Electron shell
In chemistry and atomic physics, an electron shell may be thought of as an orbit that electrons follow around an atom's nucleus. The closest shell to the nucleus is called the "1 shell" (also called the "K shell"), followed by the "2 shell" (or "L shell"), then the "3 shell" (or "M shell"), and so on further and further from the nucleus. The shells correspond to the principal quantum numbers (n = 1, 2, 3, 4 ...) or are labeled alphabetically with the letters used in X-ray notation (K, L, M, ...). A useful guide when understanding electron shells in atoms is to note that each row on the conventional periodic table of elements represents an electron shell.
Each shell can contain only a fixed number of electrons: the first shell can hold up to two electrons, the second shell can hold up to eight electrons, the third shell can hold up to 18, continiuing as the general formula of the nth shell being able to hold up to 2(n2) electrons.[1] For an explanation of why electrons exist in these shells, see electron configuration.[2]
Each shell consists of one or more subshells, and each subshell consists of one or more atomic orbitals.
History
In 1913, Niels Bohr proposed a model of the atom, giving the arrangement of electrons in their sequential orbits. At that time, Bohr allowed the capacity of the inner orbit of the atom to increase to eight electrons as the atoms got larger, and "in the scheme given below the number of electrons in this [outer] ring is arbitrary put equal to the normal valency of the corresponding element". Using these and other constraints, he proposed configurations that are in accord with those now known only for the first six elements. "From the above we are led to the following possible scheme for the arrangement of the electrons in light atoms:"[3][4]
| Element | Electrons per shell | Element | Electrons per shell | Element | Electrons per shell |
|---|---|---|---|---|---|
| 1 | 1 | 9 | 4, 4, 1 | 17 | 8, 4, 4, 1 |
| 2 | 2 | 10 | 8, 2 | 18 | 8, 8, 2 |
| 3 | 2, 1 | 11 | 8, 2, 1 | 19 | 8, 8, 2, 1 |
| 4 | 2, 2 | 12 | 8, 2, 2 | 20 | 8, 8, 2, 2 |
| 5 | 2, 3 | 13 | 8, 2, 3 | 21 | 8, 8, 2, 3 |
| 6 | 2, 4 | 14 | 8, 2, 4 | 22 | 8, 8, 2, 4 |
| 7 | 4, 3 | 15 | 8, 4, 3 | 23 | 8, 8, 4, 3 |
| 8 | 4, 2, 2 | 16 | 8, 4, 2, 2 | 24 | 8, 8, 4, 2, 2 |
The shell terminology comes from Arnold Sommerfeld's modification of the 1913 Bohr model. During this period Bohr was working with Walther Kossel, whose papers in 1914 and in 1916 called the orbits "shells".[5][6] Sommerfeld retained Bohr's planetary model, but added mildly elliptical orbits (characterized by additional quantum numbers ℓ and m) to explain the fine spectroscopic structure of some elements.[7] The multiple electrons with the same principal quantum number (n) had close orbits that formed a "shell" of positive thickness instead of the circular orbit of Bohr's model which orbits called "rings" were described by a plane.[8]
The existence of electron shells was first observed experimentally in Charles Barkla's and Henry Moseley's X-ray absorption studies. Moseley's work did not directly concern the study of electron shells, because he was trying to prove that the periodic table was not arranged by weight, but by the charge of the protons in the nucleus.[9] However, because the number of electrons in an electrically neutral atom equals the number of protons, this work was extremely important to Niels Bohr who mentioned Moseley's work several times in his 1962 interview.[10] Moseley was part of Rutherford's group, as was Niels Bohr. Moseley measured the frequencies of X-rays emitted by every element between calcium and zinc and found that the frequencies became greater as the elements got heavier. This led to the theory that electrons were emitting X-rays when they were shifted to lower shells.[11] This led to the conclusion that the electrons were in Kossel's shells with a definite limit per shell, labeling them with the letters K, L, M, N, O, P, and Q.[4][12] The origin of this terminology was alphabetic. Barkla, who worked independently from Moseley as an X-ray spectrometry experimentalist, first noticed two distinct types of scattering from shooting X-rays at elements in 1909 and named them "A" and "B". Barkla described these two types of X-ray diffraction: the first was unconnected with the type of material used in the experiment and could be polarized. The second diffraction beam he called "fluorescent" because it depended on the irradiated material.[13] It was not known what these lines meant at the time, but in 1911 Barkla decided there might be scattering lines previous to "A", so he began at "K".[14] However, later experiments indicated that the K absorption lines are produced by the innermost electrons. These letters were later found to correspond to the n values 1, 2, 3, etc. that were used in the Bohr model. They are used in the spectroscopic Siegbahn notation.
The work of assigning electrons to shells was continued from 1913 to 1925 by many chemists and a few physicists. Niels Bohr was one of the few physicists who followed the chemist's work[15] of defining the periodic table, while Arnold Sommerfeld worked more on trying to make a relativistic working model of the atom that would explain the fine structure of the spectra from a classical orbital physics standpoint through the Atombau approach.[4] Einstein and Rutherford, who did not follow chemistry, were unaware of the chemists who were developing electron shell theories of the periodic table from a chemistry point of view, such as Irving Langmuir, Charles Bury, J.J. Thomson, and Gilbert Lewis, who all introduced corrections to Bohr's model such as a maximum of two electrons in the first shell, eight in the next and so on, and were responsible for explaining valency in the outer electron shells, and the building up of atoms by adding electrons to the outer shells.[16][4] So when Bohr outlined his electron shell atomic theory in 1922, there was no mathematical formula for the theory. So Rutherford said he was hard put "to form an idea of how you arrive at your conclusions".[17][18] Einstein said of Bohr's 1922 paper that his "electron-shells of the atoms together with their significance for chemistry appeared to me like a miracle – and appears to me as a miracle even today".[19] Arnold Sommerfeld, who had followed the Atombau structure of electrons instead of Bohr who was familiar with the chemists' views of electron structure, spoke of Bohr's 1921 lecture and 1922 article on the shell model as "the greatest advance in atomic structure since 1913".[4][20][17] However, the electron shell development of Niels Bohr was basically the same theory as that of the chemist Charles Rugeley Bury in his 1921 paper.[21][4][22]
As work continued on the electron shell structure of the Sommerfeld-Bohr Model, Sommerfeld had introduced three "quantum numbers n, k, and m, that described the size of the orbit, the shape of the orbit, and the direction in which the orbit was pointing."[23] Because we use k for the Boltzmann constant, the azimuthal quantum number was changed to ℓ. When the modern quantum mechanics theory was put forward based on Heisenberg's matrix mechanics and Schrödinger's wave equation, these quantum numbers were kept in the current quantum theory but were changed to n being the principal quantum number, and m being the magnetic quantum number.
However, the final form of the electron shell model still in use today for the number of electrons in shells was discovered in 1923 by Edmund Stoner, who introduced the principle that the nth shell was described by 2(n2). Seeing this in 1925, Wolfgang Pauli added a fourth quantum number, "spin", during the old quantum theory period of the Sommerfeld-Bohr Solar System atom to complete the modern electron shell theory.[4]
Subshells
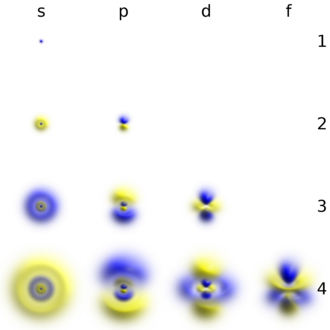
Each shell is composed of one or more subshells, which are themselves composed of atomic orbitals. For example, the first (K) shell has one subshell, called 1s; the second (L) shell has two subshells, called 2s and 2p; the third shell has 3s, 3p, and 3d; the fourth shell has 4s, 4p, 4d and 4f; the fifth shell has 5s, 5p, 5d, and 5f and can theoretically hold more in the 5g subshell that is not occupied in the ground-state electron configuration of any known element.[2] The various possible subshells are shown in the following table:
| Subshell label | ℓ | Max electrons | Shells containing it | Historical name |
|---|---|---|---|---|
| s | 0 | 2 | Every shell | sharp |
| p | 1 | 6 | 2nd shell and higher | principal |
| d | 2 | 10 | 3rd shell and higher | diffuse |
| f | 3 | 14 | 4th shell and higher | fundamental |
| g | 4 | 18 | 5th shell and higher (theoretically) | (next in alphabet after f)[24] |
- The first column is the "subshell label", a lowercase-letter label for the type of subshell. For example, the "4s subshell" is a subshell of the fourth (N) shell, with the type (s) described in the first row.
- The second column is the azimuthal quantum number (ℓ) of the subshell. The precise definition involves quantum mechanics, but it is a number that characterizes the subshell.
- The third column is the maximum number of electrons that can be put into a subshell of that type. For example, the top row says that each s-type subshell (1s, 2s, etc.) can have at most two electrons in it. Each of the following subshells (p, d, f, g) can have 4 more electrons than the one preceding it.
- The fourth column says which shells have a subshell of that type. For example, looking at the top two rows, every shell has an s subshell, while only the second shell and higher have a p subshell (i.e., there is no "1p" subshell).
- The final column gives the historical origin of the labels s, p, d, and f. They come from early studies of atomic spectral lines. The other labels, namely g, h, and i, are an alphabetic continuation following the last historically originated label of f.
Number of electrons in each shell
Each subshell is constrained to hold 4ℓ + 2 electrons at most, namely:
- Each s subshell holds at most 2 electrons
- Each p subshell holds at most 6 electrons
- Each d subshell holds at most 10 electrons
- Each f subshell holds at most 14 electrons
- Each g subshell holds at most 18 electrons
Therefore, the K shell, which contains only an s subshell, can hold up to 2 electrons; the L shell, which contains an s and a p, can hold up to 2 + 6 = 8 electrons, and so forth; in general, the nth shell can hold up to 2n2 electrons.[1]
| Shell name |
Subshell name |
Subshell max electrons |
Shell max electrons |
|---|---|---|---|
| K | 1s | 2 | 2 |
| L | 2s | 2 | 2 + 6 = 8 |
| 2p | 6 | ||
| M | 3s | 2 | 2 + 6 + 10 = 18 |
| 3p | 6 | ||
| 3d | 10 | ||
| N | 4s | 2 | 2 + 6 + 10 + 14 = 32 |
| 4p | 6 | ||
| 4d | 10 | ||
| 4f | 14 | ||
| O | 5s | 2 | 2 + 6 + 10 + 14 + 18 = 50 |
| 5p | 6 | ||
| 5d | 10 | ||
| 5f | 14 | ||
| 5g | 18 |
Although that formula gives the maximum in principle, in fact that maximum is only achieved (in known elements) for the first four shells (K, L, M, N). No known element has more than 32 electrons in any one shell.[25][26] This is because the subshells are filled according to the Aufbau principle. The first elements to have more than 32 electrons in one shell would belong to the g-block of period 8 of the periodic table. These elements would have some electrons in their 5g subshell and thus have more than 32 electrons in the O shell (fifth principal shell).
Subshell energies and filling order


Although it is sometimes stated that all the electrons in a shell have the same energy, this is an approximation. However, the electrons in one subshell do have exactly the same level of energy, with later subshells having more energy per electron than earlier ones. This effect is great enough that the energy ranges associated with shells can overlap.
The filling of the shells and subshells with electrons proceeds from subshells of lower energy to subshells of higher energy. This follows the n + ℓ rule which is also commonly known as the Madelung rule. Subshells with a lower n + ℓ value are filled before those with higher n + ℓ values. In the case of equal n + ℓ values, the subshell with a lower n value is filled first.
Because of this, the later shells are filled over vast sections of the periodic table. The K shell fills in the first period (hydrogen and helium), while the L shell fills in the second (lithium to neon). However, the M shell starts filling at sodium (element 11) but does not finish filling till copper (element 29), and the N shell is even slower: it starts filling at potassium (element 19) but does not finish filling till ytterbium (element 70). The O, P, and Q shells begin filling in the known elements (respectively at rubidium, caesium, and francium), but they are not complete even at the heaviest known element, oganesson (element 118).
List of elements with electrons per shell
The list below gives the elements arranged by increasing atomic number and shows the number of electrons per shell. At a glance, the subsets of the list show obvious patterns. In particular, every set of five elements ( electric blue) before each noble gas (group 18, yellow) heavier than helium have successive numbers of electrons in the outermost shell, namely three to seven.
Sorting the table by chemical group shows additional patterns, especially with respect to the last two outermost shells. (Elements 57 to 71 belong to the lanthanides, while 89 to 103 are the actinides.)
The list below is primarily consistent with the Aufbau principle. However, there are a number of exceptions to the rule; for example palladium (atomic number 46) has no electrons in the fifth shell, unlike other atoms with lower atomic number. The elements past 108 have such short half-lives that their electron configurations have not yet been measured, and so predictions have been inserted instead.
| Z | Element | No. of electrons/shell | Group |
|---|---|---|---|
| 1 | Hydrogen | 1 | 1 |
| 2 | Helium | 2 | 18 |
| 3 | Lithium | 2, 1 | 1 |
| 4 | Beryllium | 2, 2 | 2 |
| 5 | Boron | 2, 3 | 13 |
| 6 | Carbon | 2, 4 | 14 |
| 7 | Nitrogen | 2, 5 | 15 |
| 8 | Oxygen | 2, 6 | 16 |
| 9 | Fluorine | 2, 7 | 17 |
| 10 | Neon | 2, 8 | 18 |
| 11 | Sodium | 2, 8, 1 | 1 |
| 12 | Magnesium | 2, 8, 2 | 2 |
| 13 | Aluminium | 2, 8, 3 | 13 |
| 14 | Silicon | 2, 8, 4 | 14 |
| 15 | Phosphorus | 2, 8, 5 | 15 |
| 16 | Sulfur | 2, 8, 6 | 16 |
| 17 | Chlorine | 2, 8, 7 | 17 |
| 18 | Argon | 2, 8, 8 | 18 |
| 19 | Potassium | 2, 8, 8, 1 | 1 |
| 20 | Calcium | 2, 8, 8, 2 | 2 |
| 21 | Scandium | 2, 8, 9, 2 | 3 |
| 22 | Titanium | 2, 8, 10, 2 | 4 |
| 23 | Vanadium | 2, 8, 11, 2 | 5 |
| 24 | Chromium | 2, 8, 13, 1 | 6 |
| 25 | Manganese | 2, 8, 13, 2 | 7 |
| 26 | Iron | 2, 8, 14, 2 | 8 |
| 27 | Cobalt | 2, 8, 15, 2 | 9 |
| 28 | Nickel | 2, 8, 16, 2 | 10 |
| 29 | Copper | 2, 8, 18, 1 | 11 |
| 30 | Zinc | 2, 8, 18, 2 | 12 |
| 31 | Gallium | 2, 8, 18, 3 | 13 |
| 32 | Germanium | 2, 8, 18, 4 | 14 |
| 33 | Arsenic | 2, 8, 18, 5 | 15 |
| 34 | Selenium | 2, 8, 18, 6 | 16 |
| 35 | Bromine | 2, 8, 18, 7 | 17 |
| 36 | Krypton | 2, 8, 18, 8 | 18 |
| 37 | Rubidium | 2, 8, 18, 8, 1 | 1 |
| 38 | Strontium | 2, 8, 18, 8, 2 | 2 |
| 39 | Yttrium | 2, 8, 18, 9, 2 | 3 |
| 40 | Zirconium | 2, 8, 18, 10, 2 | 4 |
| 41 | Niobium | 2, 8, 18, 12, 1 | 5 |
| 42 | Molybdenum | 2, 8, 18, 13, 1 | 6 |
| 43 | Technetium | 2, 8, 18, 13, 2 | 7 |
| 44 | Ruthenium | 2, 8, 18, 15, 1 | 8 |
| 45 | Rhodium | 2, 8, 18, 16, 1 | 9 |
| 46 | Palladium | 2, 8, 18, 18 | 10 |
| 47 | Silver | 2, 8, 18, 18, 1 | 11 |
| 48 | Cadmium | 2, 8, 18, 18, 2 | 12 |
| 49 | Indium | 2, 8, 18, 18, 3 | 13 |
| 50 | Tin | 2, 8, 18, 18, 4 | 14 |
| 51 | Antimony | 2, 8, 18, 18, 5 | 15 |
| 52 | Tellurium | 2, 8, 18, 18, 6 | 16 |
| 53 | Iodine | 2, 8, 18, 18, 7 | 17 |
| 54 | Xenon | 2, 8, 18, 18, 8 | 18 |
| 55 | Caesium | 2, 8, 18, 18, 8, 1 | 1 |
| 56 | Barium | 2, 8, 18, 18, 8, 2 | 2 |
| 57 | Lanthanum | 2, 8, 18, 18, 9, 2 | |
| 58 | Cerium | 2, 8, 18, 19, 9, 2 | |
| 59 | Praseodymium | 2, 8, 18, 21, 8, 2 | |
| 60 | Neodymium | 2, 8, 18, 22, 8, 2 | |
| 61 | Promethium | 2, 8, 18, 23, 8, 2 | |
| 62 | Samarium | 2, 8, 18, 24, 8, 2 | |
| 63 | Europium | 2, 8, 18, 25, 8, 2 | |
| 64 | Gadolinium | 2, 8, 18, 25, 9, 2 | |
| 65 | Terbium | 2, 8, 18, 27, 8, 2 | |
| 66 | Dysprosium | 2, 8, 18, 28, 8, 2 | |
| 67 | Holmium | 2, 8, 18, 29, 8, 2 | |
| 68 | Erbium | 2, 8, 18, 30, 8, 2 | |
| 69 | Thulium | 2, 8, 18, 31, 8, 2 | |
| 70 | Ytterbium | 2, 8, 18, 32, 8, 2 | |
| 71 | Lutetium | 2, 8, 18, 32, 9, 2 | 3 |
| 72 | Hafnium | 2, 8, 18, 32, 10, 2 | 4 |
| 73 | Tantalum | 2, 8, 18, 32, 11, 2 | 5 |
| 74 | Tungsten | 2, 8, 18, 32, 12, 2 | 6 |
| 75 | Rhenium | 2, 8, 18, 32, 13, 2 | 7 |
| 76 | Osmium | 2, 8, 18, 32, 14, 2 | 8 |
| 77 | Iridium | 2, 8, 18, 32, 15, 2 | 9 |
| 78 | Platinum | 2, 8, 18, 32, 17, 1 | 10 |
| 79 | Gold | 2, 8, 18, 32, 18, 1 | 11 |
| 80 | Mercury | 2, 8, 18, 32, 18, 2 | 12 |
| 81 | Thallium | 2, 8, 18, 32, 18, 3 | 13 |
| 82 | Lead | 2, 8, 18, 32, 18, 4 | 14 |
| 83 | Bismuth | 2, 8, 18, 32, 18, 5 | 15 |
| 84 | Polonium | 2, 8, 18, 32, 18, 6 | 16 |
| 85 | Astatine | 2, 8, 18, 32, 18, 7 | 17 |
| 86 | Radon | 2, 8, 18, 32, 18, 8 | 18 |
| 87 | Francium | 2, 8, 18, 32, 18, 8, 1 | 1 |
| 88 | Radium | 2, 8, 18, 32, 18, 8, 2 | 2 |
| 89 | Actinium | 2, 8, 18, 32, 18, 9, 2 | |
| 90 | Thorium | 2, 8, 18, 32, 18, 10, 2 | |
| 91 | Protactinium | 2, 8, 18, 32, 20, 9, 2 | |
| 92 | Uranium | 2, 8, 18, 32, 21, 9, 2 | |
| 93 | Neptunium | 2, 8, 18, 32, 22, 9, 2 | |
| 94 | Plutonium | 2, 8, 18, 32, 24, 8, 2 | |
| 95 | Americium | 2, 8, 18, 32, 25, 8, 2 | |
| 96 | Curium | 2, 8, 18, 32, 25, 9, 2 | |
| 97 | Berkelium | 2, 8, 18, 32, 27, 8, 2 | |
| 98 | Californium | 2, 8, 18, 32, 28, 8, 2 | |
| 99 | Einsteinium | 2, 8, 18, 32, 29, 8, 2 | |
| 100 | Fermium | 2, 8, 18, 32, 30, 8, 2 | |
| 101 | Mendelevium | 2, 8, 18, 32, 31, 8, 2 | |
| 102 | Nobelium | 2, 8, 18, 32, 32, 8, 2 | |
| 103 | Lawrencium | 2, 8, 18, 32, 32, 8, 3 | 3 |
| 104 | Rutherfordium | 2, 8, 18, 32, 32, 10, 2 | 4 |
| 105 | Dubnium | 2, 8, 18, 32, 32, 11, 2 | 5 |
| 106 | Seaborgium | 2, 8, 18, 32, 32, 12, 2 | 6 |
| 107 | Bohrium | 2, 8, 18, 32, 32, 13, 2 | 7 |
| 108 | Hassium | 2, 8, 18, 32, 32, 14, 2 | 8 |
| 109 | Meitnerium | 2, 8, 18, 32, 32, 15, 2 (?) | 9 |
| 110 | Darmstadtium | 2, 8, 18, 32, 32, 16, 2 (?) | 10 |
| 111 | Roentgenium | 2, 8, 18, 32, 32, 17, 2 (?) | 11 |
| 112 | Copernicium | 2, 8, 18, 32, 32, 18, 2 (?) | 12 |
| 113 | Nihonium | 2, 8, 18, 32, 32, 18, 3 (?) | 13 |
| 114 | Flerovium | 2, 8, 18, 32, 32, 18, 4 (?) | 14 |
| 115 | Moscovium | 2, 8, 18, 32, 32, 18, 5 (?) | 15 |
| 116 | Livermorium | 2, 8, 18, 32, 32, 18, 6 (?) | 16 |
| 117 | Tennessine | 2, 8, 18, 32, 32, 18, 7 (?) | 17 |
| 118 | Oganesson | 2, 8, 18, 32, 32, 18, 8 (?) | 18 |
See also
References
- ^ a b Re: Why do electron shells have set limits ? madsci.org, 17 March 1999, Dan Berger, Faculty Chemistry/Science, Bluffton College
- ^ a b Electron Subshells. Corrosion Source.
- ^ Bohr, N. (1913). "On the Constitution of Atoms and Molecules, Part II. Systems containing only a Single Nucleus". Philosophical Magazine. 26: 476–502.
- ^ a b c d e f g Kragh, Helge. "Niels Bohr’s Second Atomic Theory". Historical Studies in the Physical Sciences, vol. 10, University of California Press, 1979, pp. 123–86, https://doi.org/10.2307/27757389.
- ^ W. Kossel, "Über Molekülbildung als Folge des Atombaues", Ann. Phys., 1916, 49, 229-362 (237).
- ^ Translated in Helge Kragh, Aarhus, LARS VEGARD, ATOMIC STRUCTURE, AND THE PERIODIC SYSTEM, Bull. Hist. Chem., VOLUME 37, Number 1 (2012), p.43.
- ^ Donald Sadoway, Introduction to Solid State Chemistry, Lecture 5 Archived 29 June 2011 at the Wayback Machine
- ^ Bohr, Niels (1913). On the Constitution of Atoms and Molecules, Part I. _Philosophical Magazine_ 26:1--25.
- ^ Uhler, Horace Scudder. "On Moseley’s Law for X-Ray Spectra". Proceedings of the National Academy of Sciences of the United States of America, vol. 3, no. 2, National Academy of Sciences, 1917, pp. 88–90, http://www.jstor.org/stable/83748.
- ^ Niels Bohr interview 1962 Session III https://www.aip.org/history-programs/niels-bohr-library/oral-histories/4517-3
- ^ Kumar, Manjit. Quantum: Einstein, Bohr, and the great debate about the nature of reality / Manjit Kumar.—1st American ed., 2008. Chap.4.
- ^ Barkla, Charles G. (1911). "XXXIX.The spectra of the fluorescent Röntgen radiations". Philosophical Magazine. Series 6. 22 (129): 396–412. doi:10.1080/14786440908637137.
Previously denoted by letters B and A (...). The letters K and L are, however, preferable, as it is highly probable that series of radiations both more absorbable and more penetrating exist.
- ^ Michael Eckert, Disputed discovery: the beginnings of X-ray diffraction in crystals in 1912 and its repercussions, January 2011, Acta crystallographica. Section A, Foundations of crystallography 68(1):30-39 This Laue centennial article has also been published in Zeitschrift für Kristallographie [Eckert (2012). Z. Kristallogr. 227 , 27–35].
- ^ Charles G. Barkla M.A. D.Sc. (1911) XXXIX. The spectra of the fluorescent Röntgen radiations, The London, Edinburgh, and Dublin Philosophical Magazine and Journal of Science, 22:129, 396-412, DOI: 10.1080/14786440908637137
- ^ T.Hirosigeand S.Nisio,"Formation of Bohr's Theory of Atomic Constitution",Jap. Stud.Hist.Set.,No. 3(1964),6-28.
- ^ See Periodic Table for full history.
- ^ a b Niels Bohr Collected Works, Vol. 4, p. 740. Postcard from Arnold Sommerfeld to Bohr, 7 March 1921.
- ^ Pais, Abraham (1991), Niels Bohr’s Times, in Physics, Philosophy, and Polity (Oxford: Clarendon Press), quoted p. 205.
- ^ Schilpp, Paul A. (ed.) (1969), Albert Einstein: Philosopher-Scientist (New York: MJF Books). Collection first published in 1949 as Vol. VII in the series The Library of Living Philosophers by Open Court, La Salle, IL, Einstein, Albert 'Autobiographical Notes', pp.45-47.
- ^ Kumar, Manjit. Quantum: Einstein, Bohr, and the great debate about the nature of reality / Manjit Kumar.—1st American ed., 2008. Chap.7.
- ^ Bury, Charles R. (July 1921). "Langmuir's Theory of the Arrangement of Electrons in Atoms and Molecules". Journal of the American Chemical Society. 43 (7): 1602–1609. doi:10.1021/ja01440a023. ISSN 0002-7863.
- ^ The Genesis of the Bohr Atom, John L. Heilbron and Thomas S. Kuhn, Historical Studies in the Physical Sciences, Vol. 1 (1969), pp. vi, 211-290 (81 pages), University of California Press,p. 285-286.
- ^ Kumar, Manjit. Quantum: Einstein, Bohr, and the great debate about the nature of reality / Manjit Kumar.—1st American ed., 2008. Chap.5.
- ^ Jue, T. (2009). "Quantum Mechanic Basic to Biophysical Methods". Fundamental Concepts in Biophysics. Berlin: Springer. p. 33. ISBN 978-1-58829-973-4.
- ^ Orbitals. Chem4Kids. Retrieved on 1 December 2011.
- ^ Electron & Shell Configuration Archived 28 December 2018 at the Wayback Machine. Chemistry.patent-invent.com. Retrieved on 1 December 2011.

