Bisphosphonate

Bisphosphonates are a class of drugs that prevent the loss of bone density, used to treat osteoporosis and similar diseases. They are the most commonly prescribed drugs used to treat osteoporosis.[1] They are called bisphosphonates because they have two phosphonate (PO(OH)
2) groups. They are thus also called diphosphonates (bis- or di- + phosphonate).
Evidence shows that they reduce the risk of fracture in post-menopausal women with osteoporosis.[2][3][4][5][6]
Bone tissue undergoes constant remodeling and is kept in balance (homeostasis) by osteoblasts creating bone and osteoclasts destroying bone. Bisphosphonates inhibit the digestion of bone by encouraging osteoclasts to undergo apoptosis, or cell death, thereby slowing bone loss.[7]
The uses of bisphosphonates include the prevention and treatment of osteoporosis, Paget's disease of bone, bone metastasis (with or without hypercalcemia), multiple myeloma, primary hyperparathyroidism, osteogenesis imperfecta, fibrous dysplasia, and other conditions that exhibit bone fragility.
Medical uses[edit]
Bisphosphonates are used to treat osteoporosis, osteitis deformans (Paget's disease of the bone), bone metastasis (with or without hypercalcemia), multiple myeloma, and other conditions involving fragile, breakable bone.
In osteoporosis and Paget's, the most popular first-line bisphosphonate drugs are alendronate and risedronate. If these are ineffective or if the person develops digestive tract problems, intravenous pamidronate may be used. Strontium ranelate or teriparatide are used for refractory disease. The use of strontium ranelate is restricted because of increased risk of venous thromboembolism, pulmonary embolism and serious cardiovascular disorders, including myocardial infarction.[8] In postmenopausal women, the selective estrogen receptor modulator raloxifene is occasionally administered instead of bisphosphonates. Bisphosphonates are beneficial in reducing the risk of vertebral fracture in steroid induced osteoporosis.[9]
Post-menopausal osteoporosis[edit]
Bisphosphonates are recommended as a first line treatments for post-menopausal osteoporosis.[5][10][11][12]
Long-term treatment with bisphosphonates produces anti-fracture and bone mineral density effects that persist for 3–5 years after an initial 3–5 years of treatment.[2] The bisphosphonate alendronate reduces the risk of hip, vertebral, and wrist fractures by 35-39%; zoledronate reduces the risk of hip fractures by 38% and of vertebral fractures by 62%.[3][4] Risedronate has also been shown to reduce the risk of hip fractures.[5][6]
After five years of medications by mouth or three years of intravenous medication among those at low risk, bisphosphonate treatment can be stopped.[13] In those at higher risk ten years of medication by mouth or six years of intravenous treatment may be used.[13]
Cancer[edit]
Bisphosphonates reduce the risk of fracture and bone pain[14] in people with breast,[15] lung,[16] and other metastatic cancers as well as in people with multiple myeloma.[17] In breast cancer there is mixed evidence regarding whether bisphosphonates improve survival.[15][18][19][20] A 2017 Cochrane review found that for people with early breast cancer, bisphosphonate treatment may reduce the risk of the cancer spreading to the person's bone, however, for people who had advanced breast cancer bisphosphonate treatment did not appear to reduce the risk of the cancer spreading to the bone.[15] Side effects associated with bisphosphonate treatment for people with breast cancer are mild and rare.[15]
Bisphosphonates can also reduce mortality in those with multiple myeloma and prostate cancer.[20]
Other[edit]
Evidence suggests that the use of bisphosphonates would be useful in the treatment of complex regional pain syndrome, a neuro-immune problem with high MPQ scores, low treatment efficacy and symptoms which can include regional osteoporosis. In 2009 bisphosphonates were "among the only class of medications that has survived placebo-controlled studies showing statistically significant improvement (in CRPS) with therapy."[21]
Bisphosphonates have been used to reduce fracture rates in children with the disease osteogenesis imperfecta[22] and to treat otosclerosis[23] by minimizing bone loss.
Other bisphosphonates, including medronate (R1=H, R2=H) and oxidronate (R1=H, R2=OH), are mixed with radioactive technetium and injected, as a way to image bone and detect bone disease.
Adverse effects[edit]
Common[edit]
Oral bisphosphonates can cause upset stomach and inflammation and erosions of the esophagus, which is the main problem of oral N-containing[further explanation needed] preparations, that is ones containing "normal" unbranched chains. This can be prevented by remaining seated upright for 30 to 60 minutes after taking the medication. Intravenous bisphosphonates can give fever and flu-like symptoms after the first infusion, which is thought to occur because of their potential to activate human γδ T cells.
Bisphosphonates, when administered intravenously for the treatment of cancer, have been associated with osteonecrosis of the jaw (ONJ), with the mandible twice as frequently affected as the maxilla and most cases occurring following high-dose intravenous administration used for some cancer patients. Some 60% of cases are preceded by a dental surgical procedure (that involves the bone), and it has been suggested that bisphosphonate treatment should be postponed until after any dental work to eliminate potential sites of infection (the use of antibiotics may otherwise be indicated prior to any surgery).[24]
A number of cases of severe bone, joint, or musculoskeletal pain have been reported, prompting labeling changes.[25]
Some studies have identified bisphosphonate use as a risk factor for atrial fibrillation (AF), though meta-analysis of them finds conflicting reports. As of 2008[update], the US Food and Drug Administration did not recommend any alteration in prescribing of bisphosphonates based on AF concerns.[26] More recent meta-analyses have found strong correlations between bisphosphonate use and development of AF, especially when administered intravenously,[27] but that a significantly increased risk of AF that required hospitalization did not have an attendant increased risk of stroke or cardiovascular mortality.[28]
Long-term risks[edit]
In large studies, women taking bisphosphonates for osteoporosis have had unusual fractures ("bisphosphonate fractures") in the femur (thigh bone) in the shaft (diaphysis or sub-trochanteric region) of the bone, rather than at the femoral neck, which is the most common site of fracture. However, these fractures are rare (12 in 14,195 women) compared to the common hip fractures (272 in 14,195 women), and the overall reduction in hip fractures caused by bisphosphonate is more than the increase in unusual shaft fractures.[29][obsolete source] There are concerns that long-term bisphosphonate use can result in over-suppression of bone turnover. It is hypothesized that micro-cracks in the bone are unable to heal and eventually unite and propagate, resulting in atypical fractures. Such fractures tend to heal poorly and often require some form of bone stimulation, for example bone grafting as a secondary procedure. This complication is not common, and the benefit of overall fracture reduction still holds.[29][30][non-primary source needed] In cases where there is concern of such fractures occurring, teriparatide is potentially a good alternative because it does not cause as much damage as a bisphosphonate does by suppressing bone turnover.[31]
Three meta analyses have evaluated whether bisphosphonate use is associated with an increased risk of esophageal cancer. Two studies concluded that there was no evidence of increased risk.[32][33][34]
Chemistry and classes[edit]
All bisphosphonate drugs share a common phosphorus-carbon-phosphorus "backbone":
The two PO
3 (phosphonate) groups covalently linked to carbon determine both the name "bisphosphonate" and the function of the drugs. Bis refers to the fact that there are two such groups in the molecule.
The long side-chain (R2 in the diagram) determines the chemical properties, the mode of action and the strength of bisphosphonate drugs. The short side-chain (R1), often called the 'hook', mainly influences chemical properties and pharmacokinetics.
See nitrogenous and non-nitrogenous sections in Mechanism of action below.
Pharmacokinetics[edit]
Of the bisphosphonate that is resorbed (from oral preparation) or infused (for intravenous drugs), about 50% is excreted unchanged by the kidney. The remainder has a very high affinity for bone tissue, and is rapidly adsorbed onto the bone surface. Once bisphosphonates are in bone, they have a very long elimination half-life that can exceed ten years.[35]
Mechanism of action[edit]
Bisphosphonates are structurally similar to pyrophosphate, but with a central carbon that can have up to two substituents (R1 and R2) instead of an oxygen atom. Because a bisphosphonate group mimics the structure of pyrophosphate, it can inhibit activation of enzymes that utilize pyrophosphate.
The specificity of bisphosphonate-based drugs comes from the two phosphonate groups (and possibly a hydroxyl at R1) that work together to coordinate calcium ions. Bisphosphonate molecules preferentially bind to calcium ions. The largest store of calcium in the human body is in bones, so bisphosphonates accumulate to a high concentration only in bones.
Bisphosphonates, when attached to bone tissue, are released by osteoclasts, the bone cells that break down bone tissue. Bisphosphonate molecules then attach to and enter osteoclasts where they disrupt intracellular enzymatic functions needed for bone resorption.[36]
There are two classes of bisphosphonate compounds: non-nitrogenous (no nitrogen in R2) and nitrogenous (R2 contains nitrogen). The two types of bisphosphonates work differently in inhibiting osteoclasts.
| Class | Name | R1 | R2 | Relative potency (vs Etidronate=1) |
|---|---|---|---|---|
| Etidronate (Didronel) | OH | CH3 | 1 | |
| Clodronate (Bonefos, Loron) | Cl | Cl | 10 | |
| Tiludronate (Skelid) | H | p-Chlorophenylthio | 10 | |
| Pamidronate (APD, Aredia) | OH | (CH2)2NH2 | 100 | |
| Neridronate (Nerixia[a]) | OH | (CH2)5NH2 | 100 | |
| Olpadronate | OH | (CH2)2N(CH3)2 | 500 | |
| Alendronate (Fosamax) | OH | (CH2)3NH2 | 500 | |
| Ibandronate (Boniva - US, Bonviva - Asia) | OH | (CH2)2N(CH3)(CH2)4CH3 | 1000 | |
| Risedronate (Actonel) | OH | 3-Pyridylmethyl | 2000 | |
| Zoledronate (Zometa, Aclasta) | OH | 1H-imidazol-1-ylmethyl | 10000 |
Non-nitrogenous[edit]
The non-nitrogenous bisphosphonates (diphosphonates) are metabolised in the cell to compounds that replace the terminal pyrophosphate moiety of ATP, forming a non-functional molecule that competes with adenosine triphosphate (ATP) in the cellular energy metabolism. The osteoclast initiates apoptosis and dies, leading to an overall decrease in the breakdown of bone. This type of bisphosphonate has overall more negative effects than the nitrogen containing group, and is prescribed far less often.[37]
Nitrogenous[edit]
Nitrogenous bisphosphonates act on bone metabolism by binding and blocking the enzyme farnesyl diphosphate synthase (FPPS) in the HMG-CoA reductase pathway (also known as the mevalonate pathway).[38]
Bisphosphonates that contain isoprene chains at the R1 or R2 position can impart specificity for inhibition of GGPS1.[39]
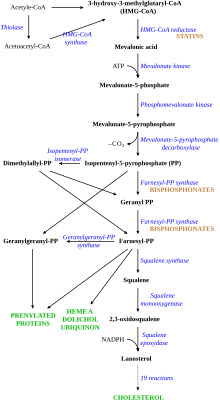
Disruption of the HMG CoA-reductase pathway at the level of FPPS prevents the formation of two metabolites (farnesol and geranylgeraniol) that are essential for connecting some small proteins to the cell membrane. This phenomenon is known as prenylation, and is important for proper sub-cellular protein trafficking (see "lipid-anchored protein" for the principles of this phenomenon).[40]
While inhibition of protein prenylation may affect many proteins found in an osteoclast, disruption to the lipid modification of Ras, Rho, Rac proteins has been speculated to underlie the effects of bisphosphonates. These proteins can affect both osteoclastogenesis, cell survival, and cytoskeletal dynamics. In particular, the cytoskeleton is vital for maintaining the "ruffled border" that is required for contact between a resorbing osteoclast and a bone surface.
Statins are another class of drugs that inhibit the HMG-CoA reductase pathway. Unlike bisphosphonates, statins do not bind to bone surfaces with high affinity, and thus are not specific for bone. Nevertheless, some studies have reported a decreased rate of fracture (an indicator of osteoporosis) and/or an increased bone mineral density in statin users. The overall efficacy of statins in the treatment of osteoporosis remains controversial.[41]
History[edit]
Bisphosphonates were developed in the 19th century but were first investigated in the 1960s for use in disorders of bone metabolism. Their non-medical use was to soften water in irrigation systems used in orange groves. The initial rationale for their use in humans was their potential in preventing the dissolution of hydroxylapatite, the principal bone mineral, thus arresting bone loss. In the 1990s their actual mechanism of action was demonstrated with the initial launch of alendronate by Merck & Co.[42]
Notes[edit]
References[edit]
- ^ National Osteoporosis Society. "Drug Treatment". U.K. National Osteoporosis Society. Archived from the original on 6 November 2012. Retrieved 7 August 2012.
- ^ a b Eriksen EF, Díez-Pérez A, Boonen S (January 2014). "Update on long-term treatment with bisphosphonates for postmenopausal osteoporosis: a systematic review". Bone. 58: 126–35. doi:10.1016/j.bone.2013.09.023. PMID 24120384.
- ^ a b Serrano AJ, Begoña L, Anitua E, Cobos R, Orive G (December 2013). "Systematic review and meta-analysis of the efficacy and safety of alendronate and zoledronate for the treatment of postmenopausal osteoporosis". Gynecol. Endocrinol. 29 (12): 1005–14. doi:10.3109/09513590.2013.813468. PMID 24063695. S2CID 20163452.
- ^ a b Gauthier, K; Bai, A; Perras, C; Cunningham, J; Ahuja, T; Richter, T; Kovacs, C (February 2012). "Denosumab, Raloxifene, and Zoledronic Acid for the Treatment of Postmenopausal Osteoporosis: Clinical Effectiveness and Harms". Rapid Response Report: Systematic Review. PMID 24278999.
- ^ a b c Watts NB, Bilezikian JP, Camacho PM, Greenspan SL, Harris ST, Hodgson SF, Kleerekoper M, Luckey MM, McClung MR, Pollack RP, Petak SM (2010). "American Association of Clinical Endocrinologists Medical Guidelines for Clinical Practice for the diagnosis and treatment of postmenopausal osteoporosis". Endocr Pract. 16 (Suppl 3): 1–37. doi:10.4158/ep.16.s3.1. PMC 4876714. PMID 21224201.
- ^ a b Kanis JA, McCloskey EV, Johansson H, Cooper C, Rizzoli R, Reginster JY (January 2013). "European guidance for the diagnosis and management of osteoporosis in postmenopausal women". Osteoporos Int. 24 (1): 23–57. doi:10.1007/s00198-012-2074-y. PMC 3587294. PMID 23079689.
- ^ Weinstein RS, Roberson PK, Manolagas SC (January 2009). "Giant osteoclast formation and long-term oral bisphosphonate therapy". N. Engl. J. Med. 360 (1): 53–62. doi:10.1056/NEJMoa0802633. PMC 2866022. PMID 19118304.
- ^ "Strontium ranelate: cardiovascular risk—restricted indication and new monitoring requirements Article date: March 2014". MHRA.
- ^ Allen, CS; Yeung, JH; Vandermeer, B; Homik, J (5 October 2016). "Bisphosphonates for steroid-induced osteoporosis". The Cochrane Database of Systematic Reviews. 2016 (10): CD001347. doi:10.1002/14651858.CD001347.pub2. PMC 6461188. PMID 27706804.
- ^ "Management of osteoporosis in postmenopausal women: 2010 position statement of The North American Menopause Society". Menopause. 17 (1): 25–54, quiz 55–6. 2010. doi:10.1097/gme.0b013e3181c617e6. PMID 20061894. S2CID 7980731.
- ^ Hauk L (August 2013). "ACOG releases practice bulletin on osteoporosis". Am Fam Physician. 88 (4): 269–75. PMID 23944732.
- ^ Compston J, Bowring C, Cooper A, et al. (August 2013). "Diagnosis and management of osteoporosis in postmenopausal women and older men in the UK: National Osteoporosis Guideline Group (NOGG) update 2013". Maturitas. 75 (4): 392–6. doi:10.1016/j.maturitas.2013.05.013. PMID 23810490.
- ^ a b Adler, Robert A; El-Hajj Fuleihan, Ghada; Bauer, Douglas C; Camacho, Pauline M; Clarke, Bart L; Clines, Gregory A; Compston, Juliet E; Drake, Matthew T; Edwards, Beatrice J; Favus, Murray J; Greenspan, Susan L; McKinney, Ross; Pignolo, Robert J; Sellmeyer, Deborah E (January 2016). "Managing Osteoporosis in Patients on Long-Term Bisphosphonate Treatment: Report of a Task Force of the American Society for Bone and Mineral Research". Journal of Bone and Mineral Research. 31 (1): 16–35. doi:10.1002/jbmr.2708. PMC 4906542. PMID 26350171.
- ^ Zhu M, Liang R, Pan LH, et al. (February 2013). "Zoledronate for metastatic bone disease and pain: a meta-analysis of randomized clinical trials". Pain Med. 14 (2): 257–64. doi:10.1111/pme.12016. PMID 23279447.
- ^ a b c d O'Carrigan, Brent; Wong, Matthew Hf; Willson, Melina L.; Stockler, Martin R.; Pavlakis, Nick; Goodwin, Annabel (2017). "Bisphosphonates and other bone agents for breast cancer". The Cochrane Database of Systematic Reviews. 10 (11): CD003474. doi:10.1002/14651858.CD003474.pub4. ISSN 1469-493X. PMC 6485886. PMID 29082518.
- ^ Lopez-Olivo MA, Shah NA, Pratt G, Risser JM, Symanski E, Suarez-Almazor ME (November 2012). "Bisphosphonates in the treatment of patients with lung cancer and metastatic bone disease: a systematic review and meta-analysis". Support Care Cancer. 20 (11): 2985–98. doi:10.1007/s00520-012-1563-z. PMC 3691019. PMID 22956190.
- ^ Mhaskar R, Kumar A, Miladinovic B, Djulbegovic B (2017). "Bisphosphonates in multiple myeloma: a network meta-analysis". Cochrane Database Syst Rev. 2017 (12): CD003188. doi:10.1002/14651858.CD003188.pub4. PMC 6486151. PMID 29253322.
- ^ Ben-Aharon I, Vidal L, Rizel S, et al. (2013). "Bisphosphonates in the adjuvant setting of breast cancer therapy--effect on survival: a systematic review and meta-analysis". PLOS ONE. 8 (8): e70044. Bibcode:2013PLoSO...870044B. doi:10.1371/journal.pone.0070044. PMC 3753308. PMID 23990894.
- ^ Zhu J, Zheng Y, Zhou Z (June 2013). "Oral adjuvant clodronate therapy could improve overall survival in early breast cancer: results from an updated systematic review and meta-analysis". Eur. J. Cancer. 49 (9): 2086–92. doi:10.1016/j.ejca.2013.01.021. PMID 23452992.
- ^ a b Van Acker, HH; Anguille, S; Willemen, Y; Smits, EL; Van Tendeloo, VF (23 November 2015). "Bisphosphonates for cancer treatment: Mechanisms of action and lessons from clinical trials". Pharmacology & Therapeutics. 158: 24–40. doi:10.1016/j.pharmthera.2015.11.008. PMID 26617219.
- ^ Pontell D (July 2008). "A clinical approach to complex regional pain syndrome". Clinics in Podiatric Medicine and Surgery. 25 (3): 361–80, vi. doi:10.1016/j.cpm.2008.02.011. PMID 18486850.
- ^ Shapiro JR, Sponsellor PD (December 2009). "Osteogenesis imperfecta: questions and answers". Current Opinion in Pediatrics. 21 (6): 709–16. doi:10.1097/MOP.0b013e328332c68f. PMID 19907330. S2CID 205834462.
- ^ Brookler K (2008). "Medical treatment of otosclerosis: rationale for use of bisphosphonates". Int Tinnitus J. 14 (2): 92–6. PMID 19205157.
- ^ Woo S, Hellstein J, Kalmar J (2006). "Narrative [corrected] review: bisphosphonates and osteonecrosis of the jaws". Ann Intern Med. 144 (10): 753–61. doi:10.7326/0003-4819-144-10-200605160-00009. PMID 16702591. S2CID 53091343.
- ^ Wysowski D, Chang J (2005). "Alendronate and risedronate: reports of severe bone, joint, and muscle pain". Arch Intern Med. 165 (3): 346–7. doi:10.1001/archinte.165.3.346-b. PMID 15710802.
- ^ "Update of Safety Review Follow-up to the October 1, 2007 Early Communication about the Ongoing Safety Review of Bisphosphonates". Postmarket Drug Safety Information for Patients and Providers. Food and Drug Administration (United States). October 2008. Retrieved 2009-07-15.
- ^ Sharma A, Einstein AJ, Vallakati A, Arbab-Zadeh A, Walker MD, Mukherjee D, Homel P, Borer JS, Lichstein E (June 1, 2014). "Risk of Atrial Fibrillation With Use of Oral and Intravenous Bisphosphonates". American Journal of Cardiology. 113 (11): 1815–1821. doi:10.1016/j.amjcard.2014.03.008. PMID 24837258.
- ^ Sharma A, Chatterjee S, Arbab-Zadeh A, Goyal S, Lichstein E, Ghosh J, Aikat S (October 2013). "Risk of serious atrial fibrillation and stroke with use of bisphosphonates: evidence from a meta-analysis". Chest. 144 (4): 1131–1322. doi:10.1378/chest.13-0675. PMID 23722644.
- ^ a b Shane E (May 2010). "Evolving data about subtrochanteric fractures and bisphosphonates". N. Engl. J. Med. 362 (19): 1825–7. doi:10.1056/NEJMe1003064. PMID 20335574.
- ^ Lenart BA, Lorich DG, Lane JM (March 2008). "Atypical fractures of the femoral diaphysis in postmenopausal women taking alendronate". N. Engl. J. Med. 358 (12): 1304–6. doi:10.1056/NEJMc0707493. PMID 18354114.
- ^ Arkan S Sayed-Noor; Bakir K Kadum; Göran O Sjödén (31 March 2010). "Bisphosphonate-induced femoral fragility fractures: What do we know?". Orthopedic Research and Reviews. 2 (1): 27–34. doi:10.2147/orr.s7521.
- ^ Andrici J, Tio M, Eslick GD (October 2012). "Meta-analysis: oral bisphosphonates and the risk of oesophageal cancer". Aliment. Pharmacol. Ther. 36 (8): 708–16. doi:10.1111/apt.12041. PMID 22966908. S2CID 5271005.
- ^ Sun K, Liu JM, Sun HX, Lu N, Ning G (January 2013). "Bisphosphonate treatment and risk of esophageal cancer: a meta-analysis of observational studies". Osteoporos Int. 24 (1): 279–86. doi:10.1007/s00198-012-2158-8. PMID 23052941. S2CID 12625687.
- ^ Oh YH, Yoon C, Park SM (October 2012). "Bisphosphonate use and gastrointestinal tract cancer risk: meta-analysis of observational studies". World J. Gastroenterol. 18 (40): 5779–88. doi:10.3748/wjg.v18.i40.5779. PMC 3484348. PMID 23155320.
- ^ "Bisphosphonates". courses.washington.edu. Retrieved 2018-09-19.
- ^ Coxon FP, Thompson K, Roelofs AJ, Ebetino FH, Rogers MJ (May 2008). "Visualizing mineral binding and uptake of bisphosphonate by osteoclasts and non-resorbing cells". Bone. 42 (5): 848–860. doi:10.1016/j.bone.2007.12.225. PMID 18325866.
- ^ Frith J, Mönkkönen J, Blackburn G, Russell R, Rogers M (1997). "Clodronate and liposome-encapsulated clodronate are metabolized to a toxic ATP analog, adenosine 5'-(beta, gamma-dichloromethylene) triphosphate, by mammalian cells in vitro". J Bone Miner Res. 12 (9): 1358–67. doi:10.1359/jbmr.1997.12.9.1358. PMID 9286751.
- ^ van Beek E, Cohen L, Leroy I, Ebetino F, Löwik C, Papapoulos S (November 2003). "Differentiating the mechanisms of antiresorptive action of nitrogen containing bisphosphonates". Bone. 33 (5): 805–11. doi:10.1016/j.bone.2003.07.007. PMID 14623056.
- ^ Wiemer, AJ; Wiemer, DF; Hohl, RJ (December 2011). "Geranylgeranyl diphosphate synthase: an emerging therapeutic target". Clinical Pharmacology and Therapeutics. 90 (6): 804–12. doi:10.1038/clpt.2011.215. PMID 22048229. S2CID 27913789.
- ^ Van Beek E, Löwik C, van der Pluijm G, Papapoulos S (1999). "The role of geranylgeranylation in bone resorption and its suppression by bisphosphonates in fetal bone explants in vitro: A clue to the mechanism of action of nitrogen-containing bisphosphonates". J Bone Miner Res. 14 (5): 722–9. doi:10.1359/jbmr.1999.14.5.722. PMID 10320520. S2CID 20316713.
- ^ Uzzan, B; et al. (2007). "Effects of statins on bone mineral density: a meta-analysis of clinical studies". Bone. 40 (6): 1581–7. doi:10.1016/j.bone.2007.02.019. PMID 17409043.
- ^ Fleisch H (2002). "Development of bisphosphonates". Breast Cancer Res. 4 (1): 30–4. doi:10.1186/bcr414. PMC 138713. PMID 11879557.
