Streptococcus pyogenes: Difference between revisions
m Reverted edits by 121.54.58.143 (talk) identified as unconstructive (HG) |
Hannes Röst (talk | contribs) added data about occurence |
||
| Line 20: | Line 20: | ||
Streptococci are [[catalase]]-negative. In ideal conditions, ''S. pyogenes'' has an incubation period of approximately 1–3 days.<ref name=WisconsinDoHS>{{cite | url=http://www.dhs.wisconsin.gov/communicable/FactSheets/StreptococcalPharyngitis.htm }}</ref> It is an infrequent, but usually pathogenic, part of the [[skin flora]]. |
Streptococci are [[catalase]]-negative. In ideal conditions, ''S. pyogenes'' has an incubation period of approximately 1–3 days.<ref name=WisconsinDoHS>{{cite | url=http://www.dhs.wisconsin.gov/communicable/FactSheets/StreptococcalPharyngitis.htm }}</ref> It is an infrequent, but usually pathogenic, part of the [[skin flora]]. |
||
It is estimated that there are more than 700 Million infections each year and over 650.000 cases of severe, invasive infections which have a mortality rate of 25 %.<ref> |
|||
{{cite journal |author=Aziz RK, Kansal R, Aronow BJ, ''et al.'' |title=Microevolution of group A streptococci in vivo: capturing regulatory networks engaged in sociomicrobiology, niche adaptation, and hypervirulence |journal=[[PLoS ONE]] |volume=5 |issue=4 |pages=e9798 |year=2010 |pmid=20418946 |pmc=2854683 |doi=10.1371/journal.pone.0009798 |url=http://dx.plos.org/10.1371/journal.pone.0009798 |accessdate=2011-08-12}} |
|||
</ref> |
|||
== Serotyping == |
== Serotyping == |
||
Revision as of 13:15, 12 August 2011
| Streptococcus pyogenes | |
|---|---|
| |
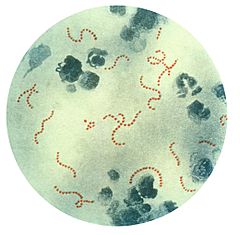
| S. pyogenes bacteria at 900x magnification. | |
| Scientific classification | |
| Kingdom: | |
| Phylum: | |
| Class: | |
| Order: | |
| Family: | |
| Genus: | |
| Species: | S. pyogenes
|
| Binomial name | |
| Streptococcus pyogenes Rosenbach 1884
| |
Streptococcus pyogenes is a spherical, Gram-positive bacterium that is the cause of Group A streptococcal infections.[1] S. pyogenes displays streptococcal group A antigen on its cell wall. S. pyogenes typically produces large zones of beta-hemolysis (the complete disruption of erythrocytes and the release of hemoglobin) when cultured on blood agar plates, and are therefore also called Group A (beta-hemolytic) Streptococcus (abbreviated GABHS).
Streptococci are catalase-negative. In ideal conditions, S. pyogenes has an incubation period of approximately 1–3 days.[2] It is an infrequent, but usually pathogenic, part of the skin flora.
It is estimated that there are more than 700 Million infections each year and over 650.000 cases of severe, invasive infections which have a mortality rate of 25 %.[3]
Serotyping
In 1928, Rebecca Lancefield published a method for serotyping S. pyogenes based on its M protein, a virulence factor displayed on its surface.[4] Later in 1946, Lancefield described the serologic classification of S. pyogenes isolates based on their surface T antigen.[5] Four of the 20 T antigens have been revealed to be pili, which are used by bacteria to attach to host cells.[6] Over 100 M serotypes and approximately 20 T serotypes are known.
Pathogenesis
S. pyogenes is the cause of many important human diseases, ranging from mild superficial skin infections to life-threatening systemic diseases.[1] Infections typically begin in the throat or skin. Examples of mild S. pyogenes infections include pharyngitis ("strep throat") and localized skin infection ("impetigo"). Erysipelas and cellulitis are characterized by multiplication and lateral spread of S. pyogenes in deep layers of the skin. S. pyogenes invasion and multiplication in the fascia can lead to necrotizing fasciitis, a potentially life-threatening condition requiring surgical treatment.
Infections due to certain strains of S. pyogenes can be associated with the release of bacterial toxins. Throat infections associated with release of certain toxins lead to scarlet fever. Other toxigenic S. pyogenes infections may lead to streptococcal toxic shock syndrome, which can be life-threatening.[1]
S. pyogenes can also cause disease in the form of postinfectious "nonpyogenic" (not associated with local bacterial multiplication and pus formation) syndromes. These autoimmune-mediated complications follow a small percentage of infections and include rheumatic fever and acute postinfectious glomerulonephritis. Both conditions appear several weeks following the initial streptococcal infection. Rheumatic fever is characterised by inflammation of the joints and/or heart following an episode of streptococcal pharyngitis. Acute glomerulonephritis, inflammation of the renal glomerulus, can follow streptococcal pharyngitis or skin infection.
This bacterium remains acutely sensitive to penicillin. Failure of treatment with penicillin is generally attributed to other local commensal organisms producing β-lactamase, or failure to achieve adequate tissue levels in the pharynx. Certain strains have developed resistance to macrolides, tetracyclines and clindamycin.
Virulence factors
S. pyogenes has several virulence factors that enable it to attach to host tissues, evade the immune response, and spread by penetrating host tissue layers.[7] A carbohydrate-based bacterial capsule composed of hyaluronic acid surrounds the bacterium, protecting it from phagocytosis by neutrophils.[1] In addition, the capsule and several factors embedded in the cell wall, including M protein, lipoteichoic acid, and protein F (SfbI) facilitate attachment to various host cells.[8] M protein also inhibits opsonization by the alternative complement pathway by binding to host complement regulators. The M protein found on some serotypes is also able to prevent opsonization by binding to fibrinogen.[1] However, the M protein is also the weakest point in this pathogen's defense, as antibodies produced by the immune system against M protein target the bacteria for engulfment by phagocytes. M proteins are unique to each strain, and identification can be used clinically to confirm the strain causing an infection.
S. pyogenes releases a number of proteins, including several virulence factors, into its host:[1]
| Name | Description |
|---|---|
| Streptolysin O | An exotoxin that is one of the bases of the organism's beta-hemolytic property. |
| Streptolysin S | A cardiotoxic exotoxin that is another beta-hemolytic component. Streptolysin S is not immunogenic and O2 stable. A potent cell poison affecting many types of cell including neutrophils, platelets, and sub-cellular organelles, streptolysin S causes an immune response and detection of antibodies to it; antistreptolysin O (ASO) can be clinically used to confirm a recent infection. |
| Streptococcal pyogenic exotoxin A (SpeA) | Superantigens secreted by many strains of S. pyogenes. This pyrogenic exotoxin is responsible for the rash of scarlet fever and many of the symptoms of streptococcal toxic shock syndrome. |
| Streptococcal pyogenic exotoxin C (SpeC) | |
| Streptokinase | Enzymatically activates plasminogen, a proteolytic enzyme, into plasmin, which in turn digests fibrin and other proteins. |
| Hyaluronidase | It is widely assumed hyaluronidase facilitates the spread of the bacteria through tissues by breaking down hyaluronic acid, an important component of connective tissue. However, very few isolates of S. pyogenes are capable of secreting active hyaluronidase due to mutations in the gene that encode the enzyme. Moreover, the few isolates that are capable of secreting hyaluronidase do not appear to need it to spread through tissues or to cause skin lesions.[9] Thus, the true role of hyaluronidase in pathogenesis, if any, remains unknown. |
| Streptodornase | Most strains of S. pyogenes secrete up to four different DNases, which are sometimes called streptodornase. The DNases protect the bacteria from being trapped in neutrophil extracellular traps (NETs) by digesting the NET's web of DNA, to which are bound neutrophil serine proteases that can kill the bacteria.[10] |
| C5a peptidase | C5a peptidase cleaves a potent neutrophil chemotaxin called C5a, which is produced by the complement system.[11] C5a peptidase is necessary to minimize the influx of neutrophils early in infection as the bacteria are attempting to colonize the host's tissue.[12] |
| Streptococcal chemokine protease | The affected tissue of patients with severe cases of necrotizing fasciitis are devoid of neutrophils.[13] The serine protease ScpC, which is released by S. pyogenes, is responsible for preventing the migration of neutrophils to the spreading infection.[14] ScpC degrades the chemokine IL-8, which would otherwise attract neutrophils to the site of infection. C5a peptidase, although required to degrade the neutrophil chemotaxin C5a in the early stages of infection, is not required for S. pyogenes to prevent the influx of neutrophils as the bacteria spread through the fascia.[12][14] |
Diagnosis
Usually, a throat swab is taken to the laboratory for testing. A Gram stain is performed to show Gram-positive cocci in chains. Then, the organism is cultured on blood agar with an added bacitracin antibiotic disk to show beta-haemolytic colonies and sensitivity (zone of inhibition around the disk) for the antibiotic. Culture on non-blood containing agar then, perform catalase test, which should show a negative reaction for all Streptococci. S. pyogenes is CAMP (not to be confused with cAMP) and hippurate tests negative. Serological identification of the organism involves testing for the presence of group A specific polysaccharide in the bacterium's cell wall using the Phadebact test.
Treatment
The treatment of choice is penicillin and the duration of treatment is well established as being 10 days minimum.[15] There is no reported instance of penicillin-resistance reported to date, although since 1985, there have been many reports of penicillin-tolerance.[16]
Macrolides, chloramphenicol and tetracyclines may be used if the strain isolated has been shown to be sensitive, but resistance is much more common.
Prevention
No vaccines are currently available to protect against S. pyogenes infection, but specific protective antibody has been shown to persist as long as 45 years after the original infection.[17]
References
- ^ a b c d e f Ryan KJ, Ray CG (editors) (2004). Sherris Medical Microbiology (4th ed.). McGraw Hill. ISBN 0-8385-8529-9.
{{cite book}}:|author=has generic name (help) - ^ http://www.dhs.wisconsin.gov/communicable/FactSheets/StreptococcalPharyngitis.htm
{{citation}}: Missing or empty|title=(help) - ^
Aziz RK, Kansal R, Aronow BJ; et al. (2010). "Microevolution of group A streptococci in vivo: capturing regulatory networks engaged in sociomicrobiology, niche adaptation, and hypervirulence". PLoS ONE. 5 (4): e9798. doi:10.1371/journal.pone.0009798. PMC 2854683. PMID 20418946. Retrieved 2011-08-12.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) CS1 maint: unflagged free DOI (link) - ^ Lancefield RC (1928). "The antigenic complex of Streptococcus hemolyticus". J Exp Med. 47 (1): 9–10. doi:10.1084/jem.47.1.91.
- ^ Lancefield RC, Dole VP (1946). "The properties of T antigen extracted from group A hemolytic streptococci". J Exp Med. 84: 449–71. doi:10.1084/jem.84.5.449.
- ^ Mora M, Bensi G, Capo S; et al. (2005). "Group A Streptococcus produce pilus-like structures containing protective antigens and Lancefield T antigens". Proc Natl Acad Sci USA. 102 (43): 15641–6. doi:10.1073/pnas.0507808102. PMC 1253647. PMID 16223875.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Patterson MJ (1996). Streptococcus. In: Baron's Medical Microbiology (Baron S et al., eds.) (4th ed.). Univ of Texas Medical Branch. ISBN 0-9631172-1-1.
- ^ Bisno AL, Brito MO, Collins CM (2003). "Molecular basis of group A streptococcal virulence". Lancet Infect Dis. 3 (4): 191–200. doi:10.1016/S1473-3099(03)00576-0. PMID 12679262.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Starr C, Engleberg N (2006). "Role of hyaluronidase in subcutaneous spread and growth of group A streptococcus". Infect Immun. 74 (1): 40–8. doi:10.1128/IAI.74.1.40-48.2006. PMC 1346594. PMID 16368955.
- ^ Buchanan J, Simpson A, Aziz R, Liu G, Kristian S, Kotb M, Feramisco J, Nizet V (2006). "DNase expression allows the pathogen group A Streptococcus to escape killing in neutrophil extracellular traps". Curr Biol. 16 (4): 396–400. doi:10.1016/j.cub.2005.12.039. PMID 16488874.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Wexler D, Chenoweth D, Cleary P (1985). "Mechanism of action of the group A streptococcal C5a inactivator". Proc Natl Acad Sci USA. 82 (23): 8144–8. doi:10.1073/pnas.82.23.8144. PMC 391459. PMID 3906656.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Ji Y, McLandsborough L, Kondagunta A, Cleary P (1996). "C5a peptidase alters clearance and trafficking of group A streptococci by infected mice". Infect Immun. 64 (2): 503–10. PMC 173793. PMID 8550199.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hidalgo-Grass C, Dan-Goor M, Maly A, Eran Y, Kwinn L, Nizet V, Ravins M, Jaffe J, Peyser A, Moses A, Hanski E (2004). "Effect of a bacterial pheromone peptide on host chemokine degradation in group A streptococcal necrotising soft-tissue infections". Lancet. 363 (9410): 696–703. doi:10.1016/S0140-6736(04)15643-2. PMID 15001327.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Hidalgo-Grass C, Mishalian I, Dan-Goor M, Belotserkovsky I, Eran Y, Nizet V, Peled A, Hanski E (2006). "A streptococcal protease that degrades CXC chemokines and impairs bacterial clearance from infected tissues". EMBO J. 25 (19): 4628–37. doi:10.1038/sj.emboj.7601327. PMC 1589981. PMID 16977314.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Falagas ME, Vouloumanou EK, Matthaiou DK, Kapaskelis AM, Karageorgopoulos DE (2008). "Effectiveness and safety of short-course vs long-course antibiotic therapy for group a beta hemolytic streptococcal tonsillopharyngitis: a meta-analysis of randomized trials". Mayo Clin Proc. 83 (8): 880–9. doi:10.4065/83.8.880. PMID 18674472.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Kim KS, Kaplan EL (1985). "Association of penicillin tolerance with failure to eradicate group A streptococci from patients with pharyngitis". J Pediatr. 107 (5): 681–4. doi:10.1016/S0022-3476(85)80392-9. PMID 3903089.
- ^ Bencivenga JF, Johnson DR, Kaplan EL (2009). "Determination of group a streptococcal anti-M type-specific antibody in sera of rheumatic fever patients after 45 years". Clinical Infectious Diseases. 49 (8): 1237–9. doi:10.1086/605673. PMID 19761409.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link)
