Phenazine
| |

| |
| Names | |
|---|---|
| Preferred IUPAC name
Phenazine[1] | |
| Other names
Dibenzopyrazine
9,10-Diazaanthracene Azophenylene acridizine | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.001.995 |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C12H8N2 | |
| Molar mass | 180.21 g/mol |
| Appearance | yellow to brown crystalline powder |
| Density | 1.25g/cm3 |
| Melting point | 174–177 °C (345–351 °F; 447–450 K) |
| Boiling point | 357.2 °C (675.0 °F; 630.3 K) at 760 mmHg |
| insoluble in water | |
| Hazards | |
| Flash point | 160.3 °C (320.5 °F; 433.4 K) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Phenazine is an organic compound with the formula (C6H4)2N2. It is a dibenzo annulated pyrazine, and the parent substance of many dyestuffs, such as the toluylene red, indulines, and safranines (and the closely related eurhodines).[2] Phenazine crystallizes in yellow needles, which are only sparingly soluble in alcohol. Sulfuric acid dissolves it, forming a deep-red solution.
Synthesis
Classically phenazine are prepared by the reaction of nitrobenzene and aniline in the Wohl-Aue reaction. Other methods include:
- pyrolysis of the barium salt of azobenzoate
- oxidation of aniline with lead oxide
- oxidation of dihydrophenazine, which is prepared by heating pyrocatechin with o-phenylenediamine.
- oxidation of ortho-aminodiphenylamine with lead peroxide.
Derivatives
- The more complex phenazines, such as the naphthophenazines, naphthazines, and naphthotolazines, may be prepared by condensing ortho-diamines with ortho-quinones[3] or by the oxidation of an ortho-diamine in the presence of α-naphthol, and by the decomposition of ortho-anilido-(-toluidido- et cetera)- azo compounds with dilute acids.
- If alkyl or aryl-ortho-diamines be used, azonium bases are obtained. The azines are mostly yellow in color, distill unchanged and are stable to oxidants. They add on alkyl iodides readily, forming alkyl azonium salts, anhydride formation also taking place between these hydroxylgroups. It dissolves in concentrated sulfuric acid with a yellowish-green fluorescence.
- The rhodamines, which are closely related to the phthaleins, are formed by the condensation of the alkyl metaaminophenols with phthalic anhydride in the presence of sulfuric acid. Their salts are fine red dyes. By the entrance of amino or hydroxyl groups into the molecule dyestuffs are formed. The mono-amino derivatives or eurhodines are obtained when the arylmonamines are condensed with orthoamino azo compounds; by condensing quinone dichlorimide or para-nitrosodimethyl aniline with monamines containing a free para position, or by oxidizing ortho-hydroxydiaminodipbenylamines. They are yellowish-red solids, which behave as weak bases, their salts undergoing hydrolytic dissociation in aqueous solution. When heated with concentrated hydrochloric acid the amino group is replaced by the hydroxyl group and the phenolic eurhodols are produced.
Aminophenazine

Many aminophenazines are prominent dyes. Two of the first synthetic dyes are aminophenazines, these include induline and nigrosin.[2]
The symmetrical diaminophenazine is the parent substance of the important dyestuff neutral red (dimethyldiaminotoluphenazine). It is obtained by the oxidation of ortho-phenylenediamine with ferric chloride.
In a related process, oxidation of a cold mixture of para-aminodimethylaniline and meta-toluylenediamine gives toluylene blue. This indamine is formed as an intermediate product and passing into the red when boiled; and also by the oxidation of dimethylparaphenylene diatnine with metatoluylene diamine. It crystallizes in orange-red needles and its alcoholic solution fluoresces strongly. It dyes silk and mordanted cotton a fine scarlet. It is known commercially as neutral red. For the phenazonium salts, see safranine. Benzo[c]cinnoline is an isomer of phenazine, to which it bears the same relation that phenanthrene bears to anthracene.
Natural products
The known biological sources of phenazine compounds are mostly bacterial in nature.[4] Some of the genera known to produce phenazines include Pseudomonas spp., Streptomyces spp., and Pantoea agglomerans. These phenazine natural products have been implicated in the virulence and competitive fitness of producing organisms. For example, the phenazine pyocyanin produced by Pseudomonas aeruginosa contributes to its ability to colonise the lungs of cystic fibrosis patients. Similarly, phenazine-1-carboxylic acid, produced by a number of Pseudomonads, increases survival in soil environments and has been shown to be essential for the biological control activity of certain strains.[5][6][7]
While bacterial phenazines are principally involved in secondary metabolisms, methanophenazine in methanogenic archaea (methanogens) is involved in primary metabolisms and are important electron carriers.[8] Methanophenazine acts as the functional equivalent of menaquinones and ubiquinones in other organisms. Methanophenazine is only known phenazine of non-bacterial origin and also is the only phenazine that engages in primary metabolisms.[9]
Biosynthesis
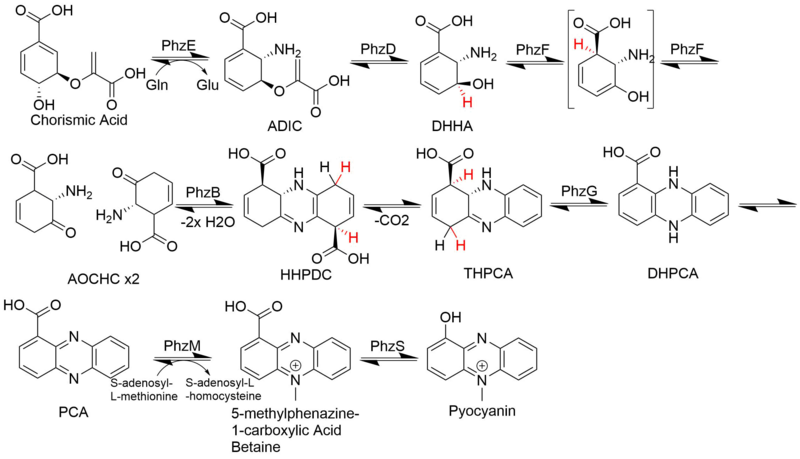
Phenazine biosynthesis branches off the shikimic acid pathway at a point subsequent to chorismic acid. Two molecules of this chorismate-derived intermediate are then brought together in a diagonally-symmetrical fashion to form the basic phenazine scaffold. Sequential modifications then lead to a variety of phenazine with differing biological activities. An example of phenazinic alkaloids are pyocyanin, saphenic acid and esmeraldins.
References
- ^ Nomenclature of Organic Chemistry : IUPAC Recommendations and Preferred Names 2013 (Blue Book). Cambridge: The Royal Society of Chemistry. 2014. p. 211. doi:10.1039/9781849733069-FP001. ISBN 978-0-85404-182-4.
- ^ a b Horst Berneth (2012). "Azine Dyes". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a03_213.pub3. ISBN 978-3527306732.
- ^ Alexander R. Surrey (1955). "Pyocyanine". Organic Syntheses; Collected Volumes, vol. 3, p. 753.
- ^ Pierson, Leland S.; Pierson, Elizabeth A. (2010). "Metabolism and function of phenazines in bacteria: impacts on the behavior of bacteria in the environment and biotechnological processes". Applied Microbiology and Biotechnology. 86 (6): 1659–1670. doi:10.1007/s00253-010-2509-3. ISSN 0175-7598. PMC 2858273. PMID 20352425.
- ^ Turner, J. M. & A. J. Messenger (1986). Occurrence, biochemistry, and physiology of phenazine pigment production. Advances in Microbial Physiology. Vol. 27. pp. 211–275. doi:10.1016/S0065-2911(08)60306-9. ISBN 978-0-12-027727-8. PMID 3532716.
- ^ McDonald, M., D. V. Mavrodi; et al. (2001). "Phenazine biosynthesis in Pseudomonas fluorescens: Branchpoint from the primary shikimate biosynthetic pathway and role of phenazine-1,6-dicarboxylic acid". J. Am. Chem. Soc. 123 (38): 9459–9460. doi:10.1021/ja011243. PMID 11562236.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Dietrich LE, Okegbe C, Price-Whelan A, Sakhtah H, Hunter RC, Newman DK (2013). "Bacterial community morphogenesis is intimately linked to the intracellular redox state" (PDF). Journal of Bacteriology. 195 (7): 1371–80. doi:10.1128/JB.02273-12. PMC 3624522. PMID 23292774.
- ^ Beifuss, Uwe; Tietze, Mario; Bäumer, Sebastian; Deppenmeier, Uwe (2000-07-17). "Methanophenazine: Structure, Total Synthesis, and Function of a New Cofactor from Methanogenic Archaea". Angewandte Chemie International Edition. 39 (14): 2470–2472. doi:10.1002/1521-3773(20000717)39:14<2470::aid-anie2470>3.0.co;2-r. ISSN 1433-7851. PMID 10941105.
- ^ Beifuss, Uwe; Tietze, Mario (2005-01-26), Mulzer, Johann (ed.), "Methanophenazine and Other Natural Biologically Active Phenazines", Natural Products Synthesis II, vol. 244, Berlin, Heidelberg: Springer Berlin Heidelberg, pp. 77–113, doi:10.1007/b96889, ISBN 978-3-540-21124-2, retrieved 2022-07-03
This article incorporates text from a publication now in the public domain: Chisholm, Hugh, ed. (1911). "Phenazine". Encyclopædia Britannica. Vol. 21 (11th ed.). Cambridge University Press. pp. 364–365.
