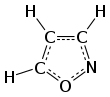
Isoxazole
| |||
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
1,2-oxazole
| |||
| Other names
isoxazole
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| ECHA InfoCard | 100.005.472 | ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| C3H3NO | |||
| Molar mass | 69.06202 g/mol | ||
| Density | 1.075 g/ml | ||
| Boiling point | 95 °C (203 °F; 368 K) | ||
| Acidity (pKa) | -3.0 (of conjugate acid) [1] | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Isoxazole is an azole with an oxygen atom next to the nitrogen. It is also the class of compounds containing this ring. Isoxazolyl is the univalent radical derived from isoxazole.
Occurrence
Isoxazole rings are found in some natural products, such as ibotenic acid and muscimol.
Pharmaceuticals and herbicides
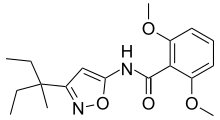
Isoxazoles also form the basis for a number of drugs,[2] including the COX-2 inhibitor valdecoxib (Bextra) and a neurotransmitter agonist AMPA. A derivative, furoxan, is a nitric oxide donor. An isoxazolyl group is found in many beta-lactamase-resistant antibiotics, such as cloxacillin, dicloxacillin and flucloxacillin. Leflunomide is an isoxazole-derivative drug. Examples of AAS containing the isoxazole ring include danazol and androisoxazole. A number of pesticides are isoxazoles.[3]
See also
- Oxazole, an analog with the nitrogen atom in position 3
- Pyrrole, an analog without the oxygen atom
- Furan, an analog without the nitrogen atom
- Simple aromatic rings
References
- ^ Zoltewicz, J. A. & Deady, L. W. Quaternization of heteroaromatic compounds. Quantitative aspects. Adv. Heterocycl. Chem. 22, 71-121 (1978).
- ^ "The recent progress of isoxazole in medicinal chemistry". Bioorganic & Medicinal Chemistry. 26 (12): 3065–3075. 2018. doi:10.1016/j.bmc.2018.05.013.
{{cite journal}}: Unknown parameter|authors=ignored (help) - ^ Clemens Lamberth (2018). "Oxazole and Isoxazole Chemistry in Crop Protection". Journal of Heterocyclic Chemistry. 55 (9): 2035–2045. doi:10.1002/jhet.3252.