Basic beryllium acetate

| |
| Names | |
|---|---|
| Systematic IUPAC name
Hexakis(μ-acetato)-μ(sup 4)-oxotetraberyllium | |
| Other names
Beryllium oxyacetate
Beryllium oxide acetate | |
| Identifiers | |
| ChemSpider | |
| ECHA InfoCard | 100.038.881 |
| EC Number |
|
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| C 12H 18Be 4O 13 | |
| Molar mass | 406.3122 g/mol |
| Appearance | colorless |
| Melting point | 285 °C (545 °F; 558 K) |
| Boiling point | 330 °C (626 °F; 603 K) |
| Solubility in chloroform | soluble |
| Hazards | |
| NIOSH (US health exposure limits): | |
PEL (Permissible)
|
TWA 0.002 mg/m3 C 0.005 mg/m3 (30 minutes), with a maximum peak of 0.025 mg/m3 (as Be)[1] |
REL (Recommended)
|
Ca C 0.0005 mg/m3 (as Be)[1] |
IDLH (Immediate danger)
|
Ca [4 mg/m3 (as Be)][1] |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Basic beryllium acetate is the chemical compound with the formula Be4O(O2CCH3)6. Although this compound has no applications and has been only lightly studied, it adopts a distinctive structure. The compound is a colourless solid that is soluble in organic solvents.
Preparation
It can be prepared by treating basic beryllium carbonate with hot acetic acid.
- 2 Be
2CO
3(OH)
2 + 6 AcOH → Be
4O(AcO)
6 + 5 H
2O + 2 CO
2
Basic beryllium acetate is insoluble in water but soluble in chloroform, consistent with it being nonpolar. It melts and sublimes in a vacuum without decomposition.[2]
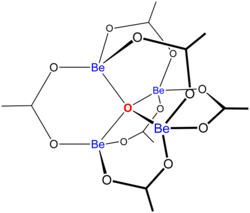
Structure
"Basic acetates" consist of an ensemble of metal centres bound to a central oxide ion, and a collection of acetate ligands. Basic beryllium acetate has a tetrahedral Be4O6+ core with acetates (CH3CO2−) spanning each of the pairs of Be2+ centres.[3][4] It is diamondoid, consisting of interlocking six-membered Be2O3C rings. The structure is relevant to its considerable stability (the compound is distillable at 330 °C).
See also
- Basic zinc acetate - isostructural
References
- ^ a b c NIOSH Pocket Guide to Chemical Hazards. "#0054". National Institute for Occupational Safety and Health (NIOSH).
- ^ Moeller, T. (1950). "Basic Beryllium Derivatives of Organic Acids". In Audrieth, L. F. (ed.). Inorganic Syntheses, Volume 3. John Wiley & Sons. p. 4. doi:10.1002/9780470132340.ch2. ISBN 978-0-470-13234-0.
- ^ Bragg, W. H. (1923). "Crystal Structure of Basic Beryllium Acetate". Nature. 111 (2790): 532. Bibcode:1923Natur.111..532B. doi:10.1038/111532a0.
- ^ Pauling, L.; Sherman, J. (1934). "The Structure of the Carboxyl Group. II. The Crystal Structure of Basic Beryllium Acetate". Proceedings of the National Academy of Sciences. 20 (6): 340. Bibcode:1934PNAS...20..340P. doi:10.1073/pnas.20.6.340.
