Acetabularia
| Acetabularia | |
|---|---|

| |
| Scientific classification | |
| (unranked): | Viridiplantae |
| Division: | Chlorophyta |
| Class: | Ulvophyceae |
| Order: | Dasycladales |
| Family: | Polyphysaceae |
| Genus: | Acetabularia Lamouroux, 1812[1] |
| Species | |
| |
Acetabularia is a genus of green algae in the family Polyphysaceae.[4] Typically found in subtropical waters, Acetabularia is a single-celled organism, but gigantic in size and complex in form, making it an excellent model organism for studying cell biology.[5] In form, the mature Acetabularia resembles the round leaves of a nasturtium, is 4 to 10 centimetres (1.6 to 3.9 in) tall and has three anatomical parts: a bottom rhizoid that resembles a set of short roots; a long stalk in the middle; and a top umbrella of branches that may fuse into a cap. Unlike other giant unicellular organisms, which are multinucleate, members of this genus possess a single nucleus located in the rhizoid, which allows the cell to regenerate completely if its cap is removed. The caps of two Acetabularia may also be exchanged, even from two different species. In addition, if a piece of the stem is removed, with no access to the nucleus in the rhizoid, this isolated stem piece will also grow a new cap.[6]

In the 1930s–1950s Joachim Hämmerling conducted experiments in which he demonstrated Acetabularia's genetic information is contained in the nucleus.[7] This was the first demonstration that genes are encoded by DNA in eukaryotes; earlier studies by Oswald Avery and others had shown that this was true for prokaryotes.
Etymology[edit]
The name, Acetabularia, derives from the Latin word acetabulum, a broad, shallow cup used for dipping bread; the upturned cap of Acetabularia resembles such a cup. For this reason, it is also sometimes called mermaid's wineglass.[8]
In the 19th century, the same designation Acetabularia was proposed by George Edward Massee for a genus of fungi (now Cyphellopus), but this usage is obsolete and considered invalid as the algal name takes precedence.[9]
Anatomy and life cycle[edit]
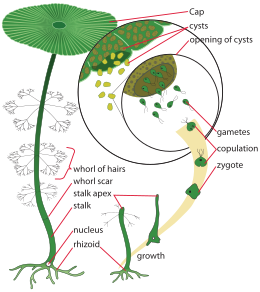
Acetabularia, as well as being unicellular, is also a uninucleate organism. It has three basic parts: its rhizoid, a short set of root-like appendages that contain the nucleus and anchor the cell to fissures in a substrate; its median stalk, which accounts for most of its length; and its apex, where its cap forms. There are usually several whorls of hair-like appendages close to the apex.[citation needed]
Acetabularia are among the largest single-celled organisms, having also a remarkably large nucleus. During sexual reproduction, the nucleus undergoes multiple rounds of mitosis, forming many daughter nuclei all within one nuclear membrane. These nuclei undergo meiosis and are transported to the tips of the branches, the sporangia, where they are released as gametes.[10]
Hämmerling's experiment[edit]
Each Acetabularia cell is composed of three segments: the "foot" or basal segment which contains the nucleus, the "stalk", and the "cap". Hämmerling exchanged caps between individuals from two species, A. mediterranea and A. crenulata. A. mediterranea has a smooth, disc-shaped cap, while A. crenulata has a branched, flower-like cap.
After the exchange, each transplanted cap gradually changed from its original form to the form typical for the species of the base it was now attached to. This showed that the nucleus controlled the form of the cap.
In another experiment, Hämmerling inserted a nucleus from one species of Acetabularia into an intact Acetabularia of a different species. The Acetabularia then produced a hybrid cap with characteristics of both species. This showed that both nuclei influenced the form of the cap.[7] Hammerling's results showed that the nucleus of a cell contains the genetic information that directs cellular development.
Morphogenesis[edit]
Although a single cell, Acetabularia exhibits a remarkably complex shape and has therefore long been a model organism for studying gene expression and morphogenesis. It seems to transport messenger RNA molecules (in an inactive riboprotein form) from the nucleus to its apical tips, where they are translated into proteins. These molecules may be activated by proteolysis of their protein carrier molecules, but this has not been verified as yet.[citation needed]
Internal chemical gradients[edit]
In addition to its gradient in specific mRNA molecules, Acetabularia exhibits concentration gradients in several types of molecules, such as ascorbic acid.[citation needed]
Circadian rhythms[edit]
Acetabularia has been used to study circadian rhythms.[11] Studies have shown Acetabularia has a diurnal circadian rhythm.[12] These rhythmic changes in respiratory and photosynthetic activity are maintained under constant conditions, even with the removal of the nucleus, showing the regulation of the rhythm is independent of the nucleus.[13] However, the nucleus isn't completely uninvolved, as it is responsible for the shifting of the cycles due to external changes. In one experiment a nucleus from a specimen trained on one circadian rhythm was transplanted into a de-nucleated plant on a rhythm that differed by 12 hours, over a period of days the donated nucleus changed the circadian rhythm of the receiving organism to that of the donor organism.[13]
Aquarium trade[edit]
Acetabularia species occasionally make their way into the aquarium trade. They are generally considered to be more difficult or unappealing macroalgae to care for in the reef aquarium, a fish-only, or a FOWLR (Fish Only With Live Rock) system, as they are delicate, readily eaten by herbivorous fish, grow slowly, and do not have the high nutrient uptake that reef aquarium refugium species (such as Chaetomorpha and Caulerpa) do. However, they are suitable for a macroalgae display tank, and thus macroalgae suppliers often carry species of Acetabularia.
See also[edit]
References[edit]
- ^ Lamouroux JVF (1812). "Extrait d'un mémoire sur la classification des Polypiers coralligènes non entièrement pierreux" [Extract from a dissertation on the classification of non-entirely stony coralligenous polyps] (PDF). Nouveaux Bulletin des Sciences, par la Société Philomathique de Paris (in French). 3: 181–188.
- ^ Saini, K. C.; Madhu, A.; Kohli, R. K.; Gupta, K.; Bast, F. (19 September 2022). "Morpho-molecular assessment of Acetabularia jalakanyakae Sp. Nov. (Dasycladales, Chlorophyta) - a new species from Andaman and Nicobar Islands, India". Indian Journal of Geo-Marine Sciences. 50 (9): 701–708. doi:10.56042/ijms.v50i09.42756. ISSN 2582-6727. S2CID 248837725.
- ^ "Indian scientists discover 'mermaid' plant species". BBC News. 17 August 2021.
- ^ Guiry, M.D.; Guiry, G.M. (2007). "Genus: Acetabularia taxonomy browser". AlgaeBase version 4.2 World-wide electronic publication, National University of Ireland, Galway. Retrieved 27 September 2007.
- ^ Mandoli, DF (1998). "Elaboration of Body Plan and Phase Change during Development of Acetabularia: How Is the Complex Architecture of a Giant Unicell Built?". Annual Review of Plant Physiology and Plant Molecular Biology. 49: 173–198. doi:10.1146/annurev.arplant.49.1.173. PMID 15012232.
- ^ B. Goodwin (1994) "How the Leopard Changed its Spots" Weidenfeld & Nicolson, London
- ^ a b Hämmerling, J. (1953). "Nucleo-cytoplasmic Relationships in the Development of Acetabularia". International Review of Cytology. 2: 475–498. doi:10.1016/S0074-7696(08)61042-6. ISBN 978-0-12-364302-5.
- ^ Lee, Robert E. (1999). Phycology. Cambridge, England: Cambridge University Press (CUP). pp. 217. ISBN 978-0-521-63883-8.
- ^ Index Fungorum.
- ^ Shihira-Ishikawa, I (1984). "Chromosome behavior in the primary nucleus of Acetabularia calyculus as revealed by epifluorescent microscopy". Protoplasma. 122 (1–2): 27–34. doi:10.1007/BF01279434. S2CID 24880240.
- ^ Sweeney, Beatrice M.; Tuffli, Charles F.; Rubin, Richard H. (1 January 1967). "The Circadian Rhythm in Photosynthesis in Acetabularia in the Presence of Actinomycin D, Puromycin, and Chloramphenicol". The Journal of General Physiology. 50 (3): 647–659. doi:10.1085/jgp.50.3.647. PMC 2225675. PMID 11526851.
- ^ Sweeney, B. M.; Haxo, F. T. (27 October 1961). "Persistence of a Photosynthetic Rhythm in Enucleated Acetabularia". Science. 134 (3487): 1361–1363. Bibcode:1961Sci...134.1361S. doi:10.1126/science.134.3487.1361. ISSN 0036-8075. PMID 17807341. S2CID 7270309.
- ^ a b Brachet, Jean (2 December 2012). Biology of Acetabularia. Elsevier. ISBN 978-0-323-15637-0.
Further reading[edit]
- Serio, Donatella; Alongi, Giuseppina; Catra, Marcello; Cormaci, Mario; Furnari, Giovanni (1 January 2006). "Changes in the benthic algal flora of Linosa Island (Straits of Sicily, Mediterranean Sea)". Botanica Marina. 49 (2). doi:10.1515/BOT.2006.018. S2CID 84853252.
- Berger, Sigrid; Liddle, Larry B. (March 2003). "The life cycle of Acetabularia (Dasycladales, Chlorophyta): textbook accounts are wrong". Phycologia. 42 (2): 204–207. doi:10.2216/i0031-8884-42-2-204.1. S2CID 84575637. ProQuest 198599797.
- Berger, Sigrid; Fettweiss, Ulrike; Gleissberg, Stefan; Liddle, Larry B.; Richter, Ursula; Sawitzky, Heiko; Zuccarello, Giuseppe C. (September 2003). "18S rDNA phylogeny and evolution of cap development in Polyphysaceae (formerly Acetabulariaceae; Dasycladales, Chlorophyta)". Phycologia. 42 (5): 506–561. doi:10.2216/i0031-8884-42-5-506.1. S2CID 83544153. ProQuest 198633648.
- Bonotto, Silvano (February 1994). "Developmental biology of Acetabularia". Journal of the Marine Biological Association of the United Kingdom. 74 (1): 93–106. doi:10.1017/S0025315400035694. S2CID 84105975.
- Berger S, de Groot E, Heuhaus G, Schweiger M (1987). "Acetabularia: a giant single cell organism with valuable advantages for cell biology". European Journal of Cell Biology. 44: 349–370.
- Puiseux-Dao S (1970). Acetabularia and Cell Biology. New York: Springer Verlag. ISBN 0-236-17738-9.
- Schweiger, H. G. (1969). "Cell Biology of Acetabularia". Current Topics in Microbiology and Immunology. Vol. 50. pp. 1–36. doi:10.1007/978-3-642-46169-9_1. ISBN 978-3-642-46171-2. PMID 4907132.
Sundry references[edit]
- Cinelli, F. (1979). "Acetabularia acetabulum (L.) Silva, Acetabularia parvula Solms-Laubach and Dasycladus vermicularis (Scopoli) Krasser (Chlorophyta, Dasycladales): ecology and distribution in the Mediterranean Sea". In Bonotto, Silvano; Kefeli, Valentin I.; Puiseux-Dao, Simone (eds.). Developmental Biology of acetabularia. Elsevier/North-Holland Biomedical Press. pp. 3–14. ISBN 978-0-444-80098-5.
- Cooper, John J.; Mandoli, Dina F. (February 1999). "Physiological Factors That Aid Differentiation of Zygotes and Early Juveniles of Acetabularia Acetabulum (Chlorophyta)". Journal of Phycology. 35 (1): 143–151. doi:10.1046/j.1529-8817.1999.3510143.x. S2CID 84315206.
- Dazy, A. C.; Borghi, H.; Durand, M.; Puiseux-Dao, S. (1984). "The effects of blue and red light on the transcellular electric potential, cytoplasmic streaming and rRNA transport in Acetabularia acetabulum". Eleventh International Seaweed Symposium. pp. 193–197. doi:10.1007/978-94-009-6560-7_33. ISBN 978-94-009-6562-1.
- Hunt, Brenda E.; Mandoli, Dina F. (June 1996). "A New, Artificial Sea Water That Facilitates Growth of Large Numbers of Cells of Acetabularia Acetabulum (Chlorophyta) and Reduces the Labor Inherent in Cell Culture1". Journal of Phycology. 32 (3): 483–495. doi:10.1111/j.0022-3646.1996.00483.x. S2CID 84001855.
- Kingsley, Roni J.; Van Gilder, Rachel; LeGeros, Racquel Z.; Watabe, Norimitsu (October 2003). "Multimineral Calcareous Deposits in the Marine Alga Acetabularia Acetabulum (Chlorophyta; Dasycladaceae)". Journal of Phycology. 39 (5): 937–947. doi:10.1046/j.1529-8817.2003.02169.x. S2CID 84484556.
- Kratz, Rene F.; Young, Philip A.; Mandoli, Dina F. (February 1998). "Timing and Light Regulation of Apical Morphogenesis During Reproductive Development in Wild-Type Populations of Acetabularia Acetabulum (Chlorophyceae)". Journal of Phycology. 34 (1): 138–146. doi:10.1046/j.1529-8817.1998.340138.x. S2CID 83759630.
- Lüttke, Angela (June 1988). "The Lack of Chloroplast DNA in Acetabularia Mediterranea (Acetabulum) (Chlorophyceae): A Reinvestigation". Journal of Phycology. 24 (2): 173–180. doi:10.1111/j.1529-8817.1988.tb04231.x. S2CID 86326553.
- Mandoli, Dina F.; Wexler, Adam; Teschmacher, Jill; Zukowski, Annette (October 1995). "Brief Incubation of Gametangia-Bearing Caps in Antibiotics Eliminates Branching in Progeny of Acetabularia Acetabulum (Chlorophyta)". Journal of Phycology. 31 (5): 844–848. doi:10.1111/j.0022-3646.1995.00844.x. S2CID 83754811.
- Menzel, Diedrik (March 1981). "Development and fine structure of plugs in the cap rays of Acetabularia acetabulum (mediterranea) (L.) Silva (Dasycladales)". Phycologia. 20 (1): 56–64. doi:10.2216/i0031-8884-20-1-56.1.
- Nishimura, N. J.; Mandoli, D. F. (May 1992). "Population analysis of reproductive cell structures of Acetabularia acetabulum (Chlorophyta)". Phycologia. 31 (3–4): 351–358. doi:10.2216/i0031-8884-31-3-4-351.1.
- Nishimura, Nathan J.; Mandoli, Dina F. (October 1992). "Vegetative Growth of Acetabularia Acetabulum (Chlorophyta): Structural Evidence for Juvenile and Adult Phases in Development1". Journal of Phycology. 28 (5): 669–677. doi:10.1111/j.0022-3646.1992.00669.x. S2CID 86616091.
- Zeller, A.; Mandoli, D. F. (March 1993). "Growth of Acetabularia acetabulum (Dasycladales, Chlorophyta) on solid substrata at specific cell densities". Phycologia. 32 (2): 136–142. doi:10.2216/i0031-8884-32-2-136.1.
External links[edit]
- www.accessexcellence.org
- Berger, Sigrid (2006).- Photo-Atlas of living Dasycladales.- ISBN 2-916733-01-9, Carnets de Géologie, Brest, Book / Livre 2006/02 (CG2006_BOOK_02), 348 p., 1 fig., 332 micrographs.
- UTEX culture collection of algae, at the University of Texas at Austin
- CCAP: Culture Collections of Algae and Protozoa
