Mutation: Difference between revisions
deleterious are usualy bad, but it does not mean a priori 'les favorable', + good recent (2007) review |
No edit summary |
||
| Line 3: | Line 3: | ||
{{evolution3}} |
{{evolution3}} |
||
[[Image:Portulaca grandiflora mutant1.jpg|thumb|right|A mutation has caused this garden [[moss rose]] to produce flowers of different colors. This is a somatic mutation that may also be passed on in the germ line.]] |
[[Image:Portulaca grandiflora mutant1.jpg|thumb|right|A mutation has caused this garden [[moss rose]] to produce flowers of different colors. This is a somatic mutation that may also be passed on in the germ line.]] |
||
In biology, '''mutations''' are changes to the [[nucleotide]] sequence of the [[genetic material]] of an organism. |
In biology, '''mutations''' are randomly derived changes to the [[nucleotide]] sequence of the [[genetic material]] of an organism. |
||
Mutations can be caused by copying errors or in the genetic material during [[cell division]], by exposure to [[mutagen]]s ([[ultraviolet]] or [[ionizing radiation]], [[mutagens|mutagenic]] chemicals, or [[virus (biology)|viruses]]), or can be induced by the organism itself, by [[cellular processes]] such as [[somatic hypermutation|hypermutation]]. In [[multicellular organisms]] with dedicated [[reproductive cells]], mutations can be subdivided into '''[[germline mutation|germ line mutation]]s''', which only can be passed on to descendants through the reproductive cells, and '''somatic mutations''', which involve cells outside the dedicated reproductive group and which are not usually transmitted to descendants. If the organism can reproduce asexually through mechanisms such as [[cutting (plant)|cuttings]] or [[budding]] the distinction can become blurred. |
Mutations can be caused by copying errors or in the genetic material during [[cell division]], by exposure to [[mutagen]]s ([[ultraviolet]] or [[ionizing radiation]], [[mutagens|mutagenic]] chemicals, or [[virus (biology)|viruses]]), or can be induced by the organism itself, by [[cellular processes]] such as [[somatic hypermutation|hypermutation]]. In [[multicellular organisms]] with dedicated [[reproductive cells]], mutations can be subdivided into '''[[germline mutation|germ line mutation]]s''', which only can be passed on to descendants through the reproductive cells, and '''somatic mutations''', which involve cells outside the dedicated reproductive group and which are not usually transmitted to descendants. If the organism can reproduce asexually through mechanisms such as [[cutting (plant)|cuttings]] or [[budding]] the distinction can become blurred. |
||
Revision as of 10:04, 14 September 2009
| Part of the Biology series on |
| Evolution |
|---|
 |
| Mechanisms and processes |
| Research and history |
| Evolutionary biology fields |

In biology, mutations are randomly derived changes to the nucleotide sequence of the genetic material of an organism.
Mutations can be caused by copying errors or in the genetic material during cell division, by exposure to mutagens (ultraviolet or ionizing radiation, mutagenic chemicals, or viruses), or can be induced by the organism itself, by cellular processes such as hypermutation. In multicellular organisms with dedicated reproductive cells, mutations can be subdivided into germ line mutations, which only can be passed on to descendants through the reproductive cells, and somatic mutations, which involve cells outside the dedicated reproductive group and which are not usually transmitted to descendants. If the organism can reproduce asexually through mechanisms such as cuttings or budding the distinction can become blurred.
For example, plants can sometimes transmit somatic mutations to their descendants asexually or sexually where flower buds develop in somatically mutated parts of plants. A new mutation that was not inherited from either parent is called a de novo mutation. The source of the mutation is unrelated to the consequence, although the consequences are related to which cells were mutated.
Nonlethal mutations accumulate within the gene pool increasing genetic variation[1]. Some genetic changes can be reduced in frequency in the gene pool by natural selection, while other "more favorable" mutations may accumulate and result in adaptive evolutionary changes.
For example, a butterfly may produce offspring with new mutations. The majority of these mutations will have no effect; but one might change the color of one of the butterfly's offspring, making it harder (or easier) for predators to see. If this color change is advantageous, the chance of this butterfly surviving and producing its own offspring are a little better, and over time the number of butterflies with this mutation may form a larger percentage of the population.
Neutral mutations are defined as mutations whose effects do not influence the fitness of an individual. These can accumulate over time due to genetic drift. It is believed that the overwhelming majority of mutations have no significant effect on an organism's fitness. Also, DNA repair mechanisms are able to mend most changes before they become permanent mutations, and many organisms have mechanisms for eliminating otherwise permanently mutated somatic cells.
Mutation is generally accepted by the scientific community as the mechanism upon which natural selection acts, providing the advantageous new traits that survive and multiply in offspring or disadvantageous traits that die out with weaker organisms.
Classification of mutation types

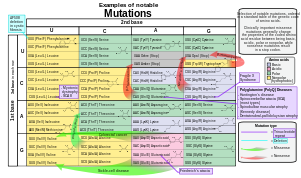
By effect on structure
The sequence of a gene can be altered in a number of ways. Gene mutations have varying effects on health depending on where they occur and whether they alter the function of essential proteins. Mutations in the structure of genes can be classified as:
- Small-scale mutations, such as those affecting a small gene in one or a few nucleotides, including:
- Point mutations, often caused by chemicals or malfunction of DNA replication, exchange a single nucleotide for another[3]. These changes are classified as transitions or transversions[4]. Most common is the transition that exchanges a purine for a purine (A ↔ G) or a pyrimidine for a pyrimidine, (C ↔ T). A transition can be caused by nitrous acid, base mis-pairing, or mutagenic base analogs such as 5-bromo-2-deoxyuridine (BrdU). Less common is a transversion, which exchanges a purine for a pyrimidine or a pyrimidine for a purine (C/T ↔ A/G). An example of a transversion is adenine (A) being converted into a cytosine (C). A point mutation can be reversed by another point mutation, in which the nucleotide is changed back to its original state (true reversion) or by second-site reversion (a complementary mutation elsewhere that results in regained gene functionality). Point mutations that occur within the protein coding region of a gene may be classified into three kinds, depending upon what the erroneous codon codes for:
- Silent mutations: which code for the same amino acid.
- Missense mutations: which code for a different amino acid.
- Nonsense mutations: which code for a stop and can truncate the protein.
- Insertions add one or more extra nucleotides into the DNA. They are usually caused by transposable elements, or errors during replication of repeating elements (e.g. AT repeats). Insertions in the coding region of a gene may alter splicing of the mRNA (splice site mutation), or cause a shift in the reading frame (frameshift), both of which can significantly alter the gene product. Insertions can be reverted by excision of the transposable element.
- Deletions remove one or more nucleotides from the DNA. Like insertions, these mutations can alter the reading frame of the gene. They are generally irreversible: though exactly the same sequence might theoretically be restored by an insertion, transposable elements able to revert a very short deletion (say 1–2 bases) in any location are either highly unlikely to exist or do not exist at all. Note that a deletion is not the exact opposite of an insertion: the former is quite random while the latter consists of a specific sequence inserting at locations that are not entirely random or even quite narrowly defined.
- Point mutations, often caused by chemicals or malfunction of DNA replication, exchange a single nucleotide for another[3]. These changes are classified as transitions or transversions[4]. Most common is the transition that exchanges a purine for a purine (A ↔ G) or a pyrimidine for a pyrimidine, (C ↔ T). A transition can be caused by nitrous acid, base mis-pairing, or mutagenic base analogs such as 5-bromo-2-deoxyuridine (BrdU). Less common is a transversion, which exchanges a purine for a pyrimidine or a pyrimidine for a purine (C/T ↔ A/G). An example of a transversion is adenine (A) being converted into a cytosine (C). A point mutation can be reversed by another point mutation, in which the nucleotide is changed back to its original state (true reversion) or by second-site reversion (a complementary mutation elsewhere that results in regained gene functionality). Point mutations that occur within the protein coding region of a gene may be classified into three kinds, depending upon what the erroneous codon codes for:
- Large-scale mutations in chromosomal structure, including:
- Amplifications (or gene duplications) leading to multiple copies of all chromosomal regions, increasing the dosage of the genes located within them.
- Deletions of large chromosomal regions, leading to loss of the genes within those regions.
- Mutations whose effect is to juxtapose previously separate pieces of DNA, potentially bringing together separate genes to form functionally distinct fusion genes (e.g. bcr-abl). These include:
- Chromosomal translocations: interchange of genetic parts from nonhomologous chromosomes.
- Interstitial deletions: an intra-chromosomal deletion that removes a segment of DNA from a single chromosome, thereby apposing previously distant genes. For example, cells isolated from a human astrocytoma, a type of brain tumor, were found to have a chromosomal deletion removing sequences between the "fused in glioblastoma" (fig) gene and the receptor tyrosine kinase "ros", producing a fusion protein (FIG-ROS). The abnormal FIG-ROS fusion protein has constitutively active kinase activity that causes oncogenic transformation (a transformation from normal cells to cancer cells).
- Chromosomal inversions: reversing the orientation of a chromosomal segment.
- Loss of heterozygosity: loss of one allele, either by a deletion or recombination event, in an organism that previously had two different alleles.
By effect on function
- Loss-of-function mutations are the result of gene product having less or no function. When the allele has a complete loss of function (null allele) it is often called an amorphic mutation. Phenotypes associated with such mutations are most often recessive. Exceptions are when the organism is haploid, or when the reduced dosage of a normal gene product is not enough for a normal phenotype (this is called haploinsufficiency).
- Gain-of-function mutations change the gene product such that it gains a new and abnormal function. These mutations usually have dominant phenotypes. Often called a neomorphic mutation.
- Dominant negative mutations (also called antimorphic mutations) have an altered gene product that acts antagonistically to the wild-type allele. These mutations usually result in an altered molecular function (often inactive) and are characterised by a dominant or semi-dominant phenotype. In humans, Marfan syndrome is an example of a dominant negative mutation occurring in an autosomal dominant disease. In this condition, the defective glycoprotein product of the fibrillin gene (FBN1) antagonizes the product of the normal allele.
- Lethal mutations are mutations that lead to the death of the organisms which carry the mutations.
- A back mutation or reversion is a point mutation that restores the original sequence and hence the original phenotype.[5]
By effect on fitness
In applied genetics it is usual to speak of mutations as either harmful or beneficial.
- A harmful mutation is a mutation that decreases the fitness of the organism.
- A beneficial mutation is a mutation that increases fitness of the organism, or which promotes traits that are desirable.
In theoretical population genetics, it is more usual to speak of such mutations as deleterious or advantageous. In the neutral theory of molecular evolution, genetic drift is the basis for most variation at the molecular level.
- A neutral mutation has no harmful or beneficial effect on the organism. Such mutations occur at a steady rate, forming the basis for the molecular clock.
- A deleterious mutation has a negative effect on the phenotype, and thus decreases the fitness of the organism.
- An advantageous mutation has a positive effect on the phenotype, and thus increases the fitness of the organism.
- A nearly neutral mutation is a mutation that may be slightly deleterious or advantageous, although most nearly neutral mutations are slightly deleterious.
By inheritance
- inheritable generic in pro-generic tissue or cells on path to be changed to gametes.
- non inheritable somatic (eg, carcinogenic mutation)
- non inheritable post mortem aDNA mutation in decaying remains.
By pattern of inheritance
The human genome contains two copies of each gene – a paternal and a maternal allele.
- A heterozygous mutation is a mutation of only one allele.
- A homozygous mutation is an identical mutation of both the paternal and maternal alleles.
- Compound heterozygous mutations or a genetic compound comprises two different mutations in the paternal and maternal alleles. [6]
- A wildtype or homozygous non-mutated organism is one in which neither allele is mutated. (Just not a mutation)
By impact on protein sequence
- A frameshift mutation is a mutation caused by insertion or deletion of a number of nucleotides that is not evenly divisible by three from a DNA sequence. Due to the triplet nature of gene expression by codons, the insertion or deletion can disrupt the reading frame, or the grouping of the codons, resulting in a completely different translation from the original. The earlier in the sequence the deletion or insertion occurs, the more altered the protein produced is.
- Missense mutations or nonsynonymous mutations are types of point mutations where a single nucleotide is changed to cause substitution of a different amino acid. This in turn can render the resulting protein nonfunctional. Such mutations are responsible for diseases such as Epidermolysis bullosa, sickle-cell disease, and SOD1 mediated ALS (Boillée 2006, p. 39).
- A neutral mutation is a mutation that occurs in an amino acid codon which results in the use of a different, but chemically similar, amino acid. This is similar to a silent mutation, where a codon mutation may encode the same amino acid (see Wobble Hypothesis); for example, a change from AUU to AUC will still encode leucine, so no discernible change occurs (a silent mutation).
- A nonsense mutation is a point mutation in a sequence of DNA that results in a premature stop codon, or a nonsense codon in the transcribed mRNA, and possibly a truncated, and often nonfunctional protein product.
- Silent mutations are mutations that do not result in a change to the amino acid sequence of a protein. They may occur in a region that does not code for a protein, or they may occur within a codon in a manner that does not alter the final amino acid sequence. The phrase silent mutation is often used interchangeably with the phrase synonymous mutation; however, synonymous mutations are a subcategory of the former, occurring only within exons. The name silent could be a misnomer. For example, a silent mutation in the exon/intron border may lead to alternative splicing by changing the splice site (see Splice site mutation), thereby leading to a changed protein.
Special classes
- Conditional mutation is a mutation that has wild-type (or less severe) phenotype under certain "permissive" environmental conditions and a mutant phenotype under certain "restrictive" conditions. For example, a temperature-sensitive mutation can cause cell death at high temperature (restrictive condition), but might have no deleterious consequences at a lower temperature (permissive condition).
Causes of mutation
Two classes of mutations are spontaneous mutations (molecular decay) and induced mutations caused by mutagens.
Spontaneous mutations on the molecular level include:
- Tautomerism – A base is changed by the repositioning of a hydrogen atom, altering the hydrogen bonding pattern of that base resulting in incorrect base pairing during replication.
- Depurination – Loss of a purine base (A or G) to form an apurinic site (AP site).
- Deamination – Hydrolysis changes a normal base to an atypical base containing a keto group in place of the original amine group. Examples include C → U and A → HX (hypoxanthine), which can be corrected by DNA repair mechanisms; and 5MeC (5-methylcytosine) → T, which is less likely to be detected as a mutation because thymine is a normal DNA base.
- Transition – A purine changes to another purine, or a pyrimidine to a pyrimidine.
- Transversion – A purine becomes a pyrimidine, or vice versa.
Induced mutations on the molecular level can be caused by:
- Chemicals
- Hydroxylamine NH2OH
- Base analogs (e.g. BrdU)
- Alkylating agents (e.g. N-ethyl-N-nitrosourea) These agents can mutate both replicating and non-replicating DNA. In contrast, a base analog can only mutate the DNA when the analog is incorporated in replicating the DNA. Each of these classes of chemical mutagens has certain effects that then lead to transitions, transversions, or deletions.
- Agents that form DNA adducts (e.g. ochratoxin A metabolites)[7]
- DNA intercalating agents (e.g. ethidium bromide)
- DNA crosslinkers
- Oxidative damage
- Nitrous acid converts amine groups on A and C to diazo groups, altering their hydrogen bonding patterns which leads to incorrect base pairing during replication.
- Radiation
- Ultraviolet radiation (nonionizing radiation). Two nucleotide bases in DNA – cytosine and thymine – are most vulnerable to radiation that can change their properties. UV light can induce adjacent thymine bases in a DNA strand to pair with each other, as a bulky dimer.
- Ionizing radiation
- Viral infections[8]
DNA has so-called hotspots, where mutations occur up to 100 times more frequently than the normal mutation rate. A hotspot can be at an unusual base, e.g., 5-methylcytosine.
Mutation rates also vary across species. Evolutionary biologists have theorized that higher mutation rates are beneficial in some situations, because they allow organisms to evolve and therefore adapt more quickly to their environments. For example, repeated exposure of bacteria to antibiotics, and selection of resistant mutants, can result in the selection of bacteria that have a much higher mutation rate than the original population (mutator strains).
Nomenclature
Nomenclature of mutations specify the type of mutation and base or amino acid changes.
- Nucleotide substitution (e.g. 76A>T) - The number is the position of the nucleotide from the 5' end, the first letter represents the wild type nucleotide, and the second letter represents the nucleotide which replaced the wild type. In the given example, the adenine at the 76th position was replaced by a thymine.
- If it becomes necessary to differentiate between mutations in genomic DNA, mitochondrial DNA, and RNA, a simple convention is used. For example, if the 100th base of a nucleotide sequence mutated from G to C, then it would be written as g.100G>C if the mutation occurred in genomic DNA, m.100G>C if the mutation occurred in mitochondrial DNA, or r.100g>c if the mutation occurred in RNA. Note that for mutations in RNA, the nucleotide code is written in lower case.
- Amino acid substitution (e.g. D111E) – The first letter is the one letter code of the wild type amino acid, the number is the position of the amino acid from the N terminus, and the second letter is the one letter code of the amino acid present in the mutation. Nonsense mutations are represented with an X for the second amino acid (e.g. D111X).
- Amino acid deletion (e.g. ΔF508) – The Greek letter Δ (delta) indicates a deletion. The letter refers to the amino acid present in the wild type and the number is the position from the N terminus of the amino acid were it to be present as in the wild type.
Harmful mutations
Changes in DNA caused by mutation can cause errors in protein sequence, creating partially or completely non-functional proteins. To function correctly, each cell depends on thousands of proteins to function in the right places at the right times. When a mutation alters a protein that plays a critical role in the body, a medical condition can result. A condition caused by mutations in one or more genes is called a genetic disorder. Some mutations alter a gene's DNA base sequence but do not change the function of the protein made by the gene. Studies in the fly Drosophila melanogaster suggest that if a mutation does change a protein, this will probably be harmful, with about 70 percent of these mutations having damaging effects, and the remainder being either neutral or weakly beneficial.[9]
If a mutation is present in a germ cell, it can give rise to offspring that carries the mutation in all of its cells. This is the case in hereditary diseases. On the other hand, a mutation can occur in a somatic cell of an organism. Such mutations will be present in all descendants of this cell, and certain mutations can cause the cell to become malignant, and thus cause cancer[10].
Often, gene mutations that could cause a genetic disorder are repaired by the DNA repair system of the cell. Each cell has a number of pathways through which enzymes recognize and repair mistakes in DNA. Because DNA can be damaged or mutated in many ways, the process of DNA repair is an important way in which the body protects itself from disease.
Beneficial mutations
Although most mutations are deleterious, mutations may have a positive effect given certain selective pressures in a population.
For example, a specific 32 base pair deletion in human CCR5 (CCR5-Δ32) confers HIV resistance to homozygotes and delays AIDS onset in heterozygotes.[11] The CCR5 mutation is more common in those of European descent. One theory for the etiology of the relatively high frequency of CCR5-Δ32 in the European population is that it conferred resistance to the bubonic plague in mid-14th century Europe. People who had this mutation were able to survive infection; thus, its frequency in the population increased.[12] It could also explain why this mutation is not found in Africa where the bubonic plague never reached. Newer theory says the selective pressure on the CCR5 Delta 32 mutation has been caused by smallpox instead of the bubonic plague.[13]
See also
References
- ^ Attention: This template ({{cite doi}}) is deprecated. To cite the publication identified by doi:10.1038/nrg2146, please use {{cite journal}} (if it was published in a bona fide academic journal, otherwise {{cite report}} with
|doi=10.1038/nrg2146instead. - ^ References for the image are found in Wikimedia Commons page at: Commons:File:Notable mutations.svg#References.
- ^ Freese, Ernst (1959). "The Difference between Spontaneous and Base-Analogue Induced Mutations of Phage T4". Proc. Natl. Acad. Sci. U.S.A. 45 (4): 622–33. doi:10.1073/pnas.45.4.622. PMC 222607. PMID 16590424.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Freese, Ernst (1959). "The Specific Mutagenic Effect of Base Analogues on Phage T4". J. Mol. Biol. 1: 87–105. doi:10.1016/S0022-2836(59)80038-3.
- ^ Ellis NA, Ciocci S, German J (2001). "Back mutation can produce phenotype reversion in Bloom syndrome somatic cells". Hum Genet. 108 (2): 167–73. doi:10.1007/s004390000447. PMID 11281456.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Medterms.com
- ^ Pfohl-Leszkowicz A, Manderville RA (2007). "Ochratoxin A: An overview on toxicity and carcinogenicity in animals and humans". Mol Nutr Food Res. 51 (1): 61–99. doi:10.1002/mnfr.200600137. PMID 17195275.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Pilon L, Langelier Y, Royal A (1986). "Herpes simplex virus type 2 mutagenesis: characterization of mutants induced at the hprt locus of nonpermissive XC cells". Mol. Cell. Biol. 6 (8): 2977–83. PMC 367868. PMID 3023954.
{{cite journal}}: Unknown parameter|day=ignored (help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Sawyer SA, Parsch J, Zhang Z, Hartl DL (2007). "Prevalence of positive selection among nearly neutral amino acid replacements in Drosophila". Proc. Natl. Acad. Sci. U.S.A. 104 (16): 6504–10. doi:10.1073/pnas.0701572104. PMID 17409186.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Ionov Y, Peinado MA, Malkhosyan S, Shibata D, Perucho M (1993). "Ubiquitous somatic mutations in simple repeated sequences reveal a new mechanism for colonic carcinogenesis". Nature. 363 (6429): 558–61. doi:10.1038/363558a0. PMID 8505985.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "CCR5 receptor gene and HIV infection, Antonio Pacheco".
- ^ "PBS:Secrets of the Dead. Case File: Mystery of the Black Death".
- ^ Galvani A, Slatkin M (2003). "Evaluating plague and smallpox as historical selective pressures for the CCR5-Δ32 HIV-resistance allele". Proc Natl Acad Sci USA. 100 (25): 15276–9. doi:10.1073/pnas.2435085100. PMC 299980. PMID 14645720.
External links
- "All About Mutations" from the Huntington's Disease Outreach Project for Education at Stanford
- Central Locus Specific Variation Database at the Institute of Genomics and Integrative Biology
- The mutations chapter of the WikiBooks General Biology textbook
- Examples of Beneficial Mutations
- Correcting mutation by gene therapy
- BBC Radio 4 In Our Time - GENETIC MUTATION - with Steve Jones - streaming audio
