Athetosis
| Athetosis | |
|---|---|
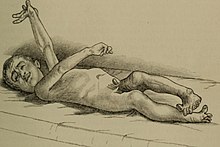 | |
| Bilateral athetosis | |
| Specialty | Neurology |
Athetosis is a symptom characterized by slow, involuntary, convoluted, writhing movements of the fingers, hands, toes, and feet and in some cases, arms, legs, neck and tongue.[1] Movements typical of athetosis are sometimes called athetoid movements. Lesions to the brain are most often the direct cause of the symptoms, particularly to the corpus striatum.[2] This symptom does not occur alone and is often accompanied by the symptoms of cerebral palsy, as it is often a result of this physical disability. Treatments for athetosis are not very effective, and in most cases are simply aimed at managing the uncontrollable movement, rather than the cause itself.[citation needed]
Signs and symptoms
[edit]Athetosis can vary from mild to severe motor dysfunction; it is generally characterized by unbalanced, involuntary movements of muscle and a difficulty maintaining a symmetrical posture. The associated motor dysfunction can be restricted to a part of the body or present throughout the body, depending on the individual and the severity of the symptom. One of the pronounced signs can be observed in the extremities in particular, as the writhing, convoluted movement of the digits.[1] Athetosis can appear as early as 18 months from birth with first signs including difficulty feeding, hypotonia, spasm, and involuntary writhing movements of the hands, feet, and face, which progressively worsen through adolescence and at times of emotional distress.[3] Athetosis is caused by lesions in several brain areas such as the hippocampus and the motor thalamus, as well as the corpus striatum;[2] therefore children during the developmental age could possibly suffer from severe communication deficits such as speech impairment, hearing loss, and failed or delayed acquirement of sitting balance, although most people with athetosis have normal or near-normal intelligence.[3]
Causes
[edit]Athetosis is a symptom primarily caused by the marbling, or degeneration of the basal ganglia.[citation needed] This degeneration is most commonly caused by complications at birth or by Huntington's disease, in addition to rare cases in which the damage may also arise later in life due to stroke or trauma.[citation needed] The two complications of particular interest are intranatal asphyxia and neonatal jaundice.
Asphyxia
[edit]Asphyxia directly causes basal ganglia damage due to lack of oxygen and therefore, insufficient nutrient supply.[citation needed] The lesions caused by asphyxia are most prominent on the caudate nucleus and the putamen.[4] However, a less-studied consequence of the resulting hypoxia is its effect on the concentrations of the neurotransmitter dopamine within the synapses of neurons in the basal ganglia. Hypoxia leads to an increase in the extracellular dopamine levels and therefore, an increase in the activity of the dopaminergic neurons. Furthermore, this increase in extracellular concentration is not caused by an increase in the neurotransmitter synthesis, but instead on inhibiting its reuptake back into the neurons and glial cells.[5] Therefore, there is an increased dopaminergic effect as dopamine remains in the synapse at higher concentrations leading to additional post-synaptic response. As a result, the uncontrollable writhing motions witnessed with athetosis deal with the over-activity of synapses within the basal ganglia.[citation needed]
Neonatal jaundice
[edit]Neonatal jaundice is the other chief complication that leads to the basal ganglia damage associated with this condition. Jaundice is caused by hyperbilirubinemia, or abnormally high levels of bilirubin in the blood. Bilirubin is usually bound to albumin immediately and sent to the liver. However, in neonatal jaundice, the concentration of bilirubin overwhelms that of albumin and some of the bilirubin remains unconjugated and can enter the brain through the blood–brain barrier.[6] Normally bilirubin would not be able to diffuse across the blood–brain barrier, but in infants, the barrier is immature and has higher permeability. Bilirubin is toxic as it prevents the phosphorylation of many proteins, including synapsin I which binds vesicles in the presynaptic terminal.[7] Therefore, it directly inhibits the exocytosis of neurotransmitters and severely hinders the synapses it affects. In autopsies of children who suffered from neonatal jaundice, chronic changes of neuronal loss, gliosis and demyelination were observed in the basal ganglia and more specifically within the globus pallidus.[6]
Thalamic stroke
[edit]Another study was done where the onset of athetoid movement followed a thalamic stroke.[citation needed] The thalamus is part of a pathway that is involved with the cortical feedback loop in which signals from the cortex are relayed through the striatum, pallidus and thalamus before making it back to the cortex.[8] The striatum receives excitatory inputs from the cortex and inhibits the pallidum. By doing so it frees the thalamus from pallidal inhibition allowing the thalamus to send excitatory outputs to the cortex. Therefore, the lesions to the thalamus or any other part of this feedback loop can result in movement disorders as they can alter the reactivity of one towards the other.[8] Also, in a case of people with thalamic stroke, a majority suffered severe sensory deficits and ataxia. It is proposed that this loss of proprioception and the ensuing loss of synergic stabilization may also lead to abnormal movements, such as those dealt with in athetosis.[8]
Fahr's syndrome
[edit]Treatments
[edit]There are several different treatment approaches to dealing with athetosis. The most common methods are the use of drugs, surgical intervention, and retraining movements of the afflicted person. It is suggested that training a person to relearn movements can be helpful in select situations. Though, generally, this type of treatment will not work, in certain cases it can be found to be very helpful in treating the symptom of athetosis.[9]
Drugs can also be used in the treatment of athetosis, however their collective effectiveness is not very convincing.[10] There is not a single drug that is a standard among treatment. Many different medicines can be used, including:
- trihexyphenidyl
- benzatropine[11]
- Curare,[9] though not practical due to respiratory paralysis
- Tetrabenazine
- Haloperidol
- Thiopropazate
- Diazepam
Most instances of drug use where the symptoms seem to be lessened tend to be in more mild cases of athetosis.[10]
Treatment by surgical intervention can obviously have the most immediate impact, again however, it is not a cure-all. In patients that have cerebral palsy as the cause of their athetosis, it has been demonstrated that a subthalamotomy tends to help relieve the extent of athetosis in approximately half of patients. Additionally, late 19th and early 20th century surgical accounts state that athetosis can be relieved by the removal of a part of the cerebral motor cortex or by cutting a part of the posterior spinal roots.[12] Patients who undergo surgical treatment to relieve the athetosis often see significant improvement in the control of their limbs and digits.[9] While surgery is often very beneficial in the short term and can produce near immediate results, in the long term it has been seen that its effects are not incredibly long lasting.[4]
Related disorders
[edit]Choreoathetosis
[edit]Chorea is another condition which results from damage to the basal ganglia. Similar to athetosis, it results from mutations affecting the pallidum inhibition of the thalamus as well as increased dopaminergic activity at the level of the striatum.[13] Considering the etiology of both disorders are fairly similar, it comes as no surprise that chorea and athetosis can and usually do occur together in a condition called choreoathetosis.
Cerebral palsy
[edit]Athetosis is a commonly occurring symptom in the disease cerebral palsy.[14] Of all people with the disease, between 16%[15] and 25%[4] of them actually exhibit the symptom of athetosis. A component of this is the finding that most often the symptoms that involve athetosis occur as a part of choreoathetosis as opposed to athetosis alone.[16]
It is also noteworthy that the presence of athetosis in cerebral palsy (as well as other conditions) causes a significant increase in a person's basal resting metabolic rate. It has been observed that those who have cerebral palsy with athetosis require approximately 500 more Calories per day than their non-cerebral palsy non-athetoid counterpart.[15]
Pseudoathetosis
[edit]Pseudoathetosis is a movement disorder, very similar to athetosis, in which the symptoms are not differentiable from those of actual athetosis, however the underlying cause is different. While actual athetosis is caused by damage to the brain, specifically in the basal ganglia,[4] pseudoathetosis is caused by the loss of proprioception.[17] The loss in proprioception is caused by damage to the area between the primary somatosensory cortex and the muscle spindles and joint receptors. Additionally, when observing an MRI, it can be seen that in the brain of a pseudoathetoid patient, lesions on the brain are not seen in the basal ganglia,[18] the area that is oftentimes the cause of athetosis.[4]
Social implications
[edit]Athetosis is characterized as a symptom that is present in many forms of central nervous system disorders that affect the body movement coordination, such as cerebral palsy. Children may struggle to engage in social communication, since the poor coordination of the tongue and mouth muscles can reduce their speech ability and hinder their social interaction to a greater degree.[19] The caregivers of the affected children are encouraged to closely monitor their nutrition and growth and to provide them with hearing aids in order to relieve their symptoms as well as support their academic plans.[20] A growing number of patients is shown to benefit from communication devices such as shorthand typing programs and computer speech devices, as well as simple picture boards.[19]
Patients living with the disorder into their adulthood often have trouble being involved in daily activities such as eating, walking, dressing, as well as performing everyday tasks. They are consistently faced with challenges that limit their ability to live on their own. They are more reluctant to be involved in social activities and romantic relationships and more likely to develop poor self-esteem and self-image related to their physical limitations as well as cognitive disabilities, though such habitual thinking is shown to decline when they feel they are accepted and supported by their peers.[21] Patients are also inclined to associate themselves with people who tend not to be engaged in physical activities, according to the September 2008 issue of "Journal of Physical Activity and Health".[22]
History
[edit]The first noted case of athetosis was discovered by W. A. Hammond and described in his book Diseases of the Nervous System in 1871.[9] Hammond was also the person who created the term "athetosis", Greek for "without position".[23] In his initial description of athetosis, the extent of the uncontrolled movement was limited to the fingers and toes. In association with this, he noted that the patients' calves and forearms were oftentimes flexed and that movements were generally slow. Over the period of time leading into the late 20th century, the definition of athetosis was expanded to include movements of the neck, tongue, face, and even the trunk. Along with the expansion of the symptoms came the recognition that it was a part of many medical conditions, including cerebral palsy and stroke.[23]
Research directions
[edit]As athetosis is relatively difficult to treat, efforts are being made to help those with the condition live and perform tasks more effectively and more efficiently. One such example of work that has been recently undertaken is a project to help those affected with athetosis to use a computer with more ease. Software for the control of the computer uses joysticks that perform linear filtering to aid in control.[24]
An additional possible treatment option for those afflicted with the symptom is neurostimulation. Studies have begun, and in cerebral palsy patients affected with dystonia-choreoathetosis, it has been demonstrated that neurostimulation has been an effective treatment in lessening symptoms in patients. There has not been a tremendous amount of experimentation, though, in this as a possible treatment option.[25]
See also
[edit]References
[edit]- ^ a b Walker, Kenneth H (1990). "Ch. 70 Involuntary Movements". Clinical Methods: The History, Physical, and Laboratory (3rd ed.). PMID 21250235.
- ^ a b "Athetosis". Health Database - Medical Ailments & Diseases. Retrieved March 22, 2011.
- ^ a b McNeil, Malcolm R. (2009). Clinical Management of Sensorimotor Speech Disorders. Thieme. ISBN 978-1-58890-514-7.
- ^ a b c d e Foley J (April 1983). "The athetoid syndrome. A review of a personal series". J. Neurol. Neurosurg. Psychiatry. 46 (4): 289–98. doi:10.1136/jnnp.46.4.289. PMC 1027350. PMID 6341510.
- ^ Akiyama Y, Koshimura K, Ohue T, Lee K, Miwa S, Yamagata, S (September 1991). "Effects of hypoxia on the activity of the dopaminergic neuron system in the rat striatum as studied by in vivo brain microdialysis". Journal of Neurochemistry. 57 (3): 997–1002. doi:10.1111/j.1471-4159.1991.tb08249.x. PMID 1861163. S2CID 42453096.
- ^ a b Martich-Kriss, V; Kollias, SS; Ball WS, Jr (April 1995). "MR findings in kernicterus". American Journal of Neuroradiology. 16 (4 Suppl): 819–21. PMC 8332263. PMID 7611048.
- ^ Hansen, TW (December 2001). "Bilirubin brain toxicity". Journal of Perinatology. 21 Suppl 1: S48–51, discussion S59–62. doi:10.1038/sj.jp.7210634. PMID 11803417.
- ^ a b c Handley, A; Medcalf, P; Hellier, K; Dutta, D (May 2009). "Movement disorders after stroke". Age and Ageing. 38 (3): 260–6. doi:10.1093/ageing/afp020. PMID 19276093.
- ^ a b c d Putnam T (May 1939). "Athetosis". Yale J. Biol. Med. 11 (5): 459–465. PMC 2602263. PMID 21433835.
- ^ a b Mawdsley C (December 1975). "Diseases of the central nervous system. Involuntary movements". Br. Med. J. 4 (5996): 572–4. doi:10.1136/bmj.4.5996.572. PMC 1675885. PMID 1203674.
- ^ Polzin, Scott J., MS, and Teresa G. Odle. "Cerebral palsy." Gale Encyclopedia of Medicine. Thomson Gale, 2006. NA. Health Reference Center Academic. Gale.
- ^ Laitinen LV (August 1970). "Neurosurgery in cerebral palsy". J. Neurol. Neurosurg. Psychiatry. 33 (4): 513–8. doi:10.1136/jnnp.33.4.513. PMC 493511. PMID 4918461.
- ^ Bhidayasiri, R; Truong, DD (September 2004). "Chorea and related disorders". Postgraduate Medical Journal. 80 (947): 527–34. doi:10.1136/pgmj.2004.019356. PMC 1743104. PMID 15356354.
- ^ Rodriguez SP, Ding D, Riviere SP (September 2010). "Algorithms for target prediction for computer users with athetosis". 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology. Vol. 2010. pp. 491–494. doi:10.1109/iembs.2010.5627118. ISBN 978-1-4244-4123-5. PMC 3253859. PMID 21096307.
- ^ a b Johnson RK, Goran MI, Ferrara MS, Poehlman ET (February 1996). "Athetosis increases resting metabolic rate in adults with cerebral palsy". J. Am. Diet. Assoc. 96 (2): 145–8. doi:10.1016/S0002-8223(96)00043-0. PMID 8557940.
- ^ O'Shea TM (December 2008). "Diagnosis, treatment, and prevention of cerebral palsy". Clin. Obstet. Gynecol. 51 (4): 816–28. doi:10.1097/GRF.0b013e3181870ba7. PMC 3051278. PMID 18981805.
- ^ Lo YL, See S (November 2010). "Images in clinical medicine. Pseudoathetosis". N. Engl. J. Med. 363 (19): e29. doi:10.1056/NEJMicm0907786. PMID 21047218.
- ^ Salih F, Zimmer C, Meierkord H (March 2007). "Parietal proprioceptive loss with pseudoathetosis". J. Neurol. 254 (3): 396–7. doi:10.1007/s00415-006-0382-x. PMID 17345036. S2CID 32393176.
- ^ a b Jeff Brody (2005). "Social Problems with Cerebral Palsy". Cerebral Palsy Lawyer - Birth Injury Attorney. Cerebral Palsy Source. Retrieved March 21, 2011.
- ^ T. Michael O’Shea (December 2009). "Diagnosis, Treatment, and Prevention of Cerebral Palsy in Near-Term/Term Infants". Clinical Obstetrics and Gynecology. 51 (4): 816–28. doi:10.1097/GRF.0b013e3181870ba7. PMC 3051278. PMID 18981805.
- ^ Lehrman, Mary (10 May 2010). "The Social, Emotional, & Psychological Effects Of Adults With Cerebral Palsy". Demand Media, Inc. Retrieved March 22, 2011.
- ^ Gaskin, Cadeyrn; Morris, Tony (September 2008). "Physical Activity, Health-Related Quality of Life, and Psychosocial Functioning of Adults With Cerebral Palsy". Journal of Physical Activity and Health. 5 (1): 146–157. doi:10.1123/jpah.5.1.146. PMID 18209260.
- ^ a b Morris JG, Jankelowitz SK, Fung VS, Clouston PD, Hayes MW, Grattan-Smith P (November 2002). "Athetosis I: historical considerations". Mov. Disord. 17 (6): 1278–80. doi:10.1002/mds.10267. PMID 12465068. S2CID 13359301.
- ^ Lopez JV, Sibenaller S, Ding D, Riviere CN (2007). "Toward filtering of athetoid movement with neural networks". Engineering in Medicine and Biology Society. 29th Annual International Conference of the IEEE. doi:10.1109/iembs.2007.4352569.
- ^ Vidailhet M, Yelnik J, Lagrange C, et al. (August 2009). "Bilateral pallidal deep brain stimulation for the treatment of patients with dystonia-choreoathetosis cerebral palsy: a prospective pilot study". Lancet Neurol. 8 (8): 709–17. doi:10.1016/S1474-4422(09)70151-6. PMID 19576854. S2CID 24345609.
