Water softening
Water softening is the removal of calcium, magnesium, and certain other metal cations in hard water. The resulting soft water requires less soap for the same cleaning effort, as soap is not wasted bonding with calcium ions. Soft water also extends the lifetime of plumbing by reducing or eliminating scale build-up in pipes and fittings. Water softening is usually achieved using lime softening or ion-exchange resins but is increasingly being accomplished using nanofiltration or reverse osmosis membranes.
Rationale
The presence of certain metal ions like calcium and magnesium principally as bicarbonates, chlorides, and sulfates in water causes a variety of problems.[1]
Hard water leads to the buildup of limescale, which can foul plumbing, and promote galvanic corrosion.[2] In industrial scale water softening plants, the effluent flow from the re-generation process can precipitate scale that can interfere with sewage systems.[3]
The slippery feeling associated with washing in soft water is caused by the weaker attraction of the soap to the water ions when the water has been stripped of its mineral content. The surface of human skin has a light charge that the soap tends to bind with, requiring more effort and a greater volume of water to remove.[4] Hard water contains calcium or magnesium ions that form insoluble salts upon reacting with soap, leaving a coating of insoluble stearates on tub and shower surfaces, commonly called soap scum.[4][5]

Methods
The most common means for removing water hardness rely on ion-exchange resin or reverse osmosis. Other approaches include precipitation methods and sequestration by the addition of chelating agents.
Ion-exchange resin method
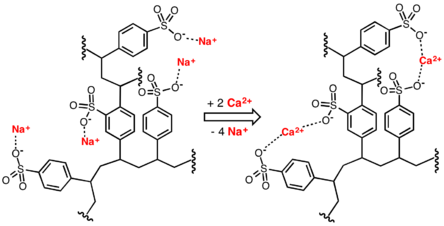
Conventional water-softening appliances intended for household use depend on an ion-exchange resin in which "hardness ions"—mainly Ca2+ and Mg2+—are exchanged for sodium ions.[6] As described by NSF/ANSI Standard 44,[7] ion-exchange devices reduce the hardness by replacing magnesium and calcium (Mg2+ and Ca2+) with sodium or potassium ions (Na+ and K+)."

Ion exchange resins are organic polymers containing anionic functional groups to which the divalent cations (Ca2+) bind more strongly than monovalent cations (Na+). Inorganic materials called zeolites also exhibit ion-exchange properties. These minerals are widely used in laundry detergents. Resins are also available to remove the carbonate, bicarbonate, and sulfate ions that are absorbed and hydroxide ions that are released from the resin.[citation needed]
When all the available Na+ ions have been replaced with calcium or magnesium ions, the resin must be recharged by eluting the Ca2+ and Mg2+ ions using a solution of sodium chloride or sodium hydroxide, depending on the type of resin used.[8] For anionic resins, regeneration typically uses a solution of sodium hydroxide (lye) or potassium hydroxide. The waste waters eluted from the ion-exchange column containing the unwanted calcium and magnesium salts are typically discharged to the sewage system.[3]
Recharge typically takes the following steps:[9]
Backwash
Water is directed through the resin in the opposite direction as normal flow, and the output is sent to a drain for disposal. This 10 minute process flushes out solids, and expands the resin bed.
Brine draw
Water is directed through a jet pump, which pulls salt water from the brine tank, before the water and brine pass through the resin bed in the normal direction. The output of this typical 30 minute process is discarded through the drain hose.
Rinse
Brine draw stops, but water continues to flow from the inlet to the outlet, gradually flushing the brine out of the resin bed. The flushing water flows slowly for several minutes, then at a faster rate for as long as an hour. At some point, the brine reservoir is refilled with fresh water.
Lime softening
Lime softening is the process in which lime is added to hard water to make it softer. It has several advantages over the ion-exchange method but is mainly suited to commercial treatment applications.[10]
Chelating agents
Chelators are used in chemical analysis, as water softeners, and are ingredients in many commercial products such as shampoos and food preservatives. Citric acid is used to soften water in soaps, personal care products and laundry detergents. A commonly used synthetic chelator is ethylenediaminetetraacetic acid (EDTA), which may exist as a tetrasodium or disodium salt. Due to environmental and aquatic toxicity concerns regarding widespread use of EDTA in household and personal care products, alternatives such as sodium phytate/phytic acid, tetrasodium glutamate diacetate and trisodium ethylenediamine disuccinate are finding more prevalent usage.
Washing soda method
In this method, water is treated with a calculated amount of washing soda (Na2CO3), which converts the chlorides and sulphates of calcium and magnesium into their respective carbonates, which get precipitated.
CaCl2 + Na2CO3 --> CaCO3 + 2NaCl
MgSO4 + Na2CO3 --> MgCO3 + Na2SO4
Distillation and rain water
Since Ca2+ and Mg2+ exist as nonvolatile salts, they can be removed by distilling the water. Distillation is too expensive in most cases. Rainwater is soft because it is naturally distilled during the water cycle of evaporation, condensation and precipitation.[11]
Reverse osmosis
Reverse osmosis uses an applied pressure gradient across a semipermeable membrane to overcome osmotic pressure and remove water molecules from the solution with hardness ions. The membrane has pores large enough to admit water molecules for passage; hardness ions such as Ca2+ and Mg2+ will not fit through the pores. The resulting soft water supply is free of hardness ions without any other ions being added. Membranes are a type of water filter requiring regular cleaning or replacement maintenance.
Distillation and reverse osmosis are the most widely used two non-chemical methods of water softening.
Non-chemical devices
Some manufacturers claim that the electrical devices they produce can affect the interaction of minerals with water so that the minerals do not bind to surfaces. Since these systems do not work by exchanging ions, like traditional water softeners do, one benefit claimed for the user is the elimination of the need to add salt to the system. Such systems do not remove minerals from the water itself. Rather, they can only alter the downstream effects that the mineral-bearing water would otherwise have. These systems do not fall within the term "water softening" but rather "water conditioning".
Similar claims for magnetic water treatment are not considered to be valid. For instance, no reduction of scale formation was found when such a magnet device was scientifically tested.[12]
Health effects
The CDC recommends limiting daily total sodium intake to 2,300 mg per day,[13] though the average American consumes 3,500 mg per day.[14] Because the amount of sodium present in drinking water—even after softening—does not represent a significant percentage of a person's daily sodium intake, the EPA considers sodium in drinking water to be unlikely to cause adverse health effects.[15]
For those who are on sodium-restricted diets, the use of a reverse osmosis system for drinking water and cooking water will remove sodium along with any other impurities that may be present. Potassium chloride can also be used as a regenerant instead of sodium chloride, although it is more costly. For people with impaired kidney function, however, elevated potassium levels, or hyperkalemia, can lead to complications such as cardiac arrhythmia.
High levels of water hardness in the home may also be linked to the development of eczema early in life.[16]
Environmental impact
Softened water (measured as residual sodium carbonate index) in which calcium and magnesium have been partly replaced by sodium is not suitable for irrigation use, as it tends to cause the development of alkali soils.[17] Non-chemical devices are often used in place of traditional water softening for this application.
See also
References
- ^ The Editors of Encyclopædia Britannica (20 July 1998). Hard water. Encyclopædia Britannica. ISBN 9781593392925. Retrieved 4 March 2015.
{{cite book}}:|last1=has generic name (help) - ^ Stephen Lower (July 2007). "Hard water and water softening". Retrieved 2007-10-08.
- ^ a b Rowe, Gary (1988). "Well Contamination By Water Softener Regeneration Discharge Water". Journal of Environmental Health. 50 (5): 272–276. JSTOR 44541189.
- ^ a b "Why can't I rinse the soap off my hands?". USGS. Retrieved 7 October 2019.
- ^ "Archived copy". Archived from the original on 2011-08-17. Retrieved 2011-08-16.
{{cite web}}: CS1 maint: archived copy as title (link) - ^ "Water Softeners". Canadian Mortgage and Housing Corporation. Archived from the original on October 10, 2006. Retrieved 2010-01-29.
- ^ Filtration Facts, September 2005, U.S. Environmental Protection Administration, pp. 6-7. Accessed 6 January 2013.
- ^ "Ion Exchange Treatment of Drinking Water" (PDF). Des.nh.gov. 2009. Retrieved 2016-07-23.
- ^ "How to Achieve Optimal Softener Performance". Chem Aqua, Inc. 2020. Retrieved December 21, 2020.
- ^ Ion Exchange vs. Lime Softening, Nancrede Engineering
- ^ Bartram, edited by Jamie; Ballance, Richard (1996). Water quality monitoring : a practical guide to the design and implementation of freshwater quality studies and monitoring programmes (1st ed.). London: E & FN Spon. ISBN 0419223207.
{{cite book}}:|first1=has generic name (help) - ^ Krauter, P. W.; Harrar, J. E.; Orloff, S. P.; Bahowick, S. M. (1 December 1996). "Test of a Magnetic Device for the Amelioration of Scale Formation at Treatment Facility D" (Document). Osti.gov. OSTI 567404.
- ^ "Salt Home — DHDSP". Cdc.gov. Retrieved 2016-07-23.
- ^ Layton, Lyndsey (20 April 2010). "FDA plans to limit amount of salt allowed in processed foods for health reasons". Washingtonpost.com.
- ^ "Drinking Water Contaminant Candidate List (CCL) and Regulatory Determination | US EPA". Water.epa.gov. 2016-05-09. Retrieved 2016-07-23.
- ^ Perkin, Michael (2016-05-18). "Hard water linked to risk of eczema in infants".
- ^ "Managing irrigation water quality" (PDF). Oregon State University. p. 12. Retrieved 2012-10-04.
