Polyacrylic acid
| |
| Names | |
|---|---|
| IUPAC name
Poly(acrylic acid), poly(1-carboxyethylene)
| |
| Other names
PAA, PAAc, Acrysol, Acumer, Alcosperse, Aquatreat, Carbomer, Sokalan
| |
| Identifiers | |
| ChEBI | |
| ChemSpider |
|
| ECHA InfoCard | 100.115.375 |
| EC Number |
|
| KEGG | |
| UNII |
|
CompTox Dashboard (EPA)
|
|
| Properties | |
| (C3H4O2)n | |
| Molar mass | variable |
| log P | 0.25700[1] |
| Hazards[2] | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| NFPA 704 (fire diamond) | |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
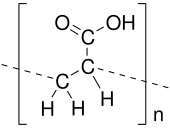
Poly(acrylic acid) (PAA; trade name Carbomer) is a polymer with the formula (CH2−CHCO2H)n. It is a derivative of acrylic acid (CH2=CHCO2H). In addition to the homopolymers, a variety of copolymers and crosslinked polymers, and partially deprotonated derivatives thereof, are known and of commercial value. In a water solution at neutral pH, PAA is an anionic polymer, i.e., many of the side chains of PAA lose their protons and acquire a negative charge. Partially or wholly deprotonated PAAs are polyelectrolytes, with the ability to absorb and retain water and swell to many times their original volume. These properties – acid–base and water-attracting – are the bases of many applications.
Synthesis
[edit]PAA, like any acrylate polymer, is usually synthesized through a process known as free radical polymerization,[3][4] though graft polymerization may also be used.[5][6] Free radical polymerization involves the conversion of monomers, in this case, acrylic acid (CH2=CHCO2H), into a polymer chain through the action of free radicals.[7][8][9] The process typically follows these steps:[10]
- Initiation: Free radicals are generated by initiators such as potassium persulfate (K2S2O8) or Azobisisobutyronitrile (AIBN). These radicals are highly reactive and can start the polymerization process by reacting with the monomer units.[11]
- Propagation: Once the radical reacts with a monomer, it creates a new radical at the end of the growing chain. This new radical can react with additional monomer units, allowing the chain to grow.[12]
- Termination: The reaction continues until two radicals recombine, or a radical is transferred to another molecule, terminating the growth of the polymer chain.[13]
- Chain transfer and inhibition: Other reactions can also occur, such as chain transfer (where the radical is transferred to a different molecule, creating a new radical) or inhibition (where impurities stop the growth of the chain).[14]
Production
[edit]The global market is estimated to be worth $3.4 billion in 2022.[15][16]
Structure and derivatives
[edit]Polyacrylic acid is a weak anionic polyelectrolyte, whose degree of ionisation is dependent on solution pH. In its non-ionised form at low pHs, PAA may associate with various non-ionic polymers (such as polyethylene oxide, poly-N-vinyl pyrrolidone, polyacrylamide, and some cellulose ethers) and form hydrogen-bonded interpolymer complexes.[17] In aqueous solutions PAA can also form polycomplexes with oppositely charged polymers such as chitosan, surfactants, and drug molecules (for example, streptomycin).[18]
Physical properties
[edit]Dry PAAs are sold as white, fluffy powders.
Derivatives
[edit]In the dry powder form of sodium polyacrylate, the positively charged sodium ions are bound to the polyacrylate, however, in aqueous solutions the sodium ions can dissociate. The presence of sodium cations allows the polymer to absorb a high amount of water.
Applications
[edit]Absorbent
[edit]PAA is widely used in dispersants. Its molecular weight has a significant impact on the rheological properties and dispersion capacity, and hence applications. The dominant application for PAA is as a superabsorbent. About 25% of PAA is used for detergents and dispersants.
Polyacrylic acid and its derivatives (particularly sodium polyacrylate) are used in disposable diapers. Acrylic acid is also the main component of Superabsorbent Polymers (SAPs), which are cross-linked polyacrylates that can absorb and retain more than 100 times of their own weight in liquid. The US Food and Drug Administration authorised the use of SAPs in packaging with indirect food contact.[19][20]
Cleaning
[edit]Detergents often contain copolymers of acrylic acid that assist in sequestering dirt. Cross-linked polyacrylic acid has also been used in the production of household products, including floor cleaners. PAA may inactivate the antiseptic chlorhexidine gluconate.[21]
Biocompatible materials
[edit]The neutralized polyacrylic acid gels are suitable biocompatible matrices for medical applications such as gels for skin care products. PAA films can be deposited on orthopaedic implants to protect them from corrosion. Crosslinked hydrogels of PAA and gelatin have also been used as medical glue.
Paints and cosmetics
[edit]Other applications involve paints and cosmetics. They stabilize suspended solid in liquids,[22] prevent emulsions from separating, and control the consistency in flow of cosmetics. Carbomer codes (910, 934, 940, 941, and 934P) are an indication of molecular weight and the specific components of the polymer. For many applications PAAs are used in form of alkali metal or ammonium salts, e.g. sodium polyacrylate.
Emerging applications
[edit]Hydrogels derived from PAA have attracted much study for use as bandages and aids for wound healing.[23]
Drilling fluid and metal quenching
[edit]A few reports were made on PAA use as deflocculant (so called alkaline polyacrylates) for oil drilling industry.[24][25]
It was also reported to be used for metal quenching in metalworking (see Sodium polyacrylate).[26]
References
[edit]- ^ "Polyacrylic acid_msds". Archived from the original on 2022-02-21. Retrieved 2018-04-23.
- ^ "C&L Inventory". echa.europa.eu. Archived from the original on 2021-04-05. Retrieved 2021-12-05.
- ^ Zhang, Rongguo; Li, Qiong; Zhang, Anfu; Liu, Yong; Lei, Jiaheng (2008-12-01). "The synthesis technique of polyacrylic acid superplasticizer". Journal of Wuhan University of Technology-Mater. Sci. Ed. 23 (6): 830–833. doi:10.1007/s11595-008-6830-y. ISSN 1993-0437. S2CID 93458743.
- ^ Hu, Yong; Jiang, Xiqun; Ding, Yin; Ge, Haixiong; Yuan, Yuyan; Yang, Changzheng (2002-08-01). "Synthesis and characterization of chitosan–poly(acrylic acid) nanoparticles". Biomaterials. 23 (15): 3193–3201. doi:10.1016/S0142-9612(02)00071-6. ISSN 0142-9612. PMID 12102191.
- ^ Li, An; Wang, Aiqin; Chen, Jianmin (2004-05-05). "Studies on poly(acrylic acid)/attapulgite superabsorbent composite. I. Synthesis and characterization". Journal of Applied Polymer Science. 92 (3): 1596–1603. doi:10.1002/app.20104. ISSN 0021-8995.
- ^ Lin, Jianming; Wu, Jihuai; Yang, Zhengfang; Pu, Minli (2001-03-01). "Synthesis and Properties of Poly(acrylic acid)/Mica Superabsorbent Nanocomposite". Macromolecular Rapid Communications. 22 (6): 422–424. doi:10.1002/1521-3927(20010301)22:6<422::AID-MARC422>3.0.CO;2-R. ISSN 1022-1336.
- ^ Bromberg, Lev E.; Barr, David P. (1999-06-01). "Aggregation Phenomena in Aqueous Solutions of Hydrophobically Modified Polyelectrolytes. A Probe Solubilization Study". Macromolecules. 32 (11): 3649–3657. Bibcode:1999MaMol..32.3649B. doi:10.1021/ma981946k. ISSN 0024-9297.
- ^ Laschewsky, André; Herfurth, Christoph; Miasnikova, Anna; Stahlhut, Frank; Weiss, Jan; Wieland, Christoph; Wischerhoff, Erik; Gradzielski, Michael; Malo de Molina, Paula (January 2003), Patil, Anjali; Ferritto, Michael S. (eds.), "Stars and Blocks: Tailoring Polymeric Rheology Modifiers for Aqueous Media by Controlled Free Radical Polymerization", ACS Symposium Series, vol. 1148, Washington, DC: American Chemical Society, pp. 125–143, doi:10.1021/bk-2013-1148.ch008, ISBN 978-0-8412-2905-1, retrieved 2023-12-20
- ^ Iqbal, Hafiz M. N.; Rasheed, Tahir; Bilal, Muhammad (2018-10-03), Ahmed, Shakeel; Hussain, Chaudhery Mustansar (eds.), "Design and Processing Aspects of Polymer and Composite Materials", Green and Sustainable Advanced Materials (1 ed.), Wiley, pp. 155–189, doi:10.1002/9781119407089.ch7, ISBN 978-1-119-40708-9, S2CID 139704996, retrieved 2023-12-20
- ^ Liu, Ke; Corrigan, Nathaniel; Postma, Almar; Moad, Graeme; Boyer, Cyrille (2020-10-27). "A Comprehensive Platform for the Design and Synthesis of Polymer Molecular Weight Distributions". Macromolecules. 53 (20): 8867–8882. Bibcode:2020MaMol..53.8867L. doi:10.1021/acs.macromol.0c01954. ISSN 0024-9297. S2CID 225117544.
- ^ Plamper, Felix A.; Becker, Harald; Lanzendörfer, Michael; Patel, Mushtaq; Wittemann, Alexander; Ballauff, Matthias; Müller, Axel H. E. (2005-09-23). "Synthesis, Characterization and Behavior in Aqueous Solution of Star-Shaped Poly(acrylic acid)". Macromolecular Chemistry and Physics. 206 (18): 1813–1825. doi:10.1002/macp.200500238. ISSN 1022-1352.
- ^ Cuti, Sergio S.; Henton, David E.; Powell, Cynthia; Reim, Robert E.; Smith, Patrick; Staples, Thomas L. (1997-04-18). "The effects of MEHQ on the polymerization of acrylic acid in the preparation of superabsorbent gels". Journal of Applied Polymer Science. 64 (3): 577–589. doi:10.1002/(SICI)1097-4628(19970418)64:3<577::AID-APP14>3.0.CO;2-V. ISSN 0021-8995.
- ^ Luciani, Carla; Choi, Kyu Yong (September 2023). "Modelling of a continuous kneader reactor for the polymerization of partially neutralized acrylic acid". The Canadian Journal of Chemical Engineering. 101 (9): 5151–5161. doi:10.1002/cjce.24898. ISSN 0008-4034. S2CID 257729740.
- ^ Nicolaÿ, Renaud; Mosnáček, Jaroslav; Kar, Kishore K.; Fruchey, Stanley O.; Cloeter, Michael D.; Harner, Richard S.; Matyjaszewski, Krzysztof (2012-03-28). "Efficient Polymerization Inhibition Systems for Acrylic Acid Distillation: Vapor-Phase Inhibitors". Industrial & Engineering Chemistry Research. 51 (12): 4467–4471. doi:10.1021/ie201709y. ISSN 0888-5885.
- ^ "Polyacrylic Acid Market Demand, Key Players, Opportunities, & Forecast Analysis By 2029". www.databridgemarketresearch.com. Retrieved 2023-12-20.
- ^ ltd, Research and Markets. "Polyacrylic Acid - Global Strategic Business Report". www.researchandmarkets.com. Retrieved 2023-12-20.
- ^ Khutoryanskiy, Vitaliy V.; Staikos, Georgios (9 March 2009). Hydrogen-bonded Interpolymer Complexes: Formation, Structure And Applications. World Scientific. ISBN 978-981-4475-04-4. OCLC 1200871469. Archived from the original on 2022-02-21. Retrieved 2022-02-21.
- ^ Nurkeeva, Zauresh S; Khutoryanskiy, Vitaliy V; Mun, Grigoriy A; Sherbakova, Marina V; Ivaschenko, Anatoliy T; Aitkhozhina, Nazira A (March 2004). "Polycomplexes of poly(acrylic acid) with streptomycin sulfate and their antibacterial activity". European Journal of Pharmaceutics and Biopharmaceutics. 57 (2): 245–9. doi:10.1016/S0939-6411(03)00149-8. PMID 15018981. Archived from the original on 21 February 2022 – via PubMed, Elsevier.
- ^ Orwoll, Robert A.; Yong, Chong S. (1999). "Poly(acrylic acid)". In Mark, James E. (ed.). Polymer Data Handbook. Oxford University Press, Inc. pp. 252–253. ISBN 978-0195107890. OCLC 39962426. Archived from the original on 2022-02-21. Retrieved 2022-02-21.
- ^ "Acrylates". The Macrogalleria. Polymer Science Learning Center. 2005. Archived from the original on 21 February 2022. Retrieved 25 June 2015.
- ^ Kaiser, Nancy; Klein, Dan; Karanja, Peter; Greten, Zachariah; Newman, Jerry (2009). "Inactivation of chlorhexidine gluconate on skin by incompatible alcohol hand sanitizing gels". American Journal of Infection Control. 37 (7): 569–73. doi:10.1016/j.ajic.2008.12.008. PMID 19398245. Archived from the original on 19 December 2021 – via PubMed, Elsevier.
- ^ Kuila, Debasish; Blay, George A.; Borjas, Ricardo E.; Hughes, Steve; Maddox, Phil; Rice, Kay; Stansbury, Wayne; Laurel, Norma (1999-08-15). "Polyacrylic acid (poly-A) as a chelant and dispersant". Journal of Applied Polymer Science. 73 (7): 1097–1115. doi:10.1002/(SICI)1097-4628(19990815)73:7<1097::AID-APP2>3.0.CO;2-F. ISSN 0021-8995.
- ^ Mogoşanu, George Dan; Grumezescu, Alexandru Mihai (25 March 2014). "Natural and synthetic polymers for wounds and burns dressing". International Journal of Pharmaceutics. 463 (2): 127–136. doi:10.1016/j.ijpharm.2013.12.015. PMID 24368109. Archived from the original on 1 February 2022.
- ^ "Deflocculants: A Detailed Overview". Archived from the original on February 26, 2021.
- ^ Petrov, N.A.; Maikobi, A.A. (December 2017). "Investigation of Uniflox Reagent for Drilling Siberian Solvent Solutions". Oil and Gas Business (6): 6–19. doi:10.17122/ogbus-2017-6-6-19 (inactive 1 November 2024). Archived from the original on 2018-06-21. Retrieved 2022-05-06.
{{cite journal}}: CS1 maint: DOI inactive as of November 2024 (link) - ^ Griffiths, W. D. (1989). The quenching characteristics of sodium polyacrylate solutions (doctoral thesis). Sheffield: Sheffield Hallam University.



