3,3'-Diindolylmethane: Difference between revisions
Added https://scitechdaily.com/90-reduction-scientists-discover-natural-molecule-that-eradicates-plaques-and-cavities/ |
→Properties: Extra text |
||
| Line 58: | Line 58: | ||
[[In vitro]], DIM has action as a [[histone deacetylase inhibitor]],<ref name=lpi/> specifically against [[HDAC1]], [[HDAC2]], and [[HDAC3]].<ref name="dietaryncbi">{{cite journal | pmc = 3255482 | pmid=22247744 | doi=10.1186/1868-7083-3-4 | volume=3 | issue=1 | title=Dietary phytochemicals, HDAC inhibition, and DNA damage/repair defects in cancer cells | year=2011 | journal=Clin Epigenetics | pages=4 | last1 = Rajendran | first1 = P | last2 = Ho | first2 = E | last3 = Williams | first3 = DE | last4 = Dashwood | first4 = RH}}</ref> DIM is a [[metabolite]] of [[indole-3-carbinol]].<ref name=lpi/><ref name="msk">{{Cite web|url=https://www.mskcc.org/cancer-care/integrative-medicine/herbs/indole-3-carbinol|title=Indole-3-Carbinol|website=Memorial Sloan Kettering Cancer Center|language=en|access-date=2019-09-03}}</ref><ref name=pubchem/> DIM was found to be a mild cannabinoid agonist with low binding affinity for both CB1 and CB2.<ref>{{cite journal |author=Andhika B. Mahardhika |display-authors=et. al. |title=Design, synthesis, and structure–activity relationships of diindolylmethane derivatives as cannabinoid CB2 receptor agonists |journal=Arch Parm |date=March 2023 |volume=356 |issue=3 |doi=10.1002/ardp.202200493|doi-access=free }}</ref><ref>{{cite journal |author=Paolo Tucci |display-authors=et. al. |title=The Plant Derived 3-3′-Diindolylmethane (DIM) Behaves as CB2 Receptor Agonist in Prostate Cancer Cellular Models |journal=International Journal of Molecular Sciences |date=2023 |volume=24 |issue=4 |doi=10.3390/ijms24043620|hdl=2164/20232 |hdl-access=free }}</ref> |
[[In vitro]], DIM has action as a [[histone deacetylase inhibitor]],<ref name=lpi/> specifically against [[HDAC1]], [[HDAC2]], and [[HDAC3]].<ref name="dietaryncbi">{{cite journal | pmc = 3255482 | pmid=22247744 | doi=10.1186/1868-7083-3-4 | volume=3 | issue=1 | title=Dietary phytochemicals, HDAC inhibition, and DNA damage/repair defects in cancer cells | year=2011 | journal=Clin Epigenetics | pages=4 | last1 = Rajendran | first1 = P | last2 = Ho | first2 = E | last3 = Williams | first3 = DE | last4 = Dashwood | first4 = RH}}</ref> DIM is a [[metabolite]] of [[indole-3-carbinol]].<ref name=lpi/><ref name="msk">{{Cite web|url=https://www.mskcc.org/cancer-care/integrative-medicine/herbs/indole-3-carbinol|title=Indole-3-Carbinol|website=Memorial Sloan Kettering Cancer Center|language=en|access-date=2019-09-03}}</ref><ref name=pubchem/> DIM was found to be a mild cannabinoid agonist with low binding affinity for both CB1 and CB2.<ref>{{cite journal |author=Andhika B. Mahardhika |display-authors=et. al. |title=Design, synthesis, and structure–activity relationships of diindolylmethane derivatives as cannabinoid CB2 receptor agonists |journal=Arch Parm |date=March 2023 |volume=356 |issue=3 |doi=10.1002/ardp.202200493|doi-access=free }}</ref><ref>{{cite journal |author=Paolo Tucci |display-authors=et. al. |title=The Plant Derived 3-3′-Diindolylmethane (DIM) Behaves as CB2 Receptor Agonist in Prostate Cancer Cellular Models |journal=International Journal of Molecular Sciences |date=2023 |volume=24 |issue=4 |doi=10.3390/ijms24043620|hdl=2164/20232 |hdl-access=free }}</ref> |
||
Researchers from [[Ben-Gurion University of the Negev]], in collaboration with teams from [[Sichuan University]] and the [[National University of Singapore]], have identified that 3,3′-Diindolylmethane (DIM) – a naturally occurring molecule also referred to as bisindole – can reduce biofilms responsible for plaque and cavities by a remarkable 90%. <ref>{{cite journal |author=Yifat Baruch |display-authors=et. al. |title=3,3′-Diindolylmethane (DIM): A Potential Therapeutic Agent against Cariogenic ''Streptococcus mutans'' Biofilm |journal=Antibiotics 2023 |date=6 June 2023 |volume=12 |issue=6 |doi= |
Researchers from [[Ben-Gurion University of the Negev]], in collaboration with teams from [[Sichuan University]] and the [[National University of Singapore]], have identified that 3,3′-Diindolylmethane (DIM) – a naturally occurring molecule also referred to as bisindole – can reduce biofilms responsible for plaque and cavities by a remarkable 90%. <ref>{{cite journal |author=Yifat Baruch |display-authors=et. al. |title=3,3′-Diindolylmethane (DIM): A Potential Therapeutic Agent against Cariogenic ''Streptococcus mutans'' Biofilm |journal=Antibiotics 2023 |date=6 June 2023 |volume=12 |issue=6 |doi=10.3390/antibiotics12061017|doi-access=free }}</ref> |
||
==See also== |
==See also== |
||
Revision as of 18:31, 9 September 2023
This article needs more reliable medical references for verification or relies too heavily on primary sources. (June 2016) |  |
| |
| Names | |
|---|---|
| Preferred IUPAC name
3,3′-Methylenedi(1H-indole) | |
| Other names
3-(1H-Indol-3-ylmethyl)-1H-indole
3,3′-Methylenebis-1H-indole DIM | |
| Identifiers | |
3D model (JSmol)
|
|
| 223072 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.124.716 |
| EC Number |
|
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C17H14N2 | |
| Molar mass | 246.313 g·mol−1 |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335, H413 | |
| P261, P264, P271, P273, P280, P302+P352, P304+P340, P305+P351+P338, P312, P321, P332+P313, P337+P313, P362, P403+P233, P405, P501 | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
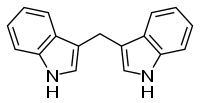
3,3′-Diindolylmethane (DIM) is a compound derived from the digestion of indole-3-carbinol, found in cruciferous vegetables, such as broccoli, Brussels sprouts, cabbage and kale.[1] It and its parent compound – indole-3-carbinol – are under laboratory research to determine their possible biological properties, particularly in anti-cancer mechanisms.[1][2][3] DIM is sold as a dietary supplement.[1]
Properties
In vitro, DIM has action as a histone deacetylase inhibitor,[1] specifically against HDAC1, HDAC2, and HDAC3.[4] DIM is a metabolite of indole-3-carbinol.[1][2][3] DIM was found to be a mild cannabinoid agonist with low binding affinity for both CB1 and CB2.[5][6]
Researchers from Ben-Gurion University of the Negev, in collaboration with teams from Sichuan University and the National University of Singapore, have identified that 3,3′-Diindolylmethane (DIM) – a naturally occurring molecule also referred to as bisindole – can reduce biofilms responsible for plaque and cavities by a remarkable 90%. [7]
See also
- Glucobrassicin, precursor to indole-3-carbinol
- Phytochemicals
References
- ^ a b c d e "Indole-3-carbinol". Micronutrient Information Center, Linus Pauling Institute, Oregon State University. 1 July 2017. Retrieved 11 July 2022.
- ^ a b "Indole-3-Carbinol". Memorial Sloan Kettering Cancer Center. Retrieved 2019-09-03.
- ^ a b "3,3'-Diindolylmethane". PubChem, US National Library of Medicine. 9 July 2022. Retrieved 11 July 2022.
- ^ Rajendran, P; Ho, E; Williams, DE; Dashwood, RH (2011). "Dietary phytochemicals, HDAC inhibition, and DNA damage/repair defects in cancer cells". Clin Epigenetics. 3 (1): 4. doi:10.1186/1868-7083-3-4. PMC 3255482. PMID 22247744.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Andhika B. Mahardhika; et al. (March 2023). "Design, synthesis, and structure–activity relationships of diindolylmethane derivatives as cannabinoid CB2 receptor agonists". Arch Parm. 356 (3). doi:10.1002/ardp.202200493.
- ^ Paolo Tucci; et al. (2023). "The Plant Derived 3-3′-Diindolylmethane (DIM) Behaves as CB2 Receptor Agonist in Prostate Cancer Cellular Models". International Journal of Molecular Sciences. 24 (4). doi:10.3390/ijms24043620. hdl:2164/20232.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Yifat Baruch; et al. (6 June 2023). "3,3′-Diindolylmethane (DIM): A Potential Therapeutic Agent against Cariogenic Streptococcus mutans Biofilm". Antibiotics 2023. 12 (6). doi:10.3390/antibiotics12061017.
External links
- Diindolylmethane Information Resource Center, University of California-Berkeley
