Coniine
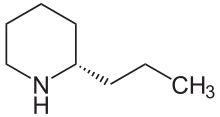 (S)-Coniine
| |
| |
| Names | |
|---|---|
| IUPAC name
(2S)-2-Propylpiperidine
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI |
|
| ChEMBL |
|
| ChemSpider |
|
| ECHA InfoCard | 100.006.621 |
| EC Number |
|
| KEGG |
|
PubChem CID
|
|
| UNII |
|
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C8H17N | |
| Molar mass | 127.231 g·mol−1 |
| Appearance | Colorless, oily liquid |
| Melting point | −2 °C (28 °F; 271 K) |
| Boiling point | 166 to 167 °C (331 to 333 °F; 439 to 440 K) |
Refractive index (nD)
|
1.4505 |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Coniine is a poisonous chemical compound, an alkaloid present in and isolable from poison hemlock (Conium maculatum), where its presence has been a source of significant economic, medical, and historico-cultural interest; coniine is also produced by the yellow pitcher plant (Sarracenia flava), and fool's parsley (Aethusa cynapium). Its ingestion and extended exposure are toxic to humans and all classes of livestock; its mechanism of poisoning involves disruption of the central nervous system, with death caused by respiratory paralysis. The biosynthesis of coniine contains as its penultimate step the non-enzymatic cyclisation of 5-oxooctylamine to γ-coniceine, a Schiff base differing from coniine only by its carbon-nitrogen double bond in the ring. This pathway results in natural coniine that is a mixture—a racemate—composed of two enantiomers, the stereoisomers (S)-(+)-coniine and (R)-(−)-coniine, depending on the direction taken by the chain that branches from the ring. Both enantiomers are toxic, with the (R)-enantiomer being the more biologically active and toxic of the two in general. Coniine holds a place in organic chemistry history as being the first of the important class of alkaloids to be synthesized, by Albert Ladenburg in 1886, and it has been synthesized in the laboratory in a number of unique ways through to modern times.
Hemlock poisoning has been a periodic human concern, a regular veterinary concern, and has had significant occurrences in human and cultural history. Notably, in 399 BC, Socrates was sentenced to death by drinking a coniine-containing mixture of poison hemlock.
Natural origins
[edit]
Poison hemlock (Conium maculatum) contains highly toxic amounts of coniine. Its presence on farmland is an issue for livestock farmers because animals will eat it if they are not well fed or the hemlock is mixed in with pasture grass.[1] The coniine is present in Conium maculatum as a mixture of the R-(−)- and S-(+)-enantiomers.[2]

Coniine is also found in Sarracenia flava, the yellow pitcher plant.[3][4] The yellow pitcher plant is a carnivorous plant endemic to the southeastern United States. The plant uses a mixture of sugar and coniine to simultaneously attract and poison insects, which then fall into a digestive tube.[5] Coniine is also found in Aethusa cynapium, commonly known as fool's parsley.[6]
History of natural isolates
[edit]The history of coniine is understandably tied to the poison hemlock plant, since the natural product was not synthesizable until the 1880s.[7] Jews in the Middle East were poisoned by coniine after consuming quail in the area that usually ate hemlock seeds, and Greeks on the island of Lesbos who also consumed quail suffered from the same poisoning, causing myoglobinuria and acute kidney injury.[8] The most famous hemlock poisoning occurred in 399 BCE, when the philosopher Socrates is believed to have consumed a liquid infused with hemlock to carry out his death sentence, his having been convicted of impiety toward the gods, and the corruption of youth.[9][10][11] Hemlock juice was often used to execute criminals in ancient Greece.[12]
Hemlock has had a limited medical use throughout history. The Greeks used it not just as capital punishment, but also as an antispasmodic and treatment for arthritis. Books from the 10th century attest to medical use by the Anglo-Saxons.[13] In the Middle Ages it was believed that hemlock could be used to cure rabies; in later European times it came to be associated with flying ointments in witchcraft. Native Americans used hemlock extract as arrow poison.[14]
While the yellow pitcher plant and fool's parsley also contain coniine, there are no reports of traditional uses for these plants.
Pharmacology and toxicology
[edit]The (R)-(−) enantiomer of coniine is the more biologically active, at least in one system (TE-671 cells expressing human fetal nicotinic neuromuscular receptors), and in mouse bioassay, the same enantiomer and the racemic mixture are about two-fold more toxic than the (S)-(+) enantiomer (see below).[2]
Coniine, as racemate or as pure enantiomer, begins by binding and stimulating the nicotinic receptor on the post-synaptic membrane of the neuromuscular junction. The subsequent depolarization results in nicotinic toxicity; as coniine stays bound to the receptor, the nerve stays depolarized, inactivating it.[15] This results, systemically, in a flaccid paralysis, an action similar to that of succinylcholine since they are both depolarizing neuromuscular blockers. Symptoms of paralysis generally occur within a half-hour, although death may take several hours. The central nervous system is not affected: the person remains conscious and aware until respiratory paralysis results in cessation of breathing. The flaccid, muscular paralysis is an ascending paralysis, lower limbs being first affected. The person may have a hypoxic convulsion just prior to death, disguised by the muscular paralysis such that the person may just weakly shudder. Cause of death is lack of oxygen to the brain and heart as a consequence of respiratory paralysis, so that a poisoned person may recover if artificial ventilation can be maintained until the toxin is removed from the victim's system.
The LD50 values (in mouse, i.v. administered) for the R-(−) and S-(+) enantiomers, and the racemate, are approximately 7 and 12, and 8 milligrams per kilogram, respectively.[2]
Chemical properties
[edit](+/–)-Coniine was first isolated by Giesecke,[16] but the formula was suggested by Blyth[17] and definitely established by Hofmann.[18][19]
D-(S)-Coniine has since been determined to be a colorless alkaline liquid, with a penetrating odour and a burning taste; has D0° 0.8626 and D19° 0.8438, refractive index n23°D 1.4505, and is dextrorotatory, [α]19°D +15.7° (see related comments under Specific rotation section below). L-(R)-Coniine has [α]21°D 15° and in other respects resembles its D-isomer, but the salts have slightly different melting points; the platinichloride has mp. 160 °C (Löffler and Friedrich report 175 °C), the aurichloride mp. 59 °C.[20][21]
Solubility
[edit]Coniine is slightly soluble (1 in 90) in cold water, less so in hot water, so that a clear cold solution becomes turbid when warmed. On the other hand, the base dissolves about 25% of water at room temperature. It mixes with alcohol in all proportions, is readily soluble in ether and most organic solvents. Coniine dissolves in carbon disulfide, forming a complex thiocarbamate.[22][23]
Crystallization
[edit]This section relies largely or entirely on a single source. (January 2017) |
Coniine solidifies into a soft crystalline mass at −2 °C. It slowly oxidizes in the air. The salts crystallize well and are soluble in water or alcohol. The hydrochloride, B•HCl, crystallizes from water in rhombs, mp. 220 °C, [α]20°D +10.1°; the hydrobromide, in needles, mp. 211 °C, and the D-acid tartrate, B•C4H6O6•2 H2O, in rhombic crystals, mp. 54 °C. The platinichloride, (B•HCl)2•PtCl4•H2O, separates from concentrated solution as an oil, which solidifies to a mass of orange-yellow crystals, mp. 175 °C (dry). The aurichloride, B•HAuCl4, crystallizes on standing, mp. 77 °C. The picrate forms small yellow needles, mp. 75 °C, from hot water. The 2,4-dinitrobenzoyl- and 3,5-dinitrobenzoyl-derivates have mps. 139.0–139.5 °C and 108–9 °C respectively.[24] The precipitate afforded by potassium cadmium iodide solution is crystalline, mp. 118 °C, while that given by nicotine with this reagent is amorphous.
Color changes
[edit]Coniine gives no coloration with sulfuric or nitric acid. Sodium nitroprusside gives a deep red color, which disappears on warming, but reappears on cooling, and is changed to blue or violet by aldehydes.[25]
Specific rotation
[edit]The stereochemical composition of "coniine" is a matter of some importance, since its two enantiomers do not have identical biological properties,[2] and many of the older pharmacological studies on this compound were carried out using the naturally-occurring isomeric mixture.[citation needed] S-(+)-Coniine has a specific rotation, [α]D, of +8.4° (c = 4.0, in CHCl3).[26] These authors note that Ladenburg's value,[27] +15°, is for a "neat", i.e. undiluted, sample. A similarly high value of +16° for the [α]D of "coniine" is given, without explicit citation of the source, in The Merck Index.[28] The value of +7.7° (c = 4.0, CHCl3) for synthetic S-(+)-coniine and -7.9° (c = 0.5, CHCl3) for synthetic R-(−)-coniine is given by other chemists.[29] The hydrochloride salts of the (S)-(+) and (R)-(−) enantiomers of coniine have values of [α]D of +4.6° and -5.2°, respectively (c = 0.5, in methanol).[2]
Synthesis
[edit]The original synthesis (shown below) of Coniine was performed by Ladenburg in 1886.[30] Ladenburg heated N-methylpyridine iodide to 250 °C, to obtain 2-methylpyridine. He then performed a Knoevenagel condensation with acetaldehyde in anhydrous zinc chloride to yield 2-propenylpyridine. In fact, Ladenburg used paraldehyde, a cyclic trimer of acetaldehyde that readily forms acetaldehyde upon heating. Finally, 2-propenylpyridine was reduced with metallic sodium in ethanol to provide racemic (±) coniine. Fractional crystallisation of racemic coniine with (+)-tartaric acid yielded enantiopure coniine.

The scheme proposed by Ladenburg gave poor yields, so the quest for alternative routes was open. A slightly better yield is observed if 2-methylpyridine and acetaldehyde are heated in a sealed tube with hydrochloric acid for 10 hours. A mixture of 2-propenylpyridine and 2-chloropropylpyridine is formed and is subsequently reduced by sodium in ethanol to give rac-coniine. Note: although the scheme below shows a single enantiomer of coniine, the final reaction produces a racemic mixture that is then separated
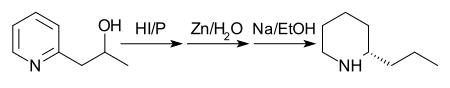
In 1907, another route with better yield was proposed. First, 2-(2'-hydroxypropyl)pyridine is reduced with phosphorus and fuming hydroiodic acid at 125 °C. Second, the product is treated with zinc dust and water. Finally, the product of the second step is treated with sodium in ethanol.[31] Note: although the graphic below shows a single enantiomer of coniine, this reaction produces a racemic mixture that is then purified and separated.
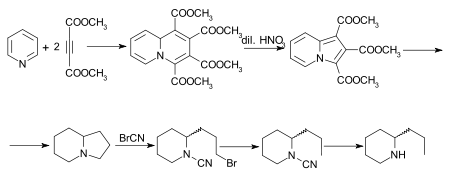
A number of other syntheses of coniine have been effected,[32] of which that of Diels and Alder is of special interest.[33] The initial adduct of pyridine and dimethyl acetylenedicarboxylate is tetramethylquinolizine-1,2,3,4-tetracarboxylate, which on oxidation with dilute nitric acid is converted into trimethyl indolizine-tricarboxylate. This, on hydrolysis and decarboxylation, furnishes indolizine, the octahydro-derivate of which, also known as octahydropyrrocoline[34] is converted by the cyanogen bromide method successively into the bromocyanamide, cyanamide and rac.-coniine. A synthesis of the alkaloid, starting from indolizine (pyrrocoline) is described by Ochiai and Tsuda.[35]
The preparation of L-(R)-coniine by the reduction of β-coniceine (L-propenylpiperidine) by Löffler and Friedrich[21] provides means for converting conhydrine to L-(R)-coniine.[36] Hess and Eichel reported,[37] incorrectly,[38] that pelletierine was the aldehyde (β-2-piperidyl-propaldehyde) corresponding to coniine, and yielded rac-coniine when its hydrazone was heated with sodium ethoxide in ethanol at 156–170 °C. According to these authors, D-(S)-coniine is rendered almost optically inactive when heated with barium hydroxide and alcohol at 180–230 °C. Leithe[39] has shown by observation of the optical rotation of (+)-pipecolic acid (piperidine-2-carboxylic acid) and some of its derivatives under varying conditions,[40] that it must belong to the D-series of amino acids.
Currently, Coniine, and many other alkaloids, can be synthesized stereoselectively.[29] For example, Pd-catalyzed 1,3-chirality transfer reaction can stereospecifically transform a single enantiomer of an allyl alcohol into a cyclic structure (in this case a piperidine).[41] In this way, starting from (S)-alcohol an (S)-enantiomer of Coniine is obtained and vice versa. Remarkably, the separation of racemic alcohol into different enantiomers is done with the help of Candida antarctica lipase.
Biosynthesis
[edit]The biosynthesis of coniine is still being investigated, but much of the pathway has been elucidated. Originally thought to use 4 acetyl groups as feed compounds for the polyketide synthase that forms coniine,[42] it is in fact derived from two malonyl and a butyryl CoA, which are derived in the usual way from acetyl-CoA.[43]
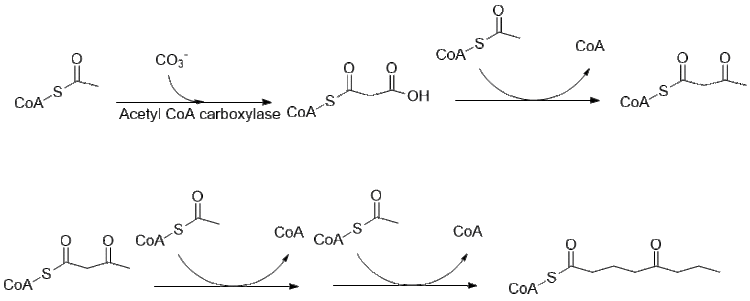
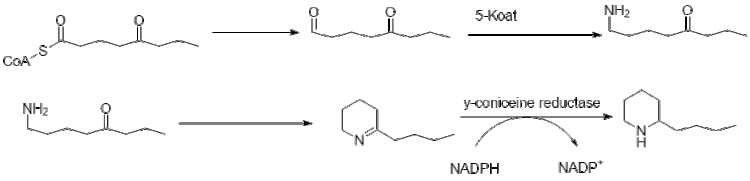
Further elongation of butyryl-CoA using 2 malonyl-CoA forms 5-ketooctanal. Ketooctanal then undergoes transamination using alanine:5-keto-octanal aminotransferase.[44] The amine then spontaneously cyclizes and is dehydrated to form the coniine precursor γ–coniceine. This is then reduced using NADPH dependent y-coniceine reductase to form coniine.
In popular culture
[edit]Coniine is the murder weapon in Agatha Christie's mystery novel Five Little Pigs.
The R and S 2-Propylpiperidine stereoisomers are a neurotoxin present in a slug-like lifeform in The Expanse. The toxin is shown as causing almost instant death upon skin contact in the show.
References
[edit]- ^ Peters, Amy; Bouska, Cassie. "Poison Hemlock". Osu Extension Service. Retrieved 3 May 2015.
- ^ a b c d e Stephen T. Lee; Benedict T. Green; Kevin D. Welch; James A. Pfister; Kip E. Panter (2008). "Stereoselective potencies and relative toxicities of coniine enantiomers". Chemical Research in Toxicology. 21 (10): 2061–2064. doi:10.1021/tx800229w. PMID 18763813.
- ^ N. V. Mody; R. Henson; P. A. Hedin; U. Kokpol; D. H. Miles (1976). "Isolation of the insect paralyzing agent coniine from Sarracenia flava". Experientia. 32 (7): 829–830. doi:10.1007/BF02003710. S2CID 38319708.
- ^ Hotti, Hannu; Rischer, Heiko (2017). "The killer of Socrates: Coniine and Related Alkaloids in the Plant Kingdom". Molecules. 22 (11): 1962. doi:10.3390/molecules22111962. PMC 6150177. PMID 29135964.
- ^ Mackie, Robin. "Yellow Pitcher Plant or Trumpets". United States Department of Agriculture. United States Department of Agriculture Forest Service. Retrieved 3 May 2015.
- ^ Clapham, Tutin, & Warburg. Flora of the British Isles (2nd ed.). p. 524.
{{cite book}}: CS1 maint: multiple names: authors list (link) - ^ Chisholm, Hugh, ed. (1911). . Encyclopædia Britannica. Vol. 6 (11th ed.). Cambridge University Press. p. 942.
- ^ "Massachusetts Medical Society: Don't Eat the Quails". www.massmed.org. Retrieved 2021-07-30.
- ^ James Warren (2001). "Socratic suicide". Journal of Hellenic Studies. 121: 91–106. doi:10.2307/631830. JSTOR 631830. PMID 19681231. S2CID 24221544.
- ^ R. G. Frey (1978). "Did Socrates commit suicide?". Philosophy. 53 (203): 106–108. doi:10.1017/S0031819100016375. S2CID 170399183.
- ^ Jackson, Henry (1911). . In Chisholm, Hugh (ed.). Encyclopædia Britannica. Vol. 25 (11th ed.). Cambridge University Press. pp. 331–338.
SOCRATES, son of the statuary Sophroniscus and of the midwife Phaenarete, was born at Athens, not earlier than 471 nor later than May or June 469 B.C. ... In 399, four years after the restoration and the amnesty, he was indicted as an offender against public morality. ... The accusation ran thus: "Socrates is guilty, firstly, of denying the gods recognized by the state and introducing new divinities, and, secondly, of corrupting the young." ... Under ordinary circumstances the condemned criminal drank the cup of hemlock on the day after the trial; but in the case of Socrates the rule that during the absence of the sacred ship sent annually to Delos no one should be put to death caused an exceptional
- ^ "The Suicide of Socrates". EyeWitness to History. Retrieved 3 May 2015.
- ^ Grieve, M. (1971). A Modern Herbal (2nd ed.). Mineola, N.Y.: Dover Publications. p. 392. ISBN 978-0-486-22798-6. Retrieved 3 May 2015.
- ^ Moser, L; Crisp, D. "Poison Hemlock" (PDF). San Francisco Peaks Weed Management. Retrieved 3 May 2015.
- ^ Goldrank, Lewis; Nelson, Lewis. Toxicologic Emergencies (11th ed.). New York: McGraw-Hill. pp. Chapter 118: Plants.
- ^ Giseke, Aug. Lud. (1827) "Ueber das wirksame Princip des Schierlings, Conium maculatum" (On the active component of hemlock, Conium maculatum), Archiv der Pharmazie, 20 (2) : 97–111. On p. 99, Giseke credits the Swiss apothecary Peschier with coining the name coniin (coniine). See footnote on p. 87 of: Peschier (1821) "Neue analytische Untersuchungen über den unter verschiedenen Himmelsstrichen gebauten Mohn; ferner über einige inländische Narcotica, und Entdeckung neuer Pflanzensäuren und Alkälien in denselben" (New analytical investigations into poppies grown in various climates; furthermore, on some domestic narcotics, and discovery of new plant acids and alkalis in the same), Neues Journal der Pharmacie für Aerzte, Apotheker und Chemiker, 5 (1) : 76–101. From p. 87: "Eine Abbildung der krystallisirten Säure s. Fig. 1 das coniumsaure Natron ist Fig. 2 abgebildet." (An illustration of the crystalline acid, see Fig. 1 ; the sodium salt of conium acid is depicted in Fig. 2.)
- ^ Blyth, J. (1849) "On the composition of coniine, and its products of decomposition," Quarterly Journal of the Chemical Society of London, 1 : 345–363. Blyth found the empirical formula of coniine to be (p. 351): C17H17N. The error in the amount of carbon is due, in part, to his having assumed that the atomic mass of carbon is 6, not 12 — a common error at the time.
- ^ Hofmann, A. W. (1881) "Einwirkung der Wärme auf die Ammoniumbasen: 2. Coniin" (Effect of heat on ammonium bases: 2. Coniine), Berichte der deutschen chemischen Gesellschaft, 14 : 705–713.
- ^ Panter, K. E. and Keeler, R. F., Ch. 5: Piperidine alkaloids of poison hemlock (Conium maculatum) in: Cheeke, Peter R., ed., Toxicants of Plant Origin: Alkaloids, vol. 1 (Boca Raton, Florida: CRC Press, Inc., 1989), p. 116.
- ^ Ahrens, Ber., 1902, 35, 1330
- ^ a b Löffler and Friedrich, Ber., 1909, 42, 107.
- ^ Melzer, Arch. Pharm., 1898, 236, 701
- ^ cf. Dilling, Pharm. J., 1909, [iv], 29, 34, 70, 102.
- ^ Späth, Kuffner and Ensfellner, Ber., 1933, 66, 596.
- ^ Gabutti, Chem. Soc. Abstr., 1906, [ii], 711.
- ^ Craig J. Cymerman; A. R. Pinder (1971). "Improved method resolution of coniine". Journal of Organic Chemistry. 36 (23): 3648–3649. doi:10.1021/jo00822a051.
- ^ A. Ladenburg (1888) Justus Liebig's Ann. Chem. 247 1-98.
- ^ The Merck Index, 15th Ed. (2013), p. 446, Monograph 2489, O'Neil: The Royal Society of Chemistry. http://www.rsc.org/Merck-Index/monograph/mono1500002489
- ^ a b D. Enders and J. Tiebes (1993) Liebig's Ann. Chem. 173-177.
- ^ Ladenburg, A. (1886-01-01). "Versuche zur Synthese des Coniin". Berichte der Deutschen Chemischen Gesellschaft. 19 (1): 439–441. doi:10.1002/cber.188601901108. ISSN 0365-9496.
- ^ Ladenburg, A. (1907-06-01). "Noch ein Wort über das Isoconlin. 14. Mitteilung über den asymmetryschen Stickstoff". Berichte der Deutschen Chemischen Gesellschaft. 40 (3): 3734–3736. doi:10.1002/cber.190704003170. ISSN 0365-9496.
- ^ Denić, Marija; Blagojević, Polina; Radulović, Niko (2013). "Synthetic approaches to coniine and other 2-alkyl piperidines". Facta universitatis - series: Physics, Chemistry and Technology. 11 (1): 1–26. doi:10.2298/FUPCT1301001D.
- ^ Diels and Alder, Annalen, 1932, 498, 16.
- ^ G. R. Clemo; G. R. Ramage (1932). "Octahydropyrrocoline". Journal of the Chemical Society: 2969–2973. doi:10.1039/JR9320002969.
- ^ Ber., 1934, 67, 1011.
- ^ Talapatra, Sunil Kumar; Talapatra, Bani (2015), Talapatra, Sunil Kumar; Talapatra, Bani (eds.), "Coniine, Conhydrine, and Pseudoconhydrine (The C-Skeleton Derived from a C8-Fatty Acid and N from Transamination)", Chemistry of Plant Natural Products: Stereochemistry, Conformation, Synthesis, Biology, and Medicine, Springer, pp. 733–748, doi:10.1007/978-3-642-45410-3_17, ISBN 978-3-642-45410-3
- ^ Ber., 1917, 50, 1192, 1386.
- ^ Pelletierine is now known to be 1-(2-piperidinyl)-2-propanone;[according to whom?] see: The Merck Index, 15th Ed. (2013), p. 1314,[original research?] Monograph 7181, O'Neil: The Royal Society of Chemistry. Available online at: http://www.rsc.org/Merck-Index/monograph/mono1500007181
- ^ Ber., 1932, 65, 927.
- ^ George William Clough (1918). "The relationship between the optical rotatory powers and the relative configurations of optically active compounds. The influence of certain inorganic haloids on the optical rotatory powers of α-hydroxy-acids, α-amino-acids, and their derivatives". Journal of the Chemical Society, Transactions. 113: 526–554. doi:10.1039/CT9181300526.
- ^ Hande, Sudhir M.; Kawai, Nobuyuki; Uenishi, Jun’ichi (2009-01-02). "An Efficient Synthesis of 2- and 2,6-Substituted Piperidines Using PdII-Catalyzed 1,3-Chirality Transfer Reaction". The Journal of Organic Chemistry. 74 (1): 244–253. doi:10.1021/jo801926g. ISSN 0022-3263. PMID 19012434.
- ^ Leete, Edward. (1964). "Biosynthesis of the Hemlock Alkaloids. The Incorporation of Acetate-1-C14 into Coniine and Conhydrine". Journal of the American Chemical Society. 86 (12): 2509–2513. doi:10.1021/ja01066a039.
- ^ Hotti, Hannu; Seppänen-Laakso, Tuulikki; Arvas, Mikko; Teeri, Teemu H.; Rischer, Heiko (2015). "Polyketide synthases from poison hemlock ( Conium maculatum L.)". The FEBS Journal. 282 (21): 4141–4156. doi:10.1111/febs.13410. PMID 26260860.
- ^ Roberts, Margaret F. (1978). "Separation of the formation of γ-coniceine and aliphatic amines from got activity in Conium maculatum". Phytochemistry. 17 (1): 107. Bibcode:1978PChem..17..107R. doi:10.1016/S0031-9422(00)89689-2.
Further reading
[edit]- Green, Benedict T.; Lee, Stephen T.; Panter, Kip E.; Brown, David R. (2012). "Piperidine Alkaloids: Human and Food Animal Teratogens" (PDF). Food and Chemical Toxicology. 50 (6): 2049–2055. doi:10.1016/j.fct.2012.03.049. PMID 22449544. Retrieved January 23, 2017.