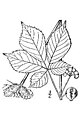
American ginseng
| American ginseng | |
|---|---|

| |
| Panax quinquefolius[1] | |
| Scientific classification | |
| Kingdom: | Plantae |
| Clade: | Tracheophytes |
| Clade: | Angiosperms |
| Clade: | Eudicots |
| Clade: | Asterids |
| Order: | Apiales |
| Family: | Araliaceae |
| Genus: | Panax |
| Subgenus: | Panax subg. Panax |
| Species: | P. quinquefolius
|
| Binomial name | |
| Panax quinquefolius | |

| |
| Synonyms[5] | |
|
Homotypic synonyms
Heterotypic synonyms
| |
American ginseng (Panax quinquefolius) is a species of flowering plant in the ivy family Araliaceae. It is native to eastern North America and introduced in China. The specific epithet quinquefolius means "five-leaved", which refers to the typical number of leaflets per leaf. It is one of a group of taxa known as "ginseng".
Europeans first became aware of American ginseng near Montreal in 1716. It has been wild-harvested and exported to Asia since 1720. Billions of plants were wild-harvested in the 19th century alone. To control international trade and prevent global extinction of the species, the United States Fish and Wildlife Service implements a CITES Export Program that authorizes 19 states and one tribe to export American ginseng from the United States. From 1978 to 2019, the bulk of exports have come from southern Appalachian states, especially Kentucky, West Virginia, and Tennessee.
The conservation status of American ginseng is globally vulnerable. It is imperiled or critically imperiled in 14 states and provinces. In Canada, the species is endangered and facing imminent extinction.
As wild populations declined in the late 19th century, American ginseng became a domesticated crop. It is cultivated primarily in Ontario, Wisconsin, British Columbia, and China. Canada is the largest producer and exporter of cultivated American ginseng in the world. It is the state herb of Wisconsin.
Description[edit]
Panax quinquefolius is a herbaceous perennial plant. Its aromatic root resembles a small parsnip that forks as it matures. The plant grows 6 to 18 in (15 to 46 cm) tall, usually bearing three compound leaves (sometimes called "prongs"), each with three to five leaflets, 2 to 5 in (5 to 13 cm) long.
Panax quinquefolius is sometimes confused with wild sarsaparilla (Aralia nudicaulis), another member of the ivy family (Araliaceae). The two species may be distinguished by their leaves. Panax quinquefolius has palmately compound leaves (with leaflets radiating from a single point) while Aralia nudicaulis has pinnately compound leaves (with leaflets arranged on either side of a central stalk).[6]
Phytochemistry[edit]

Like Asian ginseng (Panax ginseng), American ginseng contains dammarane-type ginsenosides, or saponins, as the major biologically active constituents. Dammarane-type ginsenosides include two classifications: 20(S)-protopanaxadiol (PPD) and 20(S)-protopanaxatriol (PPT). American ginseng contains high levels of Rb1, Rd (PPD classification), and Re (PPT classification) ginsenosides—higher than that of P. ginseng in one study.[7]
When taken orally, PPD-type ginsenosides are mostly metabolized by intestinal bacteria (anaerobes) to PPD monoglucoside, 20-O-beta-D-glucopyranosyl-20(S)-protopanaxadiol (M1).[8] In humans, M1 is detected in plasma starting seven hours after intake of PPD-type ginsenosides and in urine starting 12 hours after intake. These findings indicate M1 is the final metabolite of PPD-type ginsenosides.[9]
M1 is referred to in some articles as IH-901,[10] and in others as compound-K.[9]
Taxonomy[edit]


Panax quinquefolius was described as Aureliana canadensis by the French ethnologist and naturalist Joseph-François Lafitau in 1718.[11][12] As a Jesuit missionary in New France, Lafitau discovered ginseng near Montreal in 1716.[13][14] In his search for a specimen, Father Lafitau enlisted the help of the Iroquois by showing them a published botanical illustration of gin-seng,[15] a Chinese name for a plant now known as Panax ginseng.[16] The Iroquois referred to American ginseng as garent-oguen, which means "resembles man" or "a man's thigh" in Iroquoian language.
Aureliana canadensis was further described by the English naturalist Mark Catesby in 1747.[17] Catesby published a striking color illustration of a live specimen transplanted from Pennsylvania to the garden of English botanist Peter Collinson in Peckham.[18] Aureliana canadensis Lafitau ex Catesby is an invalid name since it was published prior to 1 May 1753 (Art.13.1 ICN 2018).[19]
The Swedish botanist Carl Linnaeus validly described Panax quinquefolium in 1753,[4][20] but the name was later corrected to Panax quinquefolius.[citation needed] Linnaeus placed Aureliana canadensis Lafitau ex Catesby in synonymy with Panax quinquefolius, citing both Lafitau [1718] and Catesby [1747]. Its type specimen, designated in 1991, was reportedly collected by Pehr Kalm near Quebec in 1749.[21] The specific epithet quinquefolius means "five-leaved",[22] which refers to the typical number of leaflets per leaf.
Etymology[edit]
The name ginseng derives from the Chinese herbalism term, jen-shen.[23] Other Chinese names are huaqishen (simplified Chinese: 花旗参; traditional Chinese: 花旗參; pinyin: huāqíshēn; Cantonese Yale: fākèihsām; lit. 'Flower Flag ginseng') or xiyangshen (simplified Chinese: 西洋参; traditional Chinese: 西洋參; pinyin: xīyángshēn; Cantonese Yale: sāiyèuhngsām; lit. 'west ocean ginseng').
The word "panax" is derived from the Greek 'Panakos' (panacea), in reference to the various benefits attributed to the herb.[24]
Distribution and habitat[edit]
Panax quinquefolius is native to eastern United States and southeastern Canada.[25][26][27][28] It is found primarily in the Appalachian and Ozark mountains of the United States where it prefers full shade environments in deciduous hardwood forests.[2][29] It is introduced and cultivated in the following Chinese provinces: Guizhou, Heilongjiang, Jiangsu, Jiangxi, Jilin, and Liaoning.[30]
Ecology[edit]
Panax quinquefolius is a summer flowering plant. In New England, flower buds and leaves emerge simultaneously around the middle of June, with flowers eventually appearing in July. Fruits mature to a deep red color by early September. The seeds exhibit a type of dormancy called morphophysiological dormancy, sometimes called "double dormancy", which requires two full winters to completely break dormancy. Germination finally takes place eighteen months after the fruit initially ripened.[31]
Exploitation and conservation[edit]
Europeans discovered ginseng near Montreal in 1716.[16] By 1720, ginseng from Quebec was exported to China by the Company of the Indies, a French trading company.[32] Within a few decades, ginseng populations near Montreal were suffering from overharvesting and habitat destruction, and so the wild-harvesting of ginseng began to spread south.[33] By the end of the 18th century, Albany, New York had become a trading center for ginseng. In the United States, 13.7 million kilograms of wild ginseng root were exported between 1821 and 1899,[34] with an average of about 1.7 million kilograms per decade.
| Pounds | Kilograms | Metric tons | |
|---|---|---|---|
| 1821–29 | 3,871,765 | 1,756,203 | 1,756 |
| 1830–39 | 3,192,375 | 1,448,036 | 1,448 |
| 1840–49 | 3,915,129 | 1,775,872 | 1,776 |
| 1850–59 | 1,999,999 | 907,184 | 907 |
| 1860–69 | 4,149,445 | 1,882,156 | 1,882 |
| 1870–79 | 4,041,727 | 1,833,296 | 1,833 |
| 1880–89 | 6,771,830 | 3,071,650 | 3,072 |
| 1890–99 | 2,163,302 | 981,257 | 981 |
| Total | 30,105,572 | 13,655,654 | 13,656 |
| Average per decade | 3,763,196 | 1,706,957 | 1,707 |
More than 290,000 kilograms of dry ginseng roots were exported from the United States (U.S.) in 1841. It is estimated that this represents at least 64 million roots.[35] By extrapolation, more than 3 billion roots (and therefore plants) were wild-harvested in the U.S. between 1821 and 1899.
During the twentieth century, exports of wild ginseng from the U.S. were about half of what they were during the previous century, averaging about 580,000 kilograms per decade.[36] Between 2000 and 2020, U.S. exports of wild ginseng dropped to approximately 250,000 kilograms per decade.
| Pounds | Kilograms | Metric tons | |
|---|---|---|---|
| 1980–89 | 1,034,539 | 469,259 | 469 |
| 1990–99 | 1,158,203 | 525,352 | 525 |
| 2000–09 | 648,632 | 294,215 | 294 |
| 2010–19 | 560,290 | 254,143 | 254 |
| Total | 3,401,664 | 1,542,969 | 1,542 |
| Average per decade | 850,416 | 385,742 | 385 |
The steady decline in export volume is likely the result of declining wild populations.
Export control[edit]
To control international trade, Panax quinquefolius is listed in Appendix II of the Convention on International Trade in Endangered Species (CITES).[37] CITES Appendix II includes species that, although currently not threatened with global extinction, may become so without trade controls. As of September 2022[update], nineteen (19) states and one tribe are authorized to export American ginseng from the United States.[38] The following table lists the states and tribe with an approved CITES export permit for American ginseng (each state's NatureServe conservation status is shown in parentheses):[39]
| Pounds | Kilograms | Metric tons | |
|---|---|---|---|
| Kentucky (S3) | 715,973 | 324,760 | 324.8 |
| West Virginia (S3S4) | 526,327 | 238,738 | 238.7 |
| Tennessee (S3S4) | 487,830 | 221,276 | 221.3 |
| North Carolina (S3S4) | 304,397 | 138,072 | 138.1 |
| Indiana (S3) | 299,009 | 135,628 | 135.6 |
| Virginia (S3S4) | 295,880 | 134,209 | 134.2 |
| New York (S4) | 249,903 | 113,354 | 113.4 |
| Ohio (S4) | 241,569 | 109,574 | 109.6 |
| Illinois (S3?) | 173,731 | 78,803 | 78.8 |
| Wisconsin (S4) | 102,830 | 46,643 | 46.6 |
| Missouri (S4) | 85,478 | 38,772 | 38.8 |
| Arkansas (S4) | 79,009 | 35,838 | 35.8 |
| Pennsylvania (S4) | 57,640 | 26,145 | 26.1 |
| Minnesota (S3) | 51,403 | 23,316 | 23.3 |
| Iowa (S3) | 42,402 | 19,233 | 19.2 |
| Alabama (S4) | 23,916 | 10,848 | 10.8 |
| Georgia (S3) | 23,166 | 10,508 | 10.5 |
| Maryland (S2S3) | 8,781 | 3,983 | 4.0 |
| Vermont (S3) | 6,334 | 2,873 | 2.9 |
| Menominee Nation (?) | 996 | 452 | 0.5 |
| Total | 3,776,574 | 1,713,025 | 1,713 |
From 1978 to 2019, 1,713,025 kilograms of wild and wild-simulated ginseng roots were legally exported from the United States. During this period, the bulk of exports came from southern Appalachian states including Kentucky, West Virginia, Tennessee, and North Carolina. Of these, eastern Kentucky has consistently large harvests, apparently a result of habitat abundance, very high levels of rural poverty, and a very strong local tradition of ginseng collecting.[40] American ginseng also grows wild in a number of states that do not permit export, including Connecticut (S2), Delaware (S2), Kansas (S1), Maine (S3), Massachusetts (S3), Michigan (S2S3), Nebraska (S1), New Hampshire (S2), New Jersey (S2), Oklahoma (S1), Rhode Island (S1), and South Carolina (S4).[41]
Status[edit]
As determined by the Committee on the Status of Endangered Wildlife in Canada, the conservation status of Panax quinquefolius is Endangered (E) in Canada.[42] In Ontario and Quebec, it is listed as Endangered and Threatened, respectively.[43] Both provincial designations refer to a species facing imminent extinction or extirpation. Consequently, the harvesting, possession, and export of wild American Ginseng in Canada is prohibited.
As of February 2024[update], the NatureServe conservation status of Panax quinquefolius is globally vulnerable (G3G4).[2] It is vulnerable (S3) in 14 states; imperiled (S2) in 8 states and provinces; critically imperiled (S1) in 6 states; and possibly extirpated (SH) in the District of Columbia.
In addition to (or in lieu of) the NatureServe conservation status (in parentheses below), some states designate their own conservation status:
- Connecticut (S2): Special Concern[44]
- Delaware (S2)
- Georgia (S3)
- Illinois (S3?)
- Indiana (S3)
- Iowa (S3)
- Kansas (S1)
- Kentucky (S3)
- Louisiana (S1)
- Maine (S3): Endangered[45]
- Maryland (S2S3)[46]
- Massachusetts (S3): Special Concern[47]
- Michigan (S2S3): Threatened[48]
- Minnesota (S3): Special Concern[49]
- Mississippi (S3)
- Nebraska (S1): Threatened[50]
- New Hampshire (S2): Threatened[51]
- New Jersey (S2): S1[52]
- New York (S4): Exploitably Vulnerable[53]
- North Carolina (S3S4): Rare but Relatively Secure; Exploited[54]
- Oklahoma (S1)
- Pennsylvania (S4): Pennsylvania Vulnerable[55]
- Rhode Island (S1): State Endangered[56]
- South Dakota (S1)
- Tennessee (S3S4): Special Concern; Commercially Exploited[57]
- Vermont (S3): High Priority Species of Greatest Conservation Need[58]
- Virginia (S3S4): Threatened[59]
- West Virginia (S3S4)
Panax quinquefolius is apparently secure (S4) in New York and Pennsylvania (as shown above), as well as Alabama, Arkansas, Missouri, Ohio, South Carolina, and Wisconsin.
Threats[edit]
American ginseng was formerly particularly widespread in the Appalachian and Ozark regions (and adjacent forested regions such as Pennsylvania, New York and Ontario). Due to its popularity and unique habitat requirements, the wild plant has been overharvested, as well as lost through destruction of its habitat, and is thus rare in most parts of the United States and Canada.[60][61] Ginseng is also negatively affected by deer browsing, urbanization, and habitat fragmentation.[62] Today the greatest threat to American ginseng is irresponsible digging of its wild roots for export.[2]
Cultivation[edit]
As wild populations of American ginseng began to decline in the late 19th century, a market developed for cultivated ginseng. In 1887, the tinsmith George Stanton planted ginseng in the forest around Apulia Station, a hamlet in the town of Fabius in Onondaga County, New York.[39] Within a few years, he was growing ginseng in the open under artificial shade, and by the late 1890s, he had the largest ginseng plantation in the United States.[63] In his obituary (1908), Stanton was called the Father of the Cultivated Ginseng Industry.[64][65]
Several associations were formed to support commercial growers of American ginseng. In 1902, George Stanton became the first president of the New York State Ginseng Growers Association. By 1913, the Wisconsin, Michigan, and Ohio growers' associations had the largest memberships.[66] American ginseng is the state herb of Wisconsin.[67]
American ginseng is grown commercially under artificial shade.[68] Under these conditions, a crop is harvested three to five years after seeding. Ginseng is also grown under forest-based, wild-simulated conditions, which require 6–10 years (or more) before harvest.[69] Based solely on yield, forest farming may be 1/10 as productive as commercial cultivation.[39]
American ginseng is commercially cultivated in Canada (60%), United States (30%), and China (7%).[70] In North America, it is cultivated primarily in Ontario, Wisconsin, and British Columbia. In 1994, Ontario and Wisconsin produced 1.5 and 2.0 million pounds of ginseng, respectively, while British Columbia produced 0.5 million pounds.[71] In other words, Canada and the United States each produced 2.0 million pounds (1,000 tons) of ginseng in 1994. In contrast, in 2007, Canada and the United States produced an estimated 6,486 tons and 1,054 tons (resp.) of ginseng, exporting $66 million and $37 million (resp.) worth of ginseng in 2009.[72] Based on these figures, Canada is the largest producer and exporter of American ginseng in the world.
Some states encourage the planting of ginseng both to restore natural habitats and to remove pressure from remaining wild populations. American ginseng is woods-cultivated in Colorado, Kentucky, Maine, North Carolina, Tennessee, Vermont, Virginia, and West Virginia.[73][74]
Toxicity[edit]
Individuals requiring anticoagulant therapy such as warfarin should avoid use of ginseng.[75] It is not recommended for individuals with impaired liver or renal function, or during pregnancy or breastfeeding.[75] Other adverse effects include headaches, anxiety, trouble sleeping and an upset stomach.[75]
Recent studies have shown that through the many cultivated procedures that American ginseng is grown, fungal molds, pesticides, and various metals and residues have contaminated the crop. Though these contaminating effects are not considerably substantial, they do pose health concerns that could lead to neurological problems, intoxication, cardiovascular disease and cancer.[76]
Uses[edit]
Ginseng has been a part of traditional Chinese medicine for over 2,000 years. In Asia, American ginseng is highly valued for its cooling and sedative medicinal effects (yin), whereas Asian ginseng embodies the warmer aspects of yang.[77]
American ginseng was of minor importance in traditional Native American medicine.[78][32][35] A number of the uses cited in the literature were likely adopted from the Chinese after the export trade from Canada to China began in 1720. The Iroquois ingested or smoked the roots as a panacea.[79] The Menominee in northern Wisconsin used it as a tonic and to increase one's mental capability,[80] while the Penobscot in Maine used it to promote fertility.[81] The Seminole in Florida used it for gunshot wounds.[82] Native peoples from multiple tribes gathered the roots to barter with white traders. In the late 19th century, the Cherokee sold large quantities of ginseng to traders for fifty cents a pound.[83]
Cold-fX is a product derived from the roots of American ginseng (Panax quinquefolius). The makers of Cold-fX were criticized for making health claims about the product that have never been tested or verified scientifically. Health Canada's review of the scientific literature confirmed that this is not a claim that the manufacturer is entitled to make.[84] More generally, there is no evidence that American ginseng is effective against the common cold.[75]
Culture[edit]
In the local vernacular, American ginseng has been variously known as "cheng", "chang", "sang", or "shang".[32] Those collecting it have been called "shangers" and hunting for it has been called "shanging".[85] In Appalachia, the wild-harvesting of American ginseng is called "sang hunting". Some blue-collar Appalachian families have been hunting sang for generations.[60][86]
Gallery[edit]
-
American ginseng in human figure.
-
Under wooden shade, American ginseng in late fall at Monk Garden in Wisconsin
-
A picture of the American ginseng plant with fruit.
-
American ginseng berries are ripe by late fall in Wisconsin.
-
A drawn image of the fruit and leaf of the American ginseng plant.
-
A drawn image of the American ginseng plants leaves.
-
American wild ginseng root (Panax quinquefolius). Old roots, ranging from 40–60 growth scars.
See also[edit]
- Panax trifolius, the dwarf ginseng
- Eleutherococcus senticosus, the Siberian ginseng
References[edit]
- ^ Panax_quinquefolius L., from "American medical botany being a collection of the native medicinal plants of the United States, containing their botanical history and chemical analysis, and properties and uses in medicine, diet and the arts" by Jacob Bigelow,1786/7-1879. Publication in Boston by Cummings and Hilliard,1817-1820.
- ^ a b c d "Panax quinquefolius". NatureServe Explorer. NatureServe. Retrieved 27 December 2023.
- ^ "Appendices | CITES". cites.org. Retrieved 2022-01-14.
- ^ a b "Panax quinquefolius L.". International Plant Names Index (IPNI). Royal Botanic Gardens, Kew; Harvard University Herbaria & Libraries; Australian National Botanic Gardens. Retrieved 2023-12-23.
- ^ "Panax quinquefolius L.". Plants of the World Online. Royal Botanic Gardens, Kew. Retrieved 2023-12-23.
- ^ Firestone, Chris K. (April 1, 2022). "Be aware—no foolin'!". Pennsylvania Department of Conservation and Natural Resources. Retrieved 14 January 2024.
- ^ Zhu, S.; Zou, K.; Fushimi, H.; Cai, S.; Komatsu, K. (2004). "Comparative study on triterpene saponins of ginseng drugs". Planta Medica. 70 (7): 666–677. doi:10.1055/s-2004-827192. PMID 15303259. S2CID 260251856.
- ^ Hasegawa, H.; Sung, J.-H.; Matsumiya, S.; Uchiyama, M. (1996). "Main ginseng saponin metabolites formed by intestinal bacteria". Planta Medica. 62 (5): 453–457. doi:10.1055/s-2006-957938. PMID 8923812. S2CID 260252578.
- ^ a b Tawab, M. A.; Bahr, U.; Karas, M.; Wurglics, M.; Schubert-Zsilavecz, M. (2003). "Degradation of ginsenosides in humans after oral administration". Drug Metabolism and Disposition. 31 (8): 1065–1071. doi:10.1124/dmd.31.8.1065. PMID 12867496. S2CID 17686376.
- ^ Oh, S.-H.; Lee, B.-H. (2004). "A ginseng saponin metabolite-induced apoptosis in HepG2 cells involves a mitochondria-mediated pathway and its downstream caspase-8 activation and Bid cleavage". Toxicology and Applied Pharmacology. 194 (3): 221–229. doi:10.1016/j.taap.2003.09.011. PMID 14761678.
- ^ Lafitau, Joseph-Francois (1718). Memoire presente a Son Altesse royale Monseigneur le duc dOrleans, regent du royaume de France : concernant la precieuse plante du gin-seng de Tartarie, decouverte en Canada par le P. Joseph Francois Lafitau. Paris: Chez Joseph Monge, rue S. Jacques vis-a-vis le College de Louis le Grand, a Saint Ignace. Retrieved 30 December 2023.
- ^ Belanus, Betty. "The Art of Ginseng—Illustration 4: La Plante du Gin-seng by Joseph-François Lafitau, 1718". Smithsonian Center for Folklife and Cultural Heritage. Retrieved 4 January 2024.
- ^ Carlson (1986), pp. 233–234.
- ^ Fenton, William N. (1974). "Lafitau, Joseph-François". In Halpenny, Francess G (ed.). Dictionary of Canadian Biography. Vol. III (1741–1770) (online ed.). University of Toronto Press.
- ^ Jartoux (1713), Tab. V (between pages 242 and 243).
- ^ a b Brinckmann & Huang (2018), pp. 908–910.
- ^ Catesby, Mark (1747). The Natural History of Carolina, Florida and the Bahama islands. Appendix. London. p. 16. Retrieved 3 January 2024.
- ^ Belanus, Betty. "The Art of Ginseng—Illustration 5: The Whip-Poor-Will". Smithsonian Center for Folklife and Cultural Heritage. Retrieved 4 January 2024.
- ^ "Aureliana canadensis Lafitau ex Catesby". International Plant Names Index (IPNI). Royal Botanic Gardens, Kew; Harvard University Herbaria & Libraries; Australian National Botanic Gardens. Retrieved 2024-01-08.
- ^ Linnaeus (1753), vol. 2, pp. 1058–1059.
- ^ Reveal, James L. (December 1991). "Typification of Panax quinquefolium Linnaeus (Araliaceae)". Phytologia. 71 (6): 472–474. Retrieved 24 December 2023.
- ^ Gledhill (2008), p. 324.
- ^ "Ginseng". Online Etymology Dictionary, Douglas Harper. 2018. Retrieved 14 September 2018.
- ^ "Benefits of American Ginseng: Dosage & Safety – Botanical Institute". Retrieved 2021-08-27.
- ^ USDA, NRCS (n.d.). "Panax quinquefolius". The PLANTS Database (plants.usda.gov). Greensboro, North Carolina: National Plant Data Team. Retrieved 27 December 2023.
- ^ "Panax quinquefolius". State-level distribution map from the North American Plant Atlas (NAPA). Biota of North America Program (BONAP). 2014. Retrieved 27 December 2023.
- ^ "Panax quinquefolius". County-level distribution map from the North American Plant Atlas (NAPA). Biota of North America Program (BONAP). 2014. Retrieved 27 December 2023.
- ^ "Panax quinquefolius Linnaeus". Canadensys. Retrieved 27 December 2023.
- ^ Anderson, M. Kat; Peterson, J. Scott. "American Ginseng" (PDF). USDA, NRCS. Retrieved 27 December 2023.
- ^ Xiang, Qibai; Lowry, Porter P. "Panax quinquefolius". Flora of China. Vol. 13. Retrieved 25 December 2023 – via eFloras.org, Missouri Botanical Garden, St. Louis, MO & Harvard University Herbaria, Cambridge, MA.
- ^ Hu, Rüdenberg & Del Tredici (1980), p. 630.
- ^ a b c Carlson (1986), p. 234.
- ^ Manget (2017), pp. 55–56.
- ^ Carlson (1986), p. 239 (Table 1).
- ^ a b McGraw et al. (2013), p. 2.
- ^ Liu et al. (2021), Figure 1.
- ^ "Appendices I, II and III". Convention on International Trade in Endangered Species of Wild Flora and Fauna. 2010-10-14. Retrieved 2020-01-30.
- ^ Chief, Branch of Monitoring and Consultation, Division of Scientific Authority, USFWS (1 September 2022). "General Advice for the export of wild and wild-simulated American ginseng (Panax quinquefolius) roots legally harvested during the 2022 harvest season in the 19 States and Tribe with an approved CITES Export Program for American ginseng" (PDF). Letter to Chief, Branch of Permits, Division of Management Authority, USFWS. Washington, DC: United States Fish and Wildlife Service (USFWS). Retrieved 10 January 2024.
- ^ a b c Liu et al. (2021).
- ^ Schmidt et al. (2019), p. 145.
- ^ Schmidt et al. (2019), p. 141.
- ^ "American Ginseng (Panax quinquefolius)". Government of Canada. Retrieved 6 January 2024.
- ^ "Recovery Strategy for the American Ginseng (Panax quinquefolius) in Canada 2018". Government of Canada. Retrieved 6 January 2024.
- ^ "Endangered, Threatened, and Special Concern Plants". Connecticut Department of Energy & Environmental Protection. August 5, 2015. Retrieved 15 January 2024.
- ^ "Panax quinquefolius L.". Maine Department of Agriculture, Conservation & Forestry. Retrieved 15 January 2024.
- ^ "Rare, Threatened, and Endangered Plants of Maryland" (PDF). Maryland Department of Natural Resources. March 2021. Retrieved 15 January 2024.
- ^ "American Ginseng, Panax quinquefolius L.". Massachusetts Division of Fisheries & Wildlife. Retrieved 15 January 2024.
- ^ Penskar, M. R.; Higman, P. J. (1996). "Special plant abstract for Panax quinquefolius L. (ginseng)" (PDF). Lansing: Michigan Natural Features Inventory. Retrieved 15 January 2024.
- ^ "Panax quinquefolius L. (American ginseng)". Minnesota Department of Natural Resources. Retrieved 15 January 2024.
- ^ "American ginseng". Nebraska Game and Parks Commission. Retrieved 15 January 2024.
- ^ "New Hampshire Official Rare Plants List". New Hampshire Department of Natural and Cultural Resources. 2020. Retrieved 15 January 2024.
- ^ "New Jersey State Forest Action Plan" (PDF). New Jersey Department of Environmental Protection. December 2020. Retrieved 15 January 2024.
- ^ Ring, Richard M. (December 2022). "New York Rare Plant Status Lists" (PDF). New York Natural Heritage Program. Retrieved 15 January 2024.
- ^ "Rare Plant List". North Carolina Natural Heritage Program. 2022. Retrieved 15 January 2024.
- ^ "American Ginseng in Pennsylvania". Pennsylvania Department of Conservation and Natural Resources. Retrieved 15 January 2024.
- ^ "Rhode Island Rare Plants" (PDF). Rhode Island Natural History Survey. 2016. Retrieved 15 January 2024.
- ^ "Tennessee Ginseng Program" (PDF). Tennessee Department of Environment and Conservation. Retrieved 15 January 2024.
- ^ "Species of Greatest Conservation Need" (PDF). Vermont Department of Fish & Wildlife. 2015. Retrieved 15 January 2024.
- ^ "Ginseng". Virginia Department of Agriculture and Consumer Services. Retrieved 15 January 2024.
- ^ a b Cataneo, Emily (4 November 2020). "In Appalachia, a Plan to Save Wild Ginseng". Undark. Retrieved 22 December 2023.
- ^ Beattie-Moss, Melissa (June 18, 2006). "Roots and Regulations: The unfolding story of Pennsylvania ginseng". PennState. Retrieved 28 December 2023.
- ^ "Population Biology and Conservation Ecology of American Ginseng". West Virginia University. Archived from the original on 2022-04-07.
- ^ Manget (2017), pp. 267–277.
- ^ "George Stanton and A.R. Harding, early figures in ginseng knowledge". American Ginseng: Local Knowledge, Global Roots. Smithsonian Institution. Retrieved 9 January 2024.
- ^ Bramer, J. K. (March 1908). "Obituary: George Stanton". Special Crops. 7 (67): 42–43. Retrieved 9 January 2024.
- ^ Carlson (1986), p. 235–238.
- ^ Kasper, Madeline (December 2017). "Ginseng: the official herb of Wisconsin" (PDF). Wisconsin Legislative Reference Bureau Reports. 1 (6). Retrieved 12 January 2024.
- ^ "Ginseng production in Ontario". Ontario Ministry of Agriculture, Food and Rural Affairs. September 22, 2022. Retrieved 1 January 2024.
- ^ "Wild-Simulated Forest Farming for Ginseng Production". Association for Temperate Agroforestry. Retrieved 1 January 2024.
- ^ Xiao, Max (2000-02-07). "The Canadian Ginseng Industry" (PDF). Agriculture and Agri-Food Canada. Archived (PDF) from the original on 2023-12-29. Retrieved 29 December 2023.
- ^ Jia & Zhao (2009).
- ^ Baeg & So (2013).
- ^ "There is More to a Forest than Trees". Virginia Tech. Summer 2002. Archived from the original on 2006-08-28. Retrieved 2023-12-27.
- ^ "Ginseng program". Kentucky Agriculture Department. 2017. Archived from the original on 2010-06-26. Retrieved 2018-02-20.
- ^ a b c d "Asian ginseng". National Center for Complementary and Integrative Health, US National Institutes of Health, Bethesda, MD. September 2016. Retrieved 10 February 2017.
- ^ Wang, Zengui; Huang, Linfang (March 2015). "Panax quinquefolius: An overview of the contaminants". Phytochemistry Letters. 11: 89–94. Bibcode:2015PChL...11...89W. doi:10.1016/j.phytol.2014.11.013.
- ^ "About Ontario Ginseng". Ontario Ginseng Growers Association. Retrieved 27 December 2023.
- ^ Brinckmann & Huang (2018), pp. 910–911.
- ^ "American Ginseng: Iroquois". Native American Ethnobotany Database. Retrieved 28 December 2023.
- ^ "American Ginseng: Menominee". Native American Ethnobotany Database. Retrieved 28 December 2023.
- ^ "American Ginseng: Penobscot". Native American Ethnobotany Database. Retrieved 28 December 2023.
- ^ "American Ginseng: Seminole". Native American Ethnobotany Database. Retrieved 28 December 2023.
- ^ "American Ginseng: Cherokee". Native American Ethnobotany Database. Retrieved 28 December 2023.
- ^ Charlie Gillis (2007-03-26). "COLD-fX catches the sniffles again". Maclean's Magazine. Archived from the original on 2012-02-07.
- ^ Gilman (2015), p. 492.
- ^ "Ginseng (sang), the thrill of the hunt". Pike County Historical Society. March 2022. Retrieved 24 December 2023.
Bibliography[edit]
- Baeg, In-Ho; So, Seung-Ho (2013). "The world ginseng market and the ginseng (Korea)". Journal of Ginseng Research. 37 (1): 1–7. doi:10.5142/jgr.2013.37.1. PMC 3659626. PMID 23717152.
- Brinckmann, Josef; Huang, Linfang (2018-11-01). "American Ginseng a Genuine Traditional Chinese Medicine". Medicina Nei Secoli: Journal of History of Medicine and Medical Humanities. 30 (3): 907–928. ISSN 0394-9001. Retrieved 29 December 2023.
- Burkhart, Eric P.; Jacobson, Michael G.; Finley, James (2012). "A case study of stakeholder perspective and experience with wild American ginseng (Panax quinquefolius) conservation efforts in Pennsylvania, U.S.A.: limitations to a CITES driven, top-down regulatory approach" (PDF). Biodiversity and Conservation. 21 (14): 3657–3679. Bibcode:2012BiCon..21.3657B. doi:10.1007/s10531-012-0389-9. S2CID 14768055. Retrieved 28 December 2023.
- Burkhart, Eric P. (2013). "American ginseng (Panax quinquefolius L.) floristic associations in Pennsylvania: guidance for identifying calcium-rich forest farming sites" (PDF). Agroforestry Systems. 87 (5): 1157–1172. Bibcode:2013AgrSy..87.1157B. doi:10.1007/s10457-013-9627-8. S2CID 254191922. Retrieved 22 December 2023.
- Carlson, Alvar W. (1986). "Ginseng: America's Botanical Drug Connection to the Orient" (PDF). Economic Botany. 40 (2): 233–249. doi:10.1007/BF02859148. JSTOR 4254858. S2CID 23019185. Retrieved 26 December 2023.
- Gilman, Arthur V. (2015). New Flora of Vermont. Memoirs of The New York Botanical Garden, Volume 110. Bronx, New York, USA: The New York Botanical Garden Press. ISBN 978-0-89327-516-7.
- Gledhill, David (2008). The Names of Plants (4th ed.). Cambridge University Press. ISBN 978-0-521-86645-3.
- Hu, Shiu Ying; Rüdenberg, Lily; Del Tredici, Peter (1980). "Studies of American ginsengs". Rhodora. 82 (832): 627–636. ISSN 0035-4902. JSTOR 23314100. Retrieved 2023-12-22.
- Jartoux, Pierre (1713). "XXV. The description of a tartarian plant, call'd gin-seng; with an account of its virtues. In a letter from Father Jartoux, to the Procurator General of the Missions of India and China. Taken from the tenth volume of letters of the Missionary Jesuits, printed in Paris in octavo, 1713". Philosophical Transactions of the Royal Society of London. 28 (337): 237–247. doi:10.1098/rstl.1713.0025.
- Jia, Lee; Zhao, Yuqing (2009). "Current evaluation of the millennium phytomedicine-ginseng (I): etymology, pharmacognosy, phytochemistry, market and regulations". Current Medicinal Chemistry. 16 (19): 2475–84. doi:10.2174/092986709788682146. PMC 2752963. PMID 19601793.
- Linnaeus, Carl (1753). Species Plantarum: exhibentes plantas rite cognitas, ad genera relatas, cum differentiis specificis, nominibus trivialibus, synonymis selectis, locis natalibus, secundum systema sexuale digestas (1st ed.). Stockholm: Impensis Laurentii Salvii. Retrieved 3 August 2023.
- Liu, Hong; Burkhart, Eric P.; Chen, Vivian Yi Ju; Wei, Xi (17 March 2021). "Promotion of in situ Forest Farmed American Ginseng (Panax quinquefolius L.) as a Sustainable Use Strategy: Opportunities and Challenges". Frontiers in Ecology and Evolution. 9 (652103). doi:10.3389/fevo.2021.652103.
- Manget, Thomas Luke (2017). Root digging and herb gathering: the rise and decline of the botanical drug trade in southern Appalachia (PhD). Athens: The University of Georgia.
- McGraw, James B.; Lubbers, Anne E.; Van der Voort, Martha; Mooney, Emily H.; Furedi, Mary Ann; Souther, Sara; Turner, Jessica B.; Chandler, Jennifer (2013). "Ecology and conservation of ginseng (Panax quinquefolius) in a changing world" (PDF). Annals of the New York Academy of Sciences: The Year in Ecology and Conservation Biology. 1286 (1): 62–91. Bibcode:2013NYASA1286...62M. doi:10.1111/nyas.12032. ISSN 0077-8923. PMID 23398402. S2CID 20938136. Retrieved 2 January 2024.
- Schmidt, John Paul; Cruse-Sanders, Jennifer; Chamberlain, James L.; Ferreira, Susana; Younge, John A. (March 2019). "Explaining harvests of wild-harvested herbaceous plants: American ginseng as a case study" (PDF). Biological Conservation. 231: 139–149. Bibcode:2019BCons.231..139S. doi:10.1016/j.biocon.2019.01.006. Retrieved 17 January 2024.
External links[edit]
- "American Ginseng: Local Knowledge, Global Roots". Smithsonian Institution. Retrieved 24 December 2023.
- Belanus, Betty. "The Art of Ginseng: Images that Tell the Story of American Ginseng and Reveal its Hidden Presence Throughout our Shared Lands and Histories". Smithsonian Center for Folklife and Cultural Heritage. Retrieved 3 January 2024.
- Ellison, George (November 9, 2011). "Ginseng, ferns and an ancient dialect". Smoky Mountain News. Retrieved 24 December 2023.
- "Sang". University of South Carolina. Retrieved 24 December 2023.
- "American ginseng culture in the arid climates of British Columbia" (PDF). B.C. Ministry of Agriculture, Fisheries, and Food. 1992. Retrieved 1 January 2024.
- "Wild American Ginseng". Wild American Ginseng Conservation Collaborative. Retrieved 2 January 2024.
- Rice, Laura (June 8, 2010). "Let's Eat: Adirondack Ginseng". The Adirondack Almanack. Retrieved 3 January 2024.
- Azami, Bahaji; Lena, Nisma; Yu, Qun; Shen, Yun-Hui (2021). "A Survey of the History and Applications of American Ginseng". Chinese Medicine and Culture. 4 (1): 12–18. doi:10.4103/CMAC.CMAC_41_20.
- Banks, James. "American Ginseng History". National Ginseng LLC. Retrieved 8 January 2024.
- "Jim McGraw's Lab". West Virginia University. Archived from the original on 2015-01-22.
- "Panax quinquefolius — American ginseng". Go Botany. Native Plant Trust. Retrieved 15 January 2024.