Vorinostat
 | |
| Clinical data | |
|---|---|
| Pronunciation | /vɒˈrɪnoʊstæt/ vorr-IN-oh-stat |
| Trade names | Zolinza |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a607050 |
| License data |
|
| Routes of administration | Oral (capsules) |
| ATC code | |
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Bioavailability | 1.8–11%[1] |
| Protein binding | ~71% |
| Metabolism | Hepatic glucuronidation and β-oxidation CYP system not involved |
| Metabolites | vorinostat O-glucuronide, 4-anilino-4-oxobutanoic acid (both inactive)[2] |
| Elimination half-life | ~2 hours (vorinostat and O-glucuronide), 11 hours (4-anilino-4-oxobutanoic acid) |
| Excretion | Renal (negligible) |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.207.822 |
| Chemical and physical data | |
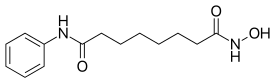
| Formula | C14H20N2O3 |
| Molar mass | 264.325 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Vorinostat (rINN),[3] also known as suberoylanilide hydroxamic acid (suberoyl+anilide+hydroxamic acid abbreviated as SAHA), is a member of a larger class of compounds that inhibit histone deacetylases (HDAC). Histone deacetylase inhibitors (HDI) have a broad spectrum of epigenetic activities.
Vorinostat is marketed under the name Zolinza (/zoʊˈlɪnzə/ zoh-LIN-zə) by Merck for the treatment of cutaneous manifestations in patients with cutaneous T cell lymphoma (CTCL) when the disease persists, gets worse, or comes back during or after two systemic therapies.[2][4] The compound was developed by Columbia University chemist Ronald Breslow and Memorial Sloan-Kettering researcher Paul Marks.[5][6]
Medical uses[edit]
Vorinostat was the first histone deacetylase inhibitor[7] approved by the U.S. Food and Drug Administration (FDA) for the treatment of CTCL on October 6, 2006.[8]
Development[edit]
In 1966, Charlotte Friend published her observation that a suspension of murine erythroleukemia cells underwent cytodifferentiation to normal erythrocytes when treated with dimethylsulfoxide (DMSO, a common drug solvent and cryoprotectant frequently used for cell culture freezing) at 280 mmolar.[9][10] Memorial Sloan-Kettering researcher Paul Marks approached Columbia University chemist Ronald Breslow about these findings and together they decided to develop more potent analogs of DMSO, in order to make use of this property for cancer treatment. Their optimization process lead to the discovery of suberoylanilide hydroxamic acid and its HDAC-inhibiting property.[6][11]
Mechanism of action[edit]
Vorinostat has been shown to bind to the active site of histone deacetylases and act as a chelator for zinc ions also found in the active site of histone deacetylases.[12] Vorinostat's inhibition of histone deacetylases results in the accumulation of acetylated histones and acetylated proteins, including transcription factors crucial for the expression of genes needed to induce cell differentiation.[12] It acts on class I, II and IV of histone deacetylase.
Clinical trials[edit]
Vorinostat has also been used to treat Sézary syndrome, another type of lymphoma closely related to CTCL.[13]
A recent study suggested that vorinostat also possesses some activity against recurrent glioblastoma multiforme, resulting in a median overall survival of 5.7 months (compared to 4–4.4 months in earlier studies).[14] Further brain tumor trials are planned in which vorinostat will be combined with other drugs.
Including vorinostat in treatment of advanced non-small-cell lung carcinoma (NSCLC) showed improved response rates and increased median progression free survival and overall survival.[15]
It has given encouraging results in a phase II trial[16] for myelodysplastic syndromes in combination with idarubicin and cytarabine.[17] It failed to demonstrate efficacy in treating acute myeloid leukemia in an earlier phase II study.[18]
Preclinical investigations[edit]
Vorinostat is being investigated as a potential HIV latency reversing agent (LRA) as part of an investigational therapeutic strategy known as "shock and kill".[19] Vorinostat was shown to reactivate HIV in latently HIV-infected T cells, both in vitro and in vivo.[20][21]
Vorinostat also has shown some activity against the pathophysiological changes in α1-antitrypsin deficiency[22] and cystic fibrosis.[23] Recent evidence also suggests vorinostat can be a therapeutic tool for Niemann-Pick type C1 (NPC1), a rare lysosomal lipid storage disease.[24]
Preclinical experiments by University of Alabama at Birmingham researchers suggest the cancer drugs vorinostat, belinostat and panobinostat might be repurposed to treat infections caused by human papillomavirus, or HPV.[25]
See also[edit]
References[edit]
- ^ "Withdrawal Assessment Report for Vorinostat MSD 100 mg Hard Capsules (vorinostat)" (PDF). European Medicines Agency. 23 October 2008. p. 9. Retrieved 1 September 2016.
- ^ a b "Zolinza (vorinostat) Capsules. Full Prescribing Information" (PDF). Merck & Co., Inc., Whitehouse Station, NJ 08889, USA. Retrieved 1 September 2016.
- ^ "International Nonproprietary Names for Pharmaceutical Substances (INN). Recommended International Nonproprietary Names: List 56" (PDF). WHO Drug Information. 20 (3): 232. 2006. Archived from the original (PDF) on July 5, 2011. Retrieved 1 September 2016.
- ^ "ZOLINZA, Merck's Investigational Medicine for Advanced Cutaneous T-Cell Lymphoma (CTCL), To Receive Priority Review from U.S. Food and Drug Administration" (Press release). Merck & Co. June 7, 2006. Archived from the original on September 14, 2006. Retrieved October 6, 2006.
- ^ Lee JH, Mahendran A, Yao Y, Ngo L, Venta-Perez G, Choy ML, et al. (September 2013). "Development of a histone deacetylase 6 inhibitor and its biological effects". Proceedings of the National Academy of Sciences of the United States of America. 110 (39): 15704–15709. Bibcode:2013PNAS..11015704L. doi:10.1073/pnas.1313893110. PMC 3785767. PMID 24023063.
- ^ a b Marks PA, Breslow R (January 2007). "Dimethyl sulfoxide to vorinostat: development of this histone deacetylase inhibitor as an anticancer drug". Nature Biotechnology. 25 (1): 84–90. doi:10.1038/nbt1272. PMID 17211407. S2CID 12656582.
- ^ "Vorinostat". HDAC Inhibitors Base.
- ^ "Zolinza (vorinostat) dosing, indications, interactions, adverse effects, and more". Medscape Reference. WebMD. Retrieved 16 February 2014.
- ^ Friend C, Patuleia MC, De Harven E (September 1966). "Erythrocytic maturation in vitro of murine (Friend) virus-induced leukemic cells". National Cancer Institute Monograph. 22: 505–522. PMID 5923328.
- ^ Friend C, Scher W, Holland JG, Sato T (February 1971). "Hemoglobin synthesis in murine virus-induced leukemic cells in vitro: stimulation of erythroid differentiation by dimethyl sulfoxide". Proceedings of the National Academy of Sciences of the United States of America. 68 (2): 378–382. Bibcode:1971PNAS...68..378F. doi:10.1073/pnas.68.2.378. PMC 388942. PMID 5277089.
- ^ Breslow R (2016-12-02). "From DMSO to the Anticancer Compound SAHA, an Unusual Intellectual Pathway for Drug Design". In Fischer J, Childers WE (eds.). Successful Drug Discovery. Weinheim, Germany: Wiley-VCH Verlag GmbH & Co. KGaA. pp. 1–11. doi:10.1002/9783527800315.ch1. ISBN 978-3-527-80031-5. Retrieved 2022-11-18.
- ^ a b Marks PA, Dokmanovic M (December 2005). "Histone deacetylase inhibitors: discovery and development as anticancer agents". Expert Opinion on Investigational Drugs. 14 (12): 1497–1511. doi:10.1038/sj.bjc.6603463. PMC 2360770. PMID 16307490.
- ^ Castoldi G, Cuneo A (May 2005). "Mycosis fungoides/Sezary's syndrome". Atlas of Genetics and Cytogenetics in Oncology and Haematology. Retrieved 2008-02-15.
- ^ "Vorinostat shows anti-cancer activity in recurrent gliomas" (Press release). Mayo Clinic. June 3, 2007. Retrieved 2007-06-03.
- ^ Ramalingam SS, Maitland ML, Frankel P, Argiris AE, Koczywas M, Gitlitz B, et al. (January 2010). "Carboplatin and Paclitaxel in combination with either vorinostat or placebo for first-line therapy of advanced non-small-cell lung cancer". Journal of Clinical Oncology. 28 (1): 56–62. doi:10.1200/JCO.2009.24.9094. PMC 2799233. PMID 19933908.
- ^ Garcia-Manero G, Tambaro FP, Bekele NB, Yang H, Ravandi F, Jabbour E, et al. (June 2012). "Phase II trial of vorinostat with idarubicin and cytarabine for patients with newly diagnosed acute myelogenous leukemia or myelodysplastic syndrome". Journal of Clinical Oncology. 30 (18): 2204–2210. doi:10.1200/JCO.2011.38.3265. PMC 4879705. PMID 22585696.
- ^ Langholtz J, Haehle M (11 January 2012). "Zolinza, Idarubicin, Cytarabine Combination Yields High Response Rates In MDS Patients (ASH 2011)". The MDS Beacon. Archived from the original on 2014-10-30. Retrieved 2012-01-17.
- ^ Schaefer EW, Loaiza-Bonilla A, Juckett M, DiPersio JF, Roy V, Slack J, et al. (October 2009). "A phase 2 study of vorinostat in acute myeloid leukemia". Haematologica. 94 (10): 1375–1382. doi:10.3324/haematol.2009.009217. PMC 2754953. PMID 19794082.
- ^ Clinical trial number NCT01319383 for "The Effect of Vorinostat on HIV RNA Expression in the Resting CD4+ T Cells of HIV+ Pts on Stable ART" at ClinicalTrials.gov
- ^ Archin NM, Espeseth A, Parker D, Cheema M, Hazuda D, Margolis DM (February 2009). "Expression of latent HIV induced by the potent HDAC inhibitor suberoylanilide hydroxamic acid". AIDS Research and Human Retroviruses. 25 (2): 207–212. doi:10.1089/aid.2008.0191. PMC 2853863. PMID 19239360.
- ^ Contreras X, Schweneker M, Chen CS, McCune JM, Deeks SG, Martin J, Peterlin BM (March 2009). "Suberoylanilide hydroxamic acid reactivates HIV from latently infected cells". The Journal of Biological Chemistry. 284 (11): 6782–6789. doi:10.1074/jbc.M807898200. PMC 2652322. PMID 19136668.
- ^ Bouchecareilh M, Hutt DM, Szajner P, Flotte TR, Balch WE (November 2012). "Histone deacetylase inhibitor (HDACi) suberoylanilide hydroxamic acid (SAHA)-mediated correction of α1-antitrypsin deficiency". The Journal of Biological Chemistry. 287 (45): 38265–38278. doi:10.1074/jbc.M112.404707. PMC 3488095. PMID 22995909.
- ^ Hutt DM, Herman D, Rodrigues AP, Noel S, Pilewski JM, Matteson J, et al. (January 2010). "Reduced histone deacetylase 7 activity restores function to misfolded CFTR in cystic fibrosis". Nature Chemical Biology. 6 (1): 25–33. doi:10.1038/nchembio.275. PMC 2901172. PMID 19966789.
- ^ Alam MS, Getz M, Haldar K (February 2016). "Chronic administration of an HDAC inhibitor treats both neurological and systemic Niemann-Pick type C disease in a mouse model". Science Translational Medicine. 8 (326): 326ra23. doi:10.1126/scitranslmed.aad9407. PMID 26888431. S2CID 5762569.
- ^ "Cancer drug may help treat human papillomavirus infections". ScienceDaily. 30 November 2018. Retrieved 2018-11-30.
External links[edit]
- Vorinostat bound to proteins in the PDB
