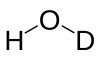
Semiheavy water
| |||
| Names | |||
|---|---|---|---|
| IUPAC name
(O-2H1)Water
| |||
| Other names
Deuterium hydrogen monoxide
Deuterium hydrogen oxide, Water-d1 , Water-d | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChemSpider | |||
| 115 | |||
PubChem CID
|
|||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| H2HO (also HDO) | |||
| Molar mass | 19.0214 g mol−1 | ||
| Appearance | Very pale blue, transparent liquid, very similar to regular water | ||
| Density | 1.054 g cm−3 | ||
| Melting point | 3.81 °C (38.86 °F; 276.96 K) | ||
| Boiling point | 101.42 °C (214.56 °F; 374.57 K) | ||
| miscible | |||
| log P | −0.65 | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Semiheavy water is the result of replacing one of the protium in light water with deuterium.[1] It exists whenever there is water with light hydrogen (protium, 1H) and deuterium (D or 2H) in the mix. This is because hydrogen atoms (hydrogen-1 and deuterium) are rapidly exchanged between water molecules. Water containing 50% H and 50% D in its hydrogen contains about 50% HDO and 25% each of H2O and D2O, in dynamic equilibrium.[2] In regular water, about 1 molecule in 3,200 is HDO (one hydrogen in 6,400 is D). By comparison, heavy water D2O[3] occurs at a proportion of about 1 molecule in 41 million (i.e., one in 6,4002). This makes semiheavy water far more common than "normal" heavy water.
The freezing point of semiheavy water is close to the freezing point of heavy water at 3.8°C compared to the 3.82°C of heavy water.
References
- ^ Tashakor S (2016-09-28). "Neutronic Investigation of Semi-Heavy Water Application in Hplwr New Flow Pattern". CNL Nuclear Review: 1–5. doi:10.12943/CNR.2016.00019.
- ^ Goncharuk VV, Kavitskaya AA, Romanyukina IY, Loboda OA (June 2013). "Revealing water's secrets: deuterium depleted water". Chemistry Central Journal. 7 (1): 103. doi:10.1186/1752-153X-7-103. PMC 3703265. PMID 23773696.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ "Heavy water | chemical compound". Encyclopedia Britannica. Retrieved 2019-04-24.
Further reading
- Schwarzer D, Lindner J, Vöhringer P (October 2005). "Energy relaxation versus spectral diffusion of the OH-stretching vibration of HOD in liquid-to-supercritical deuterated water". The Journal of Chemical Physics. 123 (16): 161105. Bibcode:2005JChPh.123p1105S. doi:10.1063/1.2110087. hdl:11858/00-001M-0000-0012-E7B7-2. PMID 16268674.