Valopicitabine
Appearance
 | |
| Clinical data | |
|---|---|
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
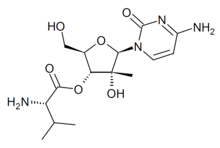
| Formula | C15H24N4O6 |
| Molar mass | 356.379 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Valopicitabine (NM-283) is an antiviral drug which was developed as a treatment for hepatitis C, though only progressed as far as Phase III clinical trials. It acts as an RNA-dependent RNA polymerase inhibitor. It is a prodrug which is converted inside the body to the active form, 2'-C-methylcytidine triphosphate.[1][2][3][4][5]
References
[edit]- ^ Pierra C, Benzaria S, Amador A, Moussa A, Mathieu S, Storer R, Gosselin G (2005). "Nm 283, an efficient prodrug of the potent anti-HCV agent 2'-C-methylcytidine". Nucleosides, Nucleotides & Nucleic Acids. 24 (5–7): 767–70. doi:10.1081/ncn-200060112. PMID 16248033. S2CID 23883085.
- ^ Toniutto P, Fabris C, Bitetto D, Fornasiere E, Rapetti R, Pirisi M (February 2007). "Valopicitabine dihydrochloride:a specific polymerase inhibitor of hepatitis C virus". Current Opinion in Investigational Drugs. 8 (2): 150–8. PMID 17328231.
- ^ Liu-Young G, Kozal MJ (June 2008). "Hepatitis C protease and polymerase inhibitors in development". AIDS Patient Care and STDs. 22 (6): 449–57. doi:10.1089/apc.2007.0199. PMC 2928549. PMID 18479202.
- ^ Carroll SS, Koeplinger K, Vavrek M, Zhang NR, Handt L, MacCoss M, et al. (August 2011). "Antiviral efficacy upon administration of a HepDirect prodrug of 2'-C-methylcytidine to hepatitis C virus-infected chimpanzees". Antimicrobial Agents and Chemotherapy. 55 (8): 3854–60. doi:10.1128/AAC.01152-10. PMC 3147631. PMID 21628542.
- ^ Krecmerova M (2017). "Amino Acid Ester Prodrugs of Nucleoside and Nucleotide Antivirals". Mini Reviews in Medicinal Chemistry. 17 (10): 818–833. doi:10.2174/1389557517666170216151601. PMID 28215138.
