Fructose 1,6-bisphosphate: Difference between revisions
m →Iron Chelation: Task 3: Fix CS1 deprecated coauthor parameter errors |
m →Iron Chelation: Added 1 doi to a journal cite using AWB (10209) |
||
| Line 102: | Line 102: | ||
==Iron Chelation== |
==Iron Chelation== |
||
Fructose 1,6-bis(phosphate) has also been implicated in the ability to bind and sequester Fe(II), a soluble form of iron whose oxidation to the insoluble Fe(III) is capable of generating reactive oxygen species via [[Fenton chemistry|Fenton]] chemistry. The ability of fructose 1,6-bis(phosphate) to bind Fe(II) may prevent such electron transfers, and thus act as an antioxidant within the body. Certain neurodegenerative diseases, like [[Alzheimer's disease|Alzheimer's]] and [[Parkinson's disease|Parkinson's]], have been linked to metal deposits with high iron content, although it is uncertain whether Fenton chemistry plays a substantial role in these diseases, or whether fructose 1,6-bis(phosphate) is capable of mitigating those effects.<ref>{{cite journal|last=Bajic|first=Aleksandar|author2=Zakrzewska J |author3=Godjevac D |author4=Andjus P |author5=Jones DR |author6=Spasic M |author7=Spasojevic I |title=Relevance of the ability of fructose 1,6-bis(phosphate) to sequester ferrous but not ferric ions|journal=Carbohydrate Research|year=2011|volume=346|pages=416–420}}</ref> |
Fructose 1,6-bis(phosphate) has also been implicated in the ability to bind and sequester Fe(II), a soluble form of iron whose oxidation to the insoluble Fe(III) is capable of generating reactive oxygen species via [[Fenton chemistry|Fenton]] chemistry. The ability of fructose 1,6-bis(phosphate) to bind Fe(II) may prevent such electron transfers, and thus act as an antioxidant within the body. Certain neurodegenerative diseases, like [[Alzheimer's disease|Alzheimer's]] and [[Parkinson's disease|Parkinson's]], have been linked to metal deposits with high iron content, although it is uncertain whether Fenton chemistry plays a substantial role in these diseases, or whether fructose 1,6-bis(phosphate) is capable of mitigating those effects.<ref>{{cite journal|last=Bajic|first=Aleksandar|author2=Zakrzewska J |author3=Godjevac D |author4=Andjus P |author5=Jones DR |author6=Spasic M |author7=Spasojevic I |title=Relevance of the ability of fructose 1,6-bis(phosphate) to sequester ferrous but not ferric ions|journal=Carbohydrate Research|year=2011|volume=346|pages=416–420 |doi=10.1016/j.carres.2010.12.008}}</ref> |
||
==See also== |
==See also== |
||
Revision as of 08:15, 24 May 2014
This article needs additional citations for verification. (December 2009) |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| ECHA InfoCard | 100.006.985 |
| MeSH | fructose-1,6-diphosphate |
PubChem CID
|
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C6H14O12P2 | |
| Molar mass | 340.116 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
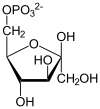
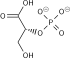
Fructose 1,6-bisphosphate, also known as Harden-Young ester, is fructose sugar phosphorylated on carbons 1 and 6 (i.e., is a fructosephosphate). The β-D-form of this compound is very common in cells. The vast majority of glucose and fructose entering a cell will become converted to fructose 1,6-bisphosphate at some point.
Fructose 1,6-bisphosphate in glycolysis
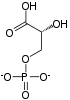
Fructose 1,6-bisphosphate lies within the glycolysis metabolic pathway and is produced by phosphorylation of fructose 6-phosphate. It is, in turn, broken down into two compounds: glyceraldehyde 3-phosphate and dihydroxyacetone phosphate. It is an allosteric activator of pyruvate kinase.
Compound C05345 at KEGG Pathway Database. Enzyme 2.7.1.11 at KEGG Pathway Database. Enzyme 3.1.3.11 at KEGG Pathway Database. Compound C05378 at KEGG Pathway Database. Enzyme 4.1.2.13 at KEGG Pathway Database. Compound C00111 at KEGG Pathway Database. Compound C00118 at KEGG Pathway Database.
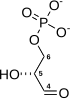
The numbering of the carbon atoms indicates the fate of the carbons according to their position in fructose 6-phosphate.
Click on genes, proteins and metabolites below to link to respective articles.[§ 1]
- ^ The interactive pathway map can be edited at WikiPathways: "GlycolysisGluconeogenesis_WP534".
Fructose 1,6-bisphosphate isomerism
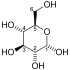
Fructose 1,6-bisphosphate has only one biologically active isomer, the β-D-form. There are many other isomers, analogous to those of fructose.
Iron Chelation
Fructose 1,6-bis(phosphate) has also been implicated in the ability to bind and sequester Fe(II), a soluble form of iron whose oxidation to the insoluble Fe(III) is capable of generating reactive oxygen species via Fenton chemistry. The ability of fructose 1,6-bis(phosphate) to bind Fe(II) may prevent such electron transfers, and thus act as an antioxidant within the body. Certain neurodegenerative diseases, like Alzheimer's and Parkinson's, have been linked to metal deposits with high iron content, although it is uncertain whether Fenton chemistry plays a substantial role in these diseases, or whether fructose 1,6-bis(phosphate) is capable of mitigating those effects.[1]
See also
References
- ^ Bajic, Aleksandar; Zakrzewska J; Godjevac D; Andjus P; Jones DR; Spasic M; Spasojevic I (2011). "Relevance of the ability of fructose 1,6-bis(phosphate) to sequester ferrous but not ferric ions". Carbohydrate Research. 346: 416–420. doi:10.1016/j.carres.2010.12.008.