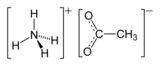
Ammonium acetate
| |

| |
| Names | |
|---|---|
| IUPAC name
Ammonium ethanoate
| |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| ECHA InfoCard | 100.010.149 |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C2H7NO2 | |
| Molar mass | 77.083 g·mol−1 |
| Appearance | White solid crystals, deliquescent |
| Odor | Slightly acetic |
| Density | 1.17 g/cm3 (20 °C)[1] 1.073 g/cm3 (25 °C) |
| Melting point | 113 °C (235 °F; 386 K) [4] |
| 102 g/100 mL (0 °C) 148 g/100 mL (4 °C)[1] 143 g/100 mL (20 °C) 533 g/100 mL (80 °C) | |
| Solubility | Soluble in alcohol, SO2, acetone, liquid ammonia[2] |
| Solubility in methanol | 7.89 g/100 mL (15 °C)[3][1] 131.24 g/100 g (94.2 °C)[2] |
| Solubility in dimethylformamide | 0.1 g/100 g[2] |
| Structure | |
| Orthorhombic | |
| Thermochemistry | |
Std enthalpy of
formation (ΔfH⦵298) |
−615 kJ/mol[2] |
| Hazards | |
| Occupational safety and health (OHS/OSH): | |
Main hazards
|
Irritant |
| GHS labelling: | |
 [3] [3]
| |
| Warning | |
| H303, H316, H320, H333[3] | |
| P281, P335[3] | |
| NFPA 704 (fire diamond) | |
| Flash point | 136 °C (277 °F; 409 K) [3] |
| Lethal dose or concentration (LD, LC): | |
LD50 (median dose)
|
386 mg/kg (mice, intravenous)[2] |
| Safety data sheet (SDS) | JT Baker |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Ammonium acetate is a chemical compound with the formula NH4CH3CO2. It is a white, hygroscopic solid and can be derived from the reaction of ammonia and acetic acid. It is available commercially.[5]
Uses
It is the main precursor to acetamide:[6]
- NH4CH3CO2 → CH3C(O)NH2 + H2O
It is also used as a diuretic.[5]
Buffer
As the salt of a weak acid and a weak base, ammonium acetate is often used with acetic acid to create a buffer solution. Ammonium acetate is volatile at low pressures. Because of this, it has been used to replace cell buffers with non-volatile salts in preparing samples for mass spectrometry.[7] It is also popular as a buffer for mobile phases for HPLC with ELSD detection for this reason. Other volatile salts that have been used for this include ammonium formate.
Other
- NH4C2H3O2 is occasionally employed as a biodegradable de-icing agent.
- Ammonium acetate is useful as a catalyst in the Knoevenagel condensation and as a source of ammonia in the Borch reaction in organic synthesis.
- a protein precipitating reagent.
- In dialysis as part of a protein purification step to remove contaminants via diffusion.
- In agricultural chemistry used as reagent for determination of soil CEC (Cation Exchange Capacity ) and determination of available potassium in soil, ammonium ion is act as an replacement cation.
Food additive
Ammonium acetate is also used as a food additive as an acidity regulator; INS number 264. It is approved for usage in Australia and New Zealand.[8]
Production
Ammonium acetate is produced by the neutralization of acetic acid with ammonium carbonate or by saturating glacial acetic acid with ammonia.[9] Obtaining crystalline ammonium acetate is difficult on account of its aqueous solution giving off ammonia when evaporated.
References
- ^ a b c Pradyot, Patnaik (2003). Handbook of Inorganic Chemicals. The McGraw-Hill Companies, Inc. ISBN 0-07-049439-8.
- ^ a b c d e http://chemister.ru/Database/properties-en.php?dbid=1&id=354
- ^ a b c d e f "Safety Data Sheet of Ammonium Acetate" (PDF). http://www.tedia.com. Tedia Company Inc. 2011-08-12. Retrieved 2014-06-10.
{{cite web}}: External link in|website= - ^ Davidson, Arthur W.; McAllister, Walter H. (1930). "SOLUTIONS OF SALTS IN PURE ACETIC ACID. II. SOLUBILITIES OF ACETATES1". Journal of the American Chemical Society. 52 (2): 507–519. doi:10.1021/ja01365a010. ISSN 0002-7863.
- ^ a b Hosea Cheung; Robin S. Tanke; G. Paul Torrence. "Acetic Acid". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a01_045.pub2. ISBN 978-3527306732.
- ^ Coleman, G. H.; Alvarado, A. M. (1923). "Acetamide". Organic Syntheses. 3: 3; Collected Volumes, vol. 1, p. 3.
- ^ Berman et al., 2008. J Am Soc Mass Spectrom, 19:1230-1236.
- ^ Australia New Zealand Food Standards Code "Standard 1.2.4 - Labelling of ingredients". Retrieved 2011-10-27.
- ^ Brannt, William (1914). A practical treatise on the manufacture of vinegar. Lancaster, PA: Henry Carey Baird & Co. pp. 316–317.

