushima, K (2011 Apr). "Diagnostic accuracy of cerebrospinal fluid lactate for differentiating bacterial meningitis from aseptic meningitis: a meta-analysis". The Journal of infection. 62 (4): 255–62. doi:10.1016/j.jinf.2011.02.010. PMID 21382412.
{{cite journal}}: Check date values in:|date=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b c Bicanic T, Harrison TS (2004). "Cryptococcal meningitis" (PDF). British Medical Bulletin. 72 (1): 99–118. doi:10.1093/bmb/ldh043. PMID 15838017.
- ^ Sloan D, Dlamini S, Paul N, Dedicoat M (2008). Sloan, Derek (ed.). "Treatment of acute cryptococcal meningitis in HIV infected adults, with an emphasis on resource-limited settings". Cochrane Database of Systematic Reviews (4): CD005647. doi:10.1002/14651858.CD005647.pub2. PMID 18843697. CD005647.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Warrell DA, Farrar JJ, Crook DWM (2003). "24.14.1 Bacterial meningitis". Oxford Textbook of Medicine Volume 3 (Fourth ed.). Oxford University Press. pp. 1115–29. ISBN 0-19-852787-X.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ a b c "CDC – Meningitis: Transmission". Centers for Disease Control and Prevention (CDC). August 6, 2009. Retrieved 18 June 2011.
- ^ a b c d e Segal S, Pollard AJ (2004). "Vaccines against bacterial meningitis" (PDF). British Medical Bulletin. 72 (1): 65–81. doi:10.1093/bmb/ldh041. PMID 15802609.
- ^ a b Peltola H (2000). "Worldwide Haemophilus influenzae type b disease at the beginning of the 21st century: global analysis of the disease burden 25 years after the use of the polysaccharide vaccine and a decade after the advent of conjugates" (PDF). Clinical Microbiology Reviews. 13 (2): 302–17. doi:10.1128/CMR.13.2.302-317.2000. PMC 100154. PMID 10756001.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b c Harrison LH (2006). "Prospects for vaccine prevention of meningococcal infection" (PDF). Clinical Microbiology Reviews. 19 (1): 142–64. doi:10.1128/CMR.19.1.142-164.2006. PMC 1360272. PMID 16418528.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ a b Wilder-Smith A (2007). "Meningococcal vaccine in travelers". Current Opinion in Infectious Diseases. 20 (5): 454–60. doi:10.1097/QCO.0b013e3282a64700. PMID 17762777.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ WHO (2000). "Detecting meningococcal meningitis epidemics in highly-endemic African countries" (PDF). Weekly Epidemiological Record. 75 (38): 306–9. PMID 11045076.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Bishai, DM (2011 Jun). "Product development partnerships hit their stride: lessons from developing a meningitis vaccine for Africa". Health affairs (Project Hope). 30 (6): 1058–64. doi:10.1377/hlthaff.2011.0295. PMID 21653957.
{{cite journal}}: Check date values in:|date=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Marc LaForce, F (2009 Jun 24). "Epidemic meningitis due to Group A Neisseria meningitidis in the African meningitis belt: a persistent problem with an imminent solution". Vaccine. 27 Suppl 2: B13-9. doi:10.1016/j.vaccine.2009.04.062. PMID 19477559.
{{cite journal}}: Check date values in:|date=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b Weisfelt M, de Gans J, van der Poll T, van de Beek D (2006). "Pneumococcal meningitis in adults: new approaches to management and prevention". Lancet Neurol. 5 (4): 332–42. doi:10.1016/S1474-4422(06)70409-4. PMID 16545750.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b Zalmanovici Trestioreanu, A (2011 Aug 10). "Antibiotics for preventing meningococcal infections". Cochrane database of systematic reviews (Online) (8): CD004785. doi:10.1002/14651858.CD004785.pub4. PMID 21833949.
{{cite journal}}: Check date values in:|date=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b Ratilal, BO (2011 Aug 10). "Antibiotic prophylaxis for preventing meningitis in patients with basilar skull fractures". Cochrane database of systematic reviews (Online) (8): CD004884. doi:10.1002/14651858.CD004884.pub3. PMID 21833952.
{{cite journal}}: Check date values in:|date=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Prasad, K (2007 Oct 17). "Third generation cephalosporins versus conventional antibiotics for treating acute bacterial meningitis". Cochrane database of systematic reviews (Online) (4): CD001832. doi:10.1002/14651858.CD001832.pub3. PMID 17943757.
{{cite journal}}: Check date values in:|date=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ van de Beek D (2010). "Adjunctive dexamethasone in bacterial meningitis: a meta-analysis of individual patient data". Lancet Neurol. 9 (3): 254–63. doi:10.1016/S1474-4422(10)70023-5. PMC 2835871. PMID 20138011.
{{cite journal}}: Invalid|display-authors=3(help); Unknown parameter|author-separator=ignored (help); Unknown parameter|month=ignored (help) - ^ a b c d Brouwer MC, McIntyre P, de Gans J, Prasad K, van de Beek D (2010). Van De Beek, Diederik (ed.). "Corticosteroids for acute bacterial meningitis". Cochrane Database of Systematic Reviews (9): CD004405. doi:10.1002/14651858.CD004405.pub3. PMID 20824838. CD004405.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Assiri AM, Alasmari FA, Zimmerman VA, Baddour LM, Erwin PJ, Tleyjeh IM (2009). "Corticosteroid administration and outcome of adolescents and adults with acute bacterial meningitis: a meta-analysis". Mayo Clin. Proc. 84 (5): 403–9. doi:10.4065/84.5.403. PMC 2676122. PMID 19411436.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Prasad, K (2008 Jan 23). "Corticosteroids for managing tuberculous meningitis". Cochrane database of systematic reviews (Online) (1): CD002244. doi:10.1002/14651858.CD002244.pub3. PMID 18254003.
{{cite journal}}: Check date values in:|date=(help); Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b de Gans J, van de Beek D (2002). "Dexamethasone in adults with bacterial meningitis" (PDF). The New England Journal of Medicine. 347 (20): 1549–56. doi:10.1056/NEJMoa021334. PMID 12432041.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ McIntyre PB; Berkey CS; King SM; et al. (1997). "Dexamethasone as adjunctive therapy in bacterial meningitis. A meta-analysis of randomized clinical trials since 1988". Journal of the American Medical Association. 278 (11): 925–31. doi:10.1001/jama.1997.03550110063038. PMID 9302246.
{{cite journal}}: Unknown parameter|author-separator=ignored (help); Unknown parameter|month=ignored (help) - ^ "Meningitis and Encephalitis Fact Sheet". National Institute of Neurological Disorders and Stroke (NINDS). 2007-12-11. Retrieved 2009-04-27.
- ^ Gottfredsson M, Perfect JR (2000). "Fungal meningitis". Seminars in Neurology. 20 (3): 307–22. doi:10.1055/s-2000-9394. PMID 11051295.
- ^ "Mortality and Burden of Disease Estimates for WHO Member States in 2002" (xls). World Health Organization (WHO). 2002.
- ^ Richardson MP, Reid A, Tarlow MJ, Rudd PT (1997). "Hearing loss during bacterial meningitis" (PDF). Archives of Disease in Childhood. 76 (2): 134–38. doi:10.1136/adc.76.2.134. PMC 1717058. PMID 9068303.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Lapeyssonnie L (1963). "Cerebrospinal meningitis in Africa". Bulletin of the World Health Organization. 28: SUPPL:1–114. PMC 2554630. PMID 14259333.
- ^ Greenwood B (1999). "Manson Lecture. Meningococcal meningitis in Africa". Trans. R. Soc. Trop. Med. Hyg. 93 (4): 341–53. doi:10.1016/S0035-9203(99)90106-2. PMID 10674069.
- ^ a b c World Health Organization (1998). Control of epidemic meningococcal disease, practical guidelines, 2nd edition, WHO/EMC/BA/98 (PDF). Vol. 3. pp. 1–83.
- ^ WHO (2003). "Detecting meningococcal meningitis epidemics in highly-endemic African countries" (PDF). Weekly Epidemiological Record. 78 (33): 294–6. PMID 14509123.
- ^ a b Arthur Earl Walker, Edward R. Laws, George B. Udvarhelyi (1998). "Infections and inflammatory involvement of the CNS". The Genesis of Neuroscience. Thieme. pp. 219–21. ISBN 1-879284-62-6.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Whytt R (1768). Observations on the Dropsy in the Brain. Edinburgh: J. Balfour.
- ^ a b c Greenwood B (2006). "100 years of epidemic meningitis in West Africa – has anything changed?" (PDF). Tropical Medicine & International health: TM & IH. 11 (6): 773–80. doi:10.1111/j.1365-3156.2006.01639.x. PMID 16771997.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Vieusseux G (1806). "Mémoire sur le Maladie qui a regne à Génève au printemps de 1805". Journal de Médecine, de Chirurgie et de Pharmacologie (Bruxelles) (in French). 11: 50–53.
- ^ Weichselbaum A (1887). "Ueber die Aetiologie der akuten Meningitis cerebro-spinalis". Fortschrift der Medizin (in German). 5: 573–583.
- ^ Flexner S (1913). "The results of the serum treatment in thirteen hundred cases of epidemic meningitis" (PDF). J Exp Med. 17 (5): 553–76. doi:10.1084/jem.17.5.553. PMC 2125091. PMID 19867668.
- ^ a b Swartz MN (2004). "Bacterial meningitis—a view of the past 90 years". The New England Journal of Medicine. 351 (18): 1826–28. doi:10.1056/NEJMp048246. PMID 15509815.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Rosenberg DH, Arling PA (1944). "Penicillin in the treatment of meningitis". Journal of the American Medical Association. 125 (15): 1011–17. doi:10.1001/jama.1944.02850330009002. reproduced in Rosenberg DH, Arling PA (1984). "Penicillin in the treatment of meningitis". Journal of the American Medical Association. 251 (14): 1870–6. doi:10.1001/jama.251.14.1870. PMID 6366279.
{{cite journal}}: Unknown parameter|month=ignored (help)
External links
