Single-layer materials
In materials science, the term single-layer materials or 2D materials refers to crystalline solids consisting of a single layer of atoms. These materials are promising for some applications but remain the focus of research. Single-layer materials derived from single elements generally carry the -ene suffix in their names, e.g. graphene. Single-layer materials that are compounds of two or more elements have -ane or -ide suffixes. 2D materials can generally be categorized as either 2D allotropes of various elements or as compounds (consisting of two or more covalently bonding elements).
It is predicted that there are hundreds of stable single-layer materials.[1][2] The atomic structure and calculated basic properties of these and many other potentially synthesisable single-layer materials, can be found in computational databases.[3] 2D materials can be produced using mainly two approaches: top-down exfoliation and bottom-up synthesis. The exfoliation methods include sonication, mechanical, hydrothermal, electrochemical, laser-assisted, and microwave-assisted exfoliation.[4]
Single element materials
[edit]C: graphene and graphyne
[edit]- Graphene

Graphene is a crystalline allotrope of carbon in the form of a nearly transparent (to visible light) one atom thick sheet. It is hundreds of times stronger than most steels by weight.[5] It has the highest known thermal and electrical conductivity, displaying current densities 1,000,000 times that of copper.[6] It was first produced in 2004.[7]
Andre Geim and Konstantin Novoselov won the 2010 Nobel Prize in Physics "for groundbreaking experiments regarding the two-dimensional material graphene". They first produced it by lifting graphene flakes from bulk graphite with adhesive tape and then transferring them onto a silicon wafer.[8]
- Graphyne
Graphyne is another 2-dimensional carbon allotrope whose structure is similar to graphene's. It can be seen as a lattice of benzene rings connected by acetylene bonds. Depending on the content of the acetylene groups, graphyne can be considered a mixed hybridization, spn, where 1 < n < 2,[9][10] compared to graphene (pure sp2) and diamond (pure sp3).
First-principle calculations using phonon dispersion curves and ab-initio finite temperature, quantum mechanical molecular dynamics simulations showed graphyne and its boron nitride analogues to be stable.[11]
The existence of graphyne was conjectured before 1960.[12] In 2010, graphdiyne (graphyne with diacetylene groups) was synthesized on copper substrates.[13] In 2022 a team claimed to have successfully used alkyne metathesis to synthesise graphyne though this claim is disputed. [14][15] However, after an investigation the team's paper was retracted by the publication citing fabricated data.[16][verification needed] Later during 2022 synthesis of multi-layered γ‑graphyne was successfully performed through the polymerization of 1,3,5-tribromo-2,4,6-triethynylbenzene under Sonogashira coupling conditions. [17][18] Recently, it has been claimed to be a competitor for graphene due to the potential of direction-dependent Dirac cones.[19][20]
B: borophene
[edit]
36 cluster might be seen as smallest borophene; front and side view
Borophene is a crystalline atomic monolayer of boron and is also known as boron sheet. First predicted by theory in the mid-1990s in a freestanding state,[21] and then demonstrated as distinct monoatomic layers on substrates by Zhang et al.,[22] different borophene structures were experimentally confirmed in 2015.[23][24]
Ge: germanene
[edit]Germanene is a two-dimensional allotrope of germanium with a buckled honeycomb structure.[25] Experimentally synthesized germanene exhibits a honeycomb structure.[26][27] This honeycomb structure consists of two hexagonal sub-lattices that are vertically displaced by 0.2 A from each other.[28]
Si: silicene
[edit]
Silicene is a two-dimensional allotrope of silicon, with a hexagonal honeycomb structure similar to that of graphene.[29][30][31] Its growth is scaffolded by a pervasive Si/Ag(111) surface alloy beneath the two-dimensional layer.[32]
Sn: stanene
[edit]
Stanene is a predicted topological insulator that may display dissipationless currents at its edges near room temperature. It is composed of tin atoms arranged in a single layer, in a manner similar to graphene.[33] Its buckled structure leads to high reactivity against common air pollutants such as NOx and COx and it is able to trap and dissociate them at low temperature.[34] A structure determination of stanene using low energy electron diffraction has shown ultra-flat stanene on a Cu(111) surface.[35]
Pb: plumbene
[edit]Plumbene is a two-dimensional allotrope of lead, with a hexagonal honeycomb structure similar to that of graphene.[36]
P: phosphorene
[edit]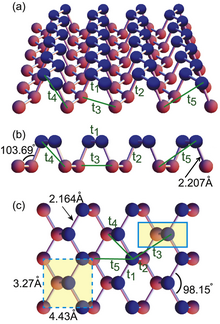
Phosphorene is a 2-dimensional, crystalline allotrope of phosphorus. Its mono-atomic hexagonal structure makes it conceptually similar to graphene. However, phosphorene has substantially different electronic properties; in particular it possesses a nonzero band gap while displaying high electron mobility.[37] This property potentially makes it a better semiconductor than graphene.[38] The synthesis of phosphorene mainly consists of micromechanical cleavage or liquid phase exfoliation methods. The former has a low yield while the latter produce free standing nanosheets in solvent and not on the solid support. The bottom-up approaches like chemical vapor deposition (CVD) are still blank because of its high reactivity. Therefore, in the current scenario, the most effective method for large area fabrication of thin films of phosphorene consists of wet assembly techniques like Langmuir-Blodgett involving the assembly followed by deposition of nanosheets on solid supports.[39]
Sb: antimonene
[edit]Antimonene is a two-dimensional allotrope of antimony, with its atoms arranged in a buckled honeycomb lattice. Theoretical calculations[40] predicted that antimonene would be a stable semiconductor in ambient conditions with suitable performance for (opto)electronics. Antimonene was first isolated in 2016 by micromechanical exfoliation[41] and it was found to be very stable under ambient conditions. Its properties make it also a good candidate for biomedical and energy applications.[42]
In a study made in 2018,[43] antimonene modified screen-printed electrodes (SPE's) were subjected to a galvanostatic charge/discharge test using a two-electrode approach to characterize their supercapacitive properties. The best configuration observed, which contained 36 nanograms of antimonene in the SPE, showed a specific capacitance of 1578 F g−1 at a current of 14 A g−1. Over 10,000 of these galvanostatic cycles, the capacitance retention values drop to 65% initially after the first 800 cycles, but then remain between 65% and 63% for the remaining 9,200 cycles. The 36 ng antimonene/SPE system also showed an energy density of 20 mW h kg−1 and a power density of 4.8 kW kg−1. These supercapacitive properties indicate that antimonene is a promising electrode material for supercapacitor systems. A more recent study,[44] concerning antimonene modified SPEs shows the inherent ability of antimonene layers to form electrochemically passivated layers to facilitate electroanalytical measurements in oxygenated environments, in which the presence of dissolved oxygens normally hinders the analytical procedure. The same study also depicts the in-situ production of antimonene oxide/PEDOT:PSS nanocomposites as electrocatalytic platforms for the determination of nitroaromatic compounds.
Bi: bismuthene
[edit]Bismuthene, the two-dimensional (2D) allotrope of bismuth, was predicted to be a topological insulator. It was predicted that bismuthene retains its topological phase when grown on silicon carbide in 2015.[45] The prediction was successfully realized and synthesized in 2016.[46] At first glance the system is similar to graphene, as the Bi atoms arrange in a honeycomb lattice. However the bandgap is as large as 800mV due to the large spin–orbit interaction (coupling) of the Bi atoms and their interaction with the substrate. Thus, room-temperature applications of the quantum spin Hall effect come into reach. It has been reported to be the largest nontrivial bandgap 2D topological insulator in its natural state.[47][48] Top-down exfoliation of bismuthene has been reported in various instances[49][50] with recent works promoting the implementation of bismuthene in the field of electrochemical sensing.[51][52] Emdadul et al.[53] predicted the mechanical strength and phonon thermal conductivity of monolayer β-bismuthene through atomic-scale analysis. The obtained room temperature (300K) fracture strength is ~4.21 N/m along the armchair direction and ~4.22 N/m along the zigzag direction. At 300 K, its Young's moduli are reported to be ~26.1 N/m and ~25.5 N/m, respectively, along the armchair and zigzag directions. In addition, their predicted phonon thermal conductivity of ~1.3 W/m∙K at 300 K is considerably lower than other analogous 2D honeycombs, making it a promising material for thermoelectric operations.
Au: goldene
[edit]On 16 April 2024, scientists from Linköping University in Sweden reported that they had produced goldene, a single layer of gold atoms 100nm wide. Lars Hultman, a materials scientist on the team behind the new research, is quoted as saying "we submit that goldene is the first free-standing 2D metal, to the best of our knowledge", meaning that it is not attached to any other material, unlike plumbene and stanene. Researchers from New York University Abu Dhabi (NYUAD) previously reported to have synthesised Goldene in 2022, however various other scientists have contended that the NYUAD team failed to prove they made a single-layer sheet of gold, as opposed to a multi-layer sheet. Goldene is expected to be used primarily for its optical properties, with applications such as sensing or as a catalyst.[54]
Metals
[edit]
Single and double atom layers of platinum in a two-dimensional film geometry has been demonstrated.[56][57] These atomically thin platinum films are epitaxially grown on graphene,[56] which imposes a compressive strain that modifies the surface chemistry of the platinum, while also allowing charge transfer through the graphene.[57] Single atom layers of palladium with the thickness down to 2.6 Å,[55] and rhodium with the thickness of less than 4 Å[58] have been synthesized and characterized with atomic force microscopy and transmission electron microscopy.
A 2D titanium formed by additive manufacturing (laser powder bed fusion) achieved greater strength than any known material (50% greater than magnesium alloy WE54). The material was arranged in a tubular lattice with a thin band running inside, merging two complementary lattice structures. This reduced by half the stress at the weakest points in the structure.[59]
2D supracrystals
[edit]The supracrystals of 2D materials have been proposed and theoretically simulated.[60][61] These monolayer crystals are built of supra atomic periodic structures where atoms in the nodes of the lattice are replaced by symmetric complexes. For example, in the hexagonal structure of graphene patterns of 4 or 6 carbon atoms would be arranged hexagonally instead of single atoms, as the repeating node in the unit cell.
2D alloys
[edit]Two-dimensional alloys (or surface alloys) are a single atomic layer of alloy that is incommensurate with the underlying substrate. One example is the 2D ordered alloys of Pb with Sn and with Bi.[62][63] Surface alloys have been found to scaffold two-dimensional layers, as in the case of silicene.[32]
Compounds
[edit]
- Boron nitride nanosheet
- Titanate nanosheet
- Borocarbonitrides
- MXenes
- 2D silica
- Niobium bromide and Niobium chloride (Nb
3[X]
8)[64][65][66]
Transition metal dichalcogenide monolayers
[edit]The most commonly studied two-dimensional transition metal dichalcogenide (TMD) is monolayer molybdenum disulfide (MoS2). Several phases are known, notably the 1T and 2H phases. The naming convention reflects the structure: the 1T phase has one "sheet" (consisting of a layer of S-Mo-S; see figure) per unit cell in a trigonal crystal system, while the 2H phase has two sheets per unit cell in a hexagonal crystal system. The 2H phase is more common, as the 1T phase is metastable and spontaneously reverts to 2H without stabilization by additional electron donors (typically surface S vacancies).[67]
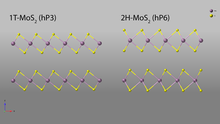
The 2H phase of MoS2 (Pearson symbol hP6; Strukturbericht designation C7) has space group P63/mmc. Each layer contains Mo surrounded by S in trigonal prismatic coordination.[68] Conversely, the 1T phase (Pearson symbol hP3) has space group P-3m1, and octahedrally-coordinated Mo; with the 1T unit cell containing only one layer, the unit cell has a c parameter slightly less than half the length of that of the 2H unit cell (5.95 Å and 12.30 Å, respectively).[69] The different crystal structures of the two phases result in differences in their electronic band structure as well. The d-orbitals of 2H-MoS2 are split into three bands: dz2, dx2-y2,xy, and dxz,yz. Of these, only the dz2 is filled; this combined with the splitting results in a semiconducting material with a bandgap of 1.9eV.[70] 1T-MoS2, on the other hand, has partially filled d-orbitals which give it a metallic character.
Because the structure consists of in-plane covalent bonds and inter-layer van der Waals interactions, the electronic properties of monolayer TMDs are highly anisotropic. For example, the conductivity of MoS2 in the direction parallel to the planar layer (0.1–1 ohm−1cm−1) is ~2200 times larger than the conductivity perpendicular to the layers.[71] There are also differences between the properties of a monolayer compared to the bulk material: the Hall mobility at room temperature is drastically lower for monolayer 2H MoS2 (0.1–10 cm2V−1s−1) than for bulk MoS2 (100–500 cm2V−1s−1). This difference arises primarily due to charge traps between the monolayer and the substrate it is deposited on.[72]
MoS2 has important applications in (electro)catalysis. As with other two-dimensional materials, properties can be highly geometry-dependent; the surface of MoS2 is catalytically inactive, but the edges can act as active sites for catalyzing reactions.[73] For this reason, device engineering and fabrication may involve considerations for maximizing catalytic surface area, for example by using small nanoparticles rather than large sheets[73] or depositing the sheets vertically rather than horizontally.[74] Catalytic efficiency also depends strongly on the phase: the aforementioned electronic properties of 2H MoS2 make it a poor candidate for catalysis applications, but these issues can be circumvented through a transition to the metallic (1T) phase. The 1T phase has more suitable properties, with a current density of 10 mA/cm2, an overpotential of −187 mV relative to RHE, and a Tafel slope of 43 mV/decade (compared to 94 mV/decade for the 2H phase).[75][76]
Graphane
[edit]
While graphene has a hexagonal honeycomb lattice structure with alternating double-bonds emerging from its sp2-bonded carbons, graphane, still maintaining the hexagonal structure, is the fully hydrogenated version of graphene with every sp3-hybrized carbon bonded to a hydrogen (chemical formula of (CH)n). Furthermore, while graphene is planar due to its double-bonded nature, graphane is rugged, with the hexagons adopting different out-of-plane structural conformers like the chair or boat, to allow for the ideal 109.5° angles which reduce ring strain, in a direct analogy to the conformers of cyclohexane.[77]
Graphane was first theorized in 2003,[78] was shown to be stable using first principles energy calculations in 2007,[79] and was first experimentally synthesized in 2009.[80] There are various experimental routes available for making graphane, including the top-down approaches of reduction of graphite in solution or hydrogenation of graphite using plasma/hydrogen gas as well as the bottom-up approach of chemical vapor deposition.[77] Graphane is an insulator, with a predicted band gap of 3.5 eV;[81] however, partially hydrogenated graphene is a semi-conductor, with the band gap being controlled by the degree of hydrogenation.[77]
Germanane
[edit]Germanane is a single-layer crystal composed of germanium with one hydrogen bonded in the z-direction for each atom.[82][83] Germanane's structure is similar to graphane, Bulk germanium does not adopt this structure. Germanane is produced in a two-step route starting with calcium germanide. From this material, the calcium (Ca) is removed by de-intercalation with HCl to give a layered solid with the empirical formula GeH.[84] The Ca sites in Zintl-phase CaGe2 interchange with the hydrogen atoms in the HCl solution, producing GeH and CaCl2.
SLSiN
[edit]SLSiN (acronym for Single-Layer Silicon Nitride), a novel 2D material introduced as the first post-graphene member of Si3N4, was first discovered computationally in 2020 via density-functional theory based simulations.[85] This new material is inherently 2D, insulator with a band-gap of about 4 eV, and stable both thermodynamically and in terms of lattice dynamics.
Combined surface alloying
[edit]Often single-layer materials, specifically elemental allotrops, are connected to the supporting substrate via surface alloys.[32][33] By now, this phenomenon has been proven via a combination of different measurement techniques for silicene,[32] for which the alloy is difficult to prove by a single technique, and hence has not been expected for a long time. Hence, such scaffolding surface alloys beneath two-dimensional materials can be also expected below other two-dimensional materials, significantly influencing the properties of the two-dimensional layer. During growth, the alloy acts as both, foundation and scaffold for the two-dimensional layer, for which it paves the way.[32]
Organic
[edit]Ni3(HITP)2 is an organic, crystalline, structurally tunable electrical conductor with a high surface area. HITP is an organic chemical (2,3,6,7,10,11-hexaaminotriphenylene). It shares graphene's hexagonal honeycomb structure. Multiple layers naturally form perfectly aligned stacks, with identical 2-nm openings at the centers of the hexagons. Room temperature electrical conductivity is ~40 S cm−1, comparable to that of bulk graphite and among the highest for any conducting metal-organic frameworks (MOFs). The temperature dependence of its conductivity is linear at temperatures between 100 K and 500 K, suggesting an unusual charge transport mechanism that has not been previously observed in organic semiconductors.[86]
The material was claimed to be the first of a group formed by switching metals and/or organic compounds. The material can be isolated as a powder or a film with conductivity values of 2 and 40 S cm−1, respectively.[87]
Polymer
[edit]Using melamine (carbon and nitrogen ring structure) as a monomer, researchers created 2DPA-1, a 2-dimensional polymer sheet held together by hydrogen bonds. The sheet forms spontaneously in solution, allowing thin films to be spin-coated. The polymer has a yield strength twice that of steel, and it resists six times more deformation force than bulletproof glass. It is impermeable to gases and liquids.[88][89]
Combinations
[edit]Single layers of 2D materials can be combined into layered assemblies. For example, bilayer graphene is a material consisting of two layers of graphene. One of the first reports of bilayer graphene was in the seminal 2004 Science paper by Geim and colleagues, in which they described devices "which contained just one, two, or three atomic layers". Layered combinations of different 2D materials are generally called van der Waals heterostructures. Twistronics is the study of how the angle (the twist) between layers of two-dimensional materials can change their electrical properties.
Characterization
[edit]Microscopy techniques such as transmission electron microscopy,[90][91][92] 3D electron diffraction,[93] scanning probe microscopy,[94] scanning tunneling microscope,[90] and atomic-force microscopy[90][92][94] are used to characterize the thickness and size of the 2D materials. Electrical properties and structural properties such as composition and defects are characterized by Raman spectroscopy,[90][92][94] X-ray diffraction,[90][92] and X-ray photoelectron spectroscopy.[95]
Mechanical characterization
[edit]The mechanical characterization of 2D materials is difficult due to ambient reactivity and substrate constraints present in many 2D materials. To this end, many mechanical properties are calculated using molecular dynamics simulations or molecular mechanics simulations. Experimental mechanical characterization is possible in 2D materials which can survive the conditions of the experimental setup as well as can be deposited on suitable substrates or exist in a free-standing form. Many 2D materials also possess out-of-plane deformation which further convolute measurements.[96]
Nanoindentation testing is commonly used to experimentally measure elastic modulus, hardness, and fracture strength of 2D materials. From these directly measured values, models exist which allow the estimation of fracture toughness, work hardening exponent, residual stress, and yield strength. These experiments are run using dedicated nanoindentation equipment or an Atomic Force Microscope (AFM). Nanoindentation experiments are generally run with the 2D material as a linear strip clamped on both ends experiencing indentation by a wedge, or with the 2D material as a circular membrane clamped around the circumference experiencing indentation by a curbed tip in the center. The strip geometry is difficult to prepare but allows for easier analysis due to linear resulting stress fields. The circular drum-like geometry is more commonly used and can be easily prepared by exfoliating samples onto a patterned substrate. The stress applied to the film in the clamping process is referred to as the residual stress. In the case of very thin layers of 2D materials bending stress is generally ignored in indentation measurements, with bending stress becoming relevant in multilayer samples. Elastic modulus and residual stress values can be extracted by determining the linear and cubic portions of the experimental force-displacement curve. The fracture stress of the 2D sheet is extracted from the applied stress at failure of the sample. AFM tip size was found to have little effect on elastic property measurement, but the breaking force was found to have a strong tip size dependence due stress concentration at the apex of the tip.[97] Using these techniques the elastic modulus and yield strength of graphene were found to be 342 N/m and 55 N/m respectively.[97]
Poisson's ratio measurements in 2D materials is generally straightforward. To get a value, a 2D sheet is placed under stress and displacement responses are measured, or an MD calculation is run. The unique structures found in 2D materials have been found to result in auxetic behavior in phosphorene[98] and graphene[99] and a Poisson's ratio of zero in triangular lattice borophene.[100]
Shear modulus measurements of graphene has been extracted by measuring a resonance frequency shift in a double paddle oscillator experiment as well as with MD simulations.[101][102]
Fracture toughness of 2D materials in Mode I (KIC) has been measured directly by stretching pre-cracked layers and monitoring crack propagation in real-time.[103] MD simulations as well as molecular mechanics simulations have also been used to calculate fracture toughness in Mode I. In anisotropic materials, such as phosphorene, crack propagation was found to happen preferentially along certain directions.[104] Most 2D materials were found to undergo brittle fracture.
Applications
[edit]The major expectation held amongst researchers is that given their exceptional properties, 2D materials will replace conventional semiconductors to deliver a new generation of electronics.
Biological applications
[edit]Research on 2D nanomaterials is still in its infancy, with the majority of research focusing on elucidating the unique material characteristics and few reports focusing on biomedical applications of 2D nanomaterials.[105] Nevertheless, recent rapid advances in 2D nanomaterials have raised important yet exciting questions about their interactions with biological moieties. 2D nanoparticles such as carbon-based 2D materials, silicate clays, transition metal dichalcogenides (TMDs), and transition metal oxides (TMOs) provide enhanced physical, chemical, and biological functionality owing to their uniform shapes, high surface-to-volume ratios, and surface charge.
Two-dimensional (2D) nanomaterials are ultrathin nanomaterials with a high degree of anisotropy and chemical functionality.[106] 2D nanomaterials are highly diverse in terms of their mechanical, chemical, and optical properties, as well as in size, shape, biocompatibility, and degradability.[107][108] These diverse properties make 2D nanomaterials suitable for a wide range of applications, including drug delivery, imaging, tissue engineering, biosensors, and gas sensors among others.[109][110] However, their low-dimension nanostructure gives them some common characteristics. For example, 2D nanomaterials are the thinnest materials known, which means that they also possess the highest specific surface areas of all known materials. This characteristic makes these materials invaluable for applications requiring high levels of surface interactions on a small scale. As a result, 2D nanomaterials are being explored for use in drug delivery systems, where they can adsorb large numbers of drug molecules and enable superior control over release kinetics.[111] Additionally, their exceptional surface area to volume ratios and typically high modulus values make them useful for improving the mechanical properties of biomedical nanocomposites and nanocomposite hydrogels, even at low concentrations. Their extreme thinness has been instrumental for breakthroughs in biosensing and gene sequencing. Moreover, the thinness of these molecules allows them to respond rapidly to external signals such as light, which has led to utility in optical therapies of all kinds, including imaging applications, photothermal therapy (PTT), and photodynamic therapy (PDT).
Despite the rapid pace of development in the field of 2D nanomaterials, these materials must be carefully evaluated for biocompatibility in order to be relevant for biomedical applications.[112] The newness of this class of materials means that even the relatively well-established 2D materials like graphene are poorly understood in terms of their physiological interactions with living tissues. Additionally, the complexities of variable particle size and shape, impurities from manufacturing, and protein and immune interactions have resulted in a patchwork of knowledge on the biocompatibility of these materials.
See also
[edit]References
[edit]- ^ Ashton, M.; Paul, J.; Sinnott, S. B.; Hennig, R. G. (2017). "Topology-Scaling Identification of Layered Solids and Stable Exfoliated 2D Materials". Phys. Rev. Lett. 118 (10): 106101. arXiv:1610.07673. Bibcode:2017PhRvL.118j6101A. doi:10.1103/PhysRevLett.118.106101. PMID 28339265. S2CID 32012137.
- ^ "Database". 2Dmaterialsweb.org. Archived from the original on 2019-01-22. Retrieved 2022-01-23.
- ^ "Computational 2D Materials Database (C2DB) — COMPUTATIONAL MATERIALS REPOSITORY". cmr.fysik.dtu.dk.
- ^ Zheng, Weiran; Lee, Lawrence Yoon Suk (2022). "Beyond sonication: Advanced exfoliation methods for scalable production of 2D materials". Matter. 5 (2): 515–545. doi:10.1016/j.matt.2021.12.010. S2CID 245902407.
- ^ Andronico, Michael (14 April 2014). "5 Ways Graphene Will Change Gadgets Forever". Laptop.
- ^ "Graphene properties". www.graphene-battery.net. 2014-05-29. Retrieved 2014-05-29.
- ^ "This Month in Physics History: October 22, 2004: Discovery of Graphene". APS News. Series II. 18 (9): 2. 2009.
- ^ "The Nobel Prize in Physics 2010". The Nobel Foundation. Retrieved 2013-12-03.
- ^ Heimann, R.B.; Evsvukov, S.E.; Koga, Y. (1997). "Carbon allotropes: a suggested classification scheme based on valence orbital hybridization". Carbon. 35 (10–11): 1654–1658. Bibcode:1997Carbo..35.1654H. doi:10.1016/S0008-6223(97)82794-7.
- ^ Enyashin, Andrey N.; Ivanovskii, Alexander L. (2011). "Graphene Allotropes". Physica Status Solidi B. 248 (8): 1879–1883. Bibcode:2011PSSBR.248.1879E. doi:10.1002/pssb.201046583. S2CID 125591804.
- ^ Özçelik, V. Ongun; Ciraci, S. (January 10, 2013). "Size Dependence in the Stabilities and Electronic Properties of α-Graphyne and Its Boron Nitride Analogue". The Journal of Physical Chemistry C. 117 (5): 2175–2182. arXiv:1301.2593. doi:10.1021/jp3111869. hdl:11693/11999. S2CID 44136901.
- ^ Balaban AT, Rentia CC, Ciupitu E (1968). "Chemical graphs. 6. Estimation of relative stability of several planar and tridimensional lattices for elementary carbon". Revue Roumaine de Chimie. 13 (2): 231–.
- ^ Li, Guoxing; Li, Yuliang; Liu, Huibiao; Guo, Yanbing; Li, Yongjun; Zhu, Daoben (2010). "Architecture of graphdiyne nanoscale films". Chemical Communications. 46 (19): 3256–3258. doi:10.1039/B922733D. PMID 20442882. S2CID 43416849.
- ^ Hu, Yiming; Wu, Chenyu; Pan, Qingyan; Jin, Yinghua; Lyu, Rui; Martinez, Vikina; Huang, Shaofeng; Wu, Jingyi; Wayment, Lacey J.; Clark, Noel A.; Raschke, Markus B.; Zhao, Yingjie; Zhang, Wei (9 May 2022). "Synthesis of γ-graphyne using dynamic covalent chemistry".
- ^ Hu, Y.; Wu, C.; Pan, Q.; Jin, Y.; Lyu, R.; Martinez, V.; Huang, S.; Wu, J.; Wayment, L.J.; Clark, N.A.; Raschke, M.B.; Zhao, Y.; Zhang, W. (2022). "Synthesis of γ-graphyne using dynamic covalent chemistry". Nature Synthesis. 1 (6): 449–454. Bibcode:2022NatSy...1..449H. doi:10.1038/s44160-022-00068-7. S2CID 248686420. (Retracted, see doi:10.1038/s44160-024-00627-0, Retraction Watch)
- ^ "Faked data prompts retraction of Nature journal study claiming creation of a new form of carbon". 3 September 2024.
- ^ Desyatkin, V. G.; Martin, W. B.; Aliev, A. E.; Chapman, N. E.; Fonseca, A. F.; Galvão, D. S.; Miller, E. R.; Stone, K. H.; Wang, Z.; Zakhidov, D.; Limpoco, F. T.; Almahdali, S. R.; Parker, S. M.; Baughman, R. H.; Rodionov, V. O. (2022). "Scalable Synthesis and Characterization of Multilayer γ‑Graphyne, New Carbon Crystals with a Small Direct Band Gap". Journal of the American Chemical Society. 144 (39): 17999–18008. arXiv:2301.05291. doi:10.1021/jacs.2c06583. PMID 36130080. S2CID 252438218.
- ^ Kang, Jun; Wei, Zhongming; Li, Jingbo (2019). "Graphyne and Its Family: Recent Theoretical Advances". ACS Applied Materials & Interfaces. 11 (3): 2692–2706. doi:10.1021/acsami.8b03338. PMID 29663794.
- ^ Gopalakrishnan, K.; Moses, Kota; Govindaraj, A.; Rao, C. N. R. (2013-12-01). "Supercapacitors based on nitrogen-doped reduced graphene oxide and borocarbonitrides". Solid State Communications. Special Issue: Graphene V: Recent Advances in Studies of Graphene and Graphene analogues. 175–176: 43–50. Bibcode:2013SSCom.175...43G. doi:10.1016/j.ssc.2013.02.005.
- ^ Schirber, Michael (24 February 2012). "Focus: Graphyne May Be Better than Graphene". Physics. 5 (24): 24. Bibcode:2012PhyOJ...5...24S. doi:10.1103/Physics.5.24.
- ^ Boustani, Ihsan (January 1997). "New quasi-planar surfaces of bare boron". Surface Science. 370 (2–3): 355–363. Bibcode:1997SurSc.370..355B. doi:10.1016/S0039-6028(96)00969-7.
- ^ Zhang, Z.; Yang, Y.; Gao, G.; Yakobson, B.I. (2 September 2015). "Two-Dimensional Boron Monolayers Mediated by Metal Substrates". Angewandte Chemie International Edition. 54 (44): 13022–13026. doi:10.1002/anie.201505425. PMID 26331848.
- ^ Mannix, A. J.; Zhou, X.-F.; Kiraly, B.; Wood, J. D.; Alducin, D.; Myers, B. D.; Liu, X.; Fisher, B. L.; Santiago, U.; Guest, J. R.; et al. (17 December 2015). "Synthesis of borophenes: Anisotropic, two-dimensional boron polymorphs". Science. 350 (6267): 1513–1516. Bibcode:2015Sci...350.1513M. doi:10.1126/science.aad1080. PMC 4922135. PMID 26680195.
- ^ Feng, Baojie; Zhang, Jin; Zhong, Qing; Li, Wenbin; Li, Shuai; Li, Hui; Cheng, Peng; Meng, Sheng; Chen, Lan; Wu, Kehui (28 March 2016). "Experimental realization of two-dimensional boron sheets". Nature Chemistry. 8 (6): 563–568. arXiv:1512.05029. Bibcode:2016NatCh...8..563F. doi:10.1038/nchem.2491. PMID 27219700. S2CID 19475989.
- ^ Bampoulis, P.; Zhang, L.; Safaei, A.; van Gastel, R.; Poelsema, B.; Zandvliet, H. J. W. (2014). "Germanene termination of Ge2Pt crystals on Ge(110)". Journal of Physics: Condensed Matter. 26 (44): 442001. arXiv:1706.00697. Bibcode:2014JPCM...26R2001B. doi:10.1088/0953-8984/26/44/442001. PMID 25210978. S2CID 36478002.
- ^ Derivaz, Mickael; Dentel, Didier; Stephan, Regis; Hanf, Marie-Christine; Mehdaoui, Ahmed; Sonnet, Philippe; Pirri, Carmelo (2015). "Continuous germanene layer on Al (111)". Nano Letters. 15 (4). ACS Publications: 2510–2516. Bibcode:2015NanoL..15.2510D. doi:10.1021/acs.nanolett.5b00085. PMID 25802988.
- ^ Yuhara, J.; Shimazu, H.; Ito, K.; Ohta, A.; Kurosawa, M.; Nakatake, M.; Le Lay, Guy (2018). "Germanene Epitaxial Growth by Segregation through Ag(111) Thin Films on Ge(111)". ACS Nano. 12 (11): 11632–11637. doi:10.1021/acsnano.8b07006. PMID 30371060. S2CID 53102735.
- ^ Lee, Kangho; Kim, Hye-Young; Lotya, Mustafa; Coleman, Jonathan N.; Kim, Gyu-Tae; Duesberg, Georg S. (2011-09-22). "Electrical Characteristics of Molybdenum Disulfide Flakes Produced by Liquid Exfoliation". Advanced Materials. 23 (36): 4178–4182. Bibcode:2011AdM....23.4178L. doi:10.1002/adma.201101013. PMID 21823176. S2CID 205240634.
- ^ Xu, Mingsheng; Liang, Tao; Shi, Minmin; Chen, Hongzheng (2013). "Graphene-like two-dimensional materials". Chemical Reviews. 113 (5). ACS Publications: 3766–3798. doi:10.1021/cr300263a. PMID 23286380.
- ^ Cahangirov, S.; Topsakal, M.; Aktürk, E.; Şahin, H.; Ciraci, S. (2009). "Two- and One-Dimensional Honeycomb Structures of Silicon and Germanium". Phys. Rev. Lett. 102 (23): 236804. arXiv:0811.4412. Bibcode:2009PhRvL.102w6804C. doi:10.1103/PhysRevLett.102.236804. PMID 19658958. S2CID 22106457.
- ^ Stephan, Régis; Hanf, Marie-Christine; Sonnet, Philippe (2014). "Spatial analysis of interactions at the silicene/Ag interface: first principles study". Journal of Physics: Condensed Matter. 27 (1). IOP Publishing: 015002. doi:10.1088/0953-8984/27/1/015002. PMID 25407116. S2CID 39842095.
- ^ a b c d e Küchle, Johannes T.; Baklanov, Aleksandr; Seitsonen, Ari P.; Ryan, Paul T.P.; Feulner, Peter; Pendem, Prashanth; Lee, Tien-Lin; Muntwiler, Matthias; Schwarz, Martin; Haag, Felix; Barth, Johannes V.; Auwärter, Willi; Duncan, David A.; Allegretti, Francesco (2022). "Silicene's pervasive surface alloy on Ag(111): a scaffold for two-dimensional growth". 2D Materials. 9 (4): 045021. Bibcode:2022TDM.....9d5021K. doi:10.1088/2053-1583/ac8a01. S2CID 251637081.
- ^ a b Yuhara, J.; Fujii, Y.; Isobe, N.; Nakatake, M.; Lede, X.; Rubio, A.; Le Lay, G. (2018). "Large Area Planar Stanene Epitaxially Grown on Ag(111)". 2D Materials. 5 (2). 025002. Bibcode:2018TDM.....5b5002Y. doi:10.1088/2053-1583/aa9ea0. hdl:21.11116/0000-0001-A92C-0.
- ^ Takahashi, L.; Takahashi, K. (2015). "Low temperature pollutant trapping and dissociation over two-dimensional tin". Physical Chemistry Chemical Physics. 17 (33): 21394–21396. Bibcode:2015PCCP...1721394T. doi:10.1039/C5CP03382A. PMID 26226204. Supporting Information
- ^ Ahmed, Rezwan; Nakagawa, Takeshi; Mizuno, Seigi (2020). "Structure determination of ultra-flat stanene on Cu(111) using low energy electron diffraction". Surface Science. 691: 121498. Bibcode:2020SurSc.69121498A. doi:10.1016/j.susc.2019.121498. S2CID 203142186.
- ^ Yuhara, J.; He, B.; Le Lay, G. (2019). "Graphene's Latest Cousin: Plumbene Epitaxial Growth on a "Nano WaterCube"". Advanced Materials. 31 (27): 1901017. Bibcode:2019AdM....3101017Y. doi:10.1002/adma.201901017. PMID 31074927. S2CID 149446617..
- ^ Berger, Andy (July 17, 2015). "Beyond Graphene, a Zoo of New 2-D Materials". Discover Magazine. Archived from the original on 2019-11-01. Retrieved 2015-09-19.
- ^ Li, L.; Yu, Y.; Ye, G. J.; Ge, Q.; Ou, X.; Wu, H.; Feng, D.; Chen, X. H.; Zhang, Y. (2014). "Black phosphorus field-effect transistors". Nature Nanotechnology. 9 (5): 372–377. arXiv:1401.4117. Bibcode:2014NatNa...9..372L. doi:10.1038/nnano.2014.35. PMID 24584274. S2CID 17218693.
- ^ Ritu, Harneet (2016). "Large Area Fabrication of Semiconducting Phosphorene by Langmuir-Blodgett Assembly". Sci. Rep. 6: 34095. arXiv:1605.00875. Bibcode:2016NatSR...634095K. doi:10.1038/srep34095. PMC 5037434. PMID 27671093.
- ^ Zhang, S.; Yan, Z.; Li, Y.; Chen, Z.; Zeng, H. (2015). "Atomically Thin Arsenene and Antimonene: Semimetal-Semiconductor and Indirect-Direct Band-Gap Transitions". Angew. Chem. Int. Ed. 54 (10): 3112–3115. doi:10.1002/anie.201411246. PMID 25564773.
- ^ Ares, P.; Aguilar-Galindo, F.; Rodríguez-San-Miguel, D.; Aldave, D. A.; Díaz-Tendero, S.; Alcamí, M.; Martín, F.; Gómez-Herrero, J.; Zamora, F. (2016). "Mechanical Isolation of Highly Stable Antimonene under Ambient Conditions". Adv. Mater. 28 (30): 6332–6336. arXiv:1608.06859. Bibcode:2016AdM....28.6332A. doi:10.1002/adma.201602128. hdl:10486/672484. PMID 27272099. S2CID 8296292.
- ^ Ares, P.; Palacios, J. J.; Abellán, G.; Gómez-Herrero, J.; Zamora, F. (2018). "Recent progress on antimonene: a new bidimensional material". Adv. Mater. 30 (2): 1703771. Bibcode:2018AdM....3003771A. doi:10.1002/adma.201703771. hdl:10486/688820. PMID 29076558. S2CID 205282902.
- ^ Martínez-Periñán, Emiliano; Down, Michael P.; Gibaja, Carlos; Lorenzo, Encarnación; Zamora, Félix; Banks, Craig E. (2018). "Antimonene: A Novel 2D Nanomaterial for Supercapacitor Applications" (PDF). Advanced Energy Materials. 8 (11): 1702606. Bibcode:2018AdEnM...802606M. doi:10.1002/aenm.201702606. hdl:10486/688798. ISSN 1614-6840. S2CID 103042887.
- ^ Lazanas, Alexandros Ch.; Prodromidis, Mamas I. (April 2022). "Electrochemical performance of passivated antimonene nanosheets and of in-situ prepared antimonene oxide-PEDOT:PSS modified screen-printed graphite electrodes". Electrochimica Acta. 410: 140033. doi:10.1016/j.electacta.2022.140033. S2CID 246598714.
- ^ Hsu, Chia-Hsiu; Huang, Zhi-Quan; Chuang, Feng-Chuan; Kuo, Chien-Cheng; Liu, Yu-Tzu; Lin, Hsin; Bansil, Arun (2015-02-10). "The nontrivial electronic structure of Bi/Sb honeycombs on SiC(0001)". New Journal of Physics. 17 (2): 025005. Bibcode:2015NJPh...17b5005H. doi:10.1088/1367-2630/17/2/025005.
- ^ Reis, Felix; Li, Gang; Dudy, Lenart; Bauernfiend, Maximilian; Glass, Stefan; Hanke, Werner; Thomale, Ronny; Schaefer, Joerg; Claessen, Ralph (July 21, 2017). "Bismuthene on a SiC substrate: A candidate for a high-temperature quantum spin Hall material". Science. 357 (6348): 287–290. arXiv:1608.00812. Bibcode:2017Sci...357..287R. doi:10.1126/science.aai8142. PMID 28663438. S2CID 23323210.
- ^ Liu, Zheng; Liu, Chao-Xing; Wu, Yong-Shi; Duan, Wen-Hui; Liu, Feng; Wu, Jian (2011-09-23). "Stable NontrivialZ2Topology in Ultrathin Bi (111) Films: A First-Principles Study". Physical Review Letters. 107 (13): 136805. arXiv:1104.0978. Bibcode:2011PhRvL.107m6805L. doi:10.1103/physrevlett.107.136805. ISSN 0031-9007. PMID 22026889. S2CID 10121875.
- ^ Murakami, Shuichi (2006-12-06). "Quantum Spin Hall Effect and Enhanced Magnetic Response by Spin-Orbit Coupling". Physical Review Letters. 97 (23): 236805. arXiv:cond-mat/0607001. Bibcode:2006PhRvL..97w6805M. doi:10.1103/physrevlett.97.236805. ISSN 0031-9007. PMID 17280226. S2CID 34984890.
- ^ Qi-Qi, Yang (2 October 2018). "2D bismuthene fabricated via acid-intercalated exfoliation showing strong nonlinear near-infrared responses for mode-locking lasers". Nanoscale. 10 (45): 21106–21115. doi:10.1039/c8nr06797j. PMID 30325397.
- ^ Gusmao, Rui; Sofer, Zdenek; Bousa, Daniel; Pumera, Martin (29 July 2017). "Pnictogens (As, Sb, Bi) Nanosheets by Shear Exfoliation Using Kitchen Blenders for Electrochemical Applications". Angewandte Chemie International Edition. 56 (46): 14417–14422. doi:10.1002/anie.201706389. PMID 28755460. S2CID 22513370.
- ^ Martinez, Carmen C.; Gusmao, Rui; Sofer, Zdenek; Pumera, Martin (2019). "Pnictogen-Based Enzymatic Phenol Biosensors: Phosphorene, Arsenene, Antimonene, and Bismuthene". Angewandte Chemie International Edition. 58 (1): 134–138. doi:10.1002/anie.201808846. PMID 30421531. S2CID 53291371.
- ^ Lazanas, Alexandros Ch.; Tsirka, Kyriaki; Paipetis, Alkiviadis S.; Prodromidis, Mamas I. (2020). "2D bismuthene/graphene modified electrodes for the ultra-sensitive stripping voltammetric determination of lead and cadmium". Electrochimica Acta. 336: 135726. doi:10.1016/j.electacta.2020.135726. S2CID 214292108.
- ^ Chowdhury, Emdadul Haque; Rahman, Md. Habibur; Bose, Pritom; Jayan, Rahul; Islam, Md Mahbubul (2020). "Atomic-scale analysis of the physical strength and phonon transport mechanisms of monolayer β-bismuthene". Physical Chemistry Chemical Physics. 22 (48): 28238–28255. Bibcode:2020PCCP...2228238C. doi:10.1039/d0cp04785f. ISSN 1463-9076. PMID 33295342. S2CID 228079431.
- ^ Peplow, Mark (18 April 2024). "Meet 'goldene': this gilded cousin of graphene is also one atom thick". Nature. Retrieved 19 April 2024.
- ^ a b Yin, Xi; Liu, Xinhong; Pan, Yung-Tin; Walsh, Kathleen A.; Yang, Hong (November 4, 2014). "Hanoi Tower-like Multilayered Ultrathin Palladium Nanosheets". Nano Letters. 14 (12): 7188–7194. Bibcode:2014NanoL..14.7188Y. doi:10.1021/nl503879a. PMID 25369350.
- ^ a b Abdelhafiz, Ali; Vitale, Adam; Buntin, Parker; deGlee, Ben; Joiner, Corey; Robertson, Alex; Vogel, Eric M.; Warner, Jamie; Alamgir, Faisal M. (2018). "Epitaxial and atomically thin graphene–metal hybrid catalyst films: the dual role of graphene as the support and the chemically-transparent protective cap". Energy & Environmental Science. 11 (6): 1610–1616. doi:10.1039/c8ee00539g.
- ^ a b Abdelhafiz, Ali; Vitale, Adam; Joiner, Corey; Vogel, Eric; Alamgir, Faisal M. (2015-03-16). "Layer-by-Layer Evolution of Structure, Strain, and Activity for the Oxygen Evolution Reaction in Graphene-Templated Pt Monolayers". ACS Applied Materials & Interfaces. 7 (11): 6180–6188. doi:10.1021/acsami.5b00182. PMID 25730297.
- ^ Duan, Haohong; Yan, Ning; Yu, Rong; Chang, Chun-Ran; Zhou, Gang; Hu, Han-Shi; Rong, Hongpan; Niu, Zhiqiang; Mao, Junjie; Asakura, Hiroyuki; Tanaka, Tsunehiro; Dyson, Paul Joseph; Li, Jun; Li, Yadong (17 January 2014). "Ultrathin rhodium nanosheets". Nature Communications. 5: 3093. Bibcode:2014NatCo...5.3093D. doi:10.1038/ncomms4093. PMID 24435210.
- ^ Paleja, Ameya (2024-02-26). "Australian scientists 3D print titanium structure with supernatural strength". Interesting Engineering. Retrieved 2024-02-27.
- ^ Kochaev, A. I.; Karenin, A.A.; Meftakhutdinov, R.M.; Brazhe, R.A. (2012). "2D supracrystals as a promising materials for planar nanoacoustoelectronics". Journal of Physics: Conference Series. 345 (1): 012007. Bibcode:2012JPhCS.345a2007K. doi:10.1088/1742-6596/345/1/012007.
- ^ Brazhe, R. A.; Kochaev, A. I. (2012). "Flexural waves in graphene and 2D supracrystals". Physics of the Solid State. 54 (8): 1612–1614. Bibcode:2012PhSS...54.1612B. doi:10.1134/S1063783412080069. S2CID 120094142.
- ^ Yuhara, J.; Schmid, M.; Varga, P. (2003). "A two-dimensional alloy of immiscible metals, The single and binary monolayer films of Pb and Sn on Rh(111)". Phys. Rev. B. 67 (19): 195407. Bibcode:2003PhRvB..67s5407Y. doi:10.1103/PhysRevB.67.195407.
- ^ Yuhara, J.; Yokoyama, M.; Matsui, T. (2011). "Two-dimensional solid solution alloy of Bi-Pb binary films on Rh(111)". J. Appl. Phys. 110 (7): 074314–074314–4. Bibcode:2011JAP...110g4314Y. doi:10.1063/1.3650883.
- ^ Blain, Loz (2022-04-28). "One-way superconducting diode has massive implications for electronics". New Atlas. Retrieved 2022-04-29.
- ^ Pasco, Christopher M.; Baggari, Ismail El; Bianco, Elisabeth; Kourkoutis, Lena F.; McQueen, Tyrel M. (2019-07-23). "Tunable Magnetic Transition to a Singlet Ground State in a 2D Van der Waals Layered Trimerized Kagom\'e Magnet". ACS Nano. 13 (8): 9457–9463. arXiv:1907.10108v1. doi:10.1021/acsnano.9b04392. PMID 31310516. S2CID 197422328.
- ^ Wu, Heng; Wang, Yaojia; Xu, Yuanfeng; Sivakumar, Pranava K.; Pasco, Chris; Filippozzi, Ulderico; Parkin, Stuart S. P.; Zeng, Yu-Jia; McQueen, Tyrel; Ali, Mazhar N. (April 2022). "The field-free Josephson diode in a van der Waals heterostructure". Nature. 604 (7907): 653–656. arXiv:2103.15809. Bibcode:2022Natur.604..653W. doi:10.1038/s41586-022-04504-8. ISSN 1476-4687. PMID 35478238. S2CID 248414862.
- ^ Gan, Xiaorong; Lee, Lawrence Yoon Suk; Wong, Kwok-yin; Lo, Tsz Wing; Ho, Kwun Hei; Lei, Dang Yuan; Zhao, Huimin (2018). "2H/1T Phase Transition of Multilayer MoS2 by Electrochemical Incorporation of S Vacancies". ACS Applied Energy Materials. 1 (9): 4754–4765. doi:10.1021/acsaem.8b00875. S2CID 106014720.
- ^ Dickinson, Roscoe G.; Pauling, Linus (1923). "The Crystal Structure of Molybdenite". Journal of the American Chemical Society. 45 (6): 1466–1471. doi:10.1021/ja01659a020.
- ^ Fang, Yuqiang; Pan, Jie; He, Jianqiao; Luo, Ruichun; Wang, Dong; Che, Xiangli; Bu, Kejun; Zhao, Wei; Liu, Pan; Mu, Gang; Zhang, Hui; Lin, Tianquan; Huang, Fuqiang (2018-01-26). "Structure Re-determination and Superconductivity Observation of Bulk 1T MoS 2". Angewandte Chemie International Edition. 57 (5): 1232–1235. arXiv:1712.09248. doi:10.1002/anie.201710512. PMID 29210496. S2CID 205406195.
- ^ Splendiani, Andrea; Sun, Liang; Zhang, Yuanbo; Li, Tianshu; Kim, Jonghwan; Chim, Chi-Yung; Galli, Giulia; Wang, Feng (2010-04-14). "Emerging Photoluminescence in Monolayer MoS 2". Nano Letters. 10 (4): 1271–1275. Bibcode:2010NanoL..10.1271S. doi:10.1021/nl903868w. ISSN 1530-6984. PMID 20229981.
- ^ Tributsch, H. (1977). "Layer-Type Transition Metal Dichalcogenides – a New Class of Electrodes for Electrochemical Solar Cells". Berichte der Bunsengesellschaft für physikalische Chemie. 81 (4): 361–369. doi:10.1002/bbpc.19770810403.
- ^ Fivaz, R.; Mooser, E. (1967-11-15). "Mobility of Charge Carriers in Semiconducting Layer Structures". Physical Review. 163 (3): 743–755. Bibcode:1967PhRv..163..743F. doi:10.1103/PhysRev.163.743. ISSN 0031-899X.
- ^ a b Salazar, Norberto; Rangarajan, Srinivas; Rodríguez-Fernández, Jonathan; Mavrikakis, Manos; Lauritsen, Jeppe V. (2020-08-31). "Site-dependent reactivity of MoS2 nanoparticles in hydrodesulfurization of thiophene". Nature Communications. 11 (1): 4369. doi:10.1038/s41467-020-18183-4. ISSN 2041-1723. PMC 7459117. PMID 32868769.
- ^ Kong, Desheng; Wang, Haotian; Cha, Judy J.; Pasta, Mauro; Koski, Kristie J.; Yao, Jie; Cui, Yi (2013-03-13). "Synthesis of MoS 2 and MoSe 2 Films with Vertically Aligned Layers". Nano Letters. 13 (3): 1341–1347. Bibcode:2013NanoL..13.1341K. doi:10.1021/nl400258t. ISSN 1530-6984. PMID 23387444.
- ^ Lukowski, Mark A.; Daniel, Andrew S.; Meng, Fei; Forticaux, Audrey; Li, Linsen; Jin, Song (2013-07-17). "Enhanced Hydrogen Evolution Catalysis from Chemically Exfoliated Metallic MoS 2 Nanosheets". Journal of the American Chemical Society. 135 (28): 10274–10277. doi:10.1021/ja404523s. ISSN 0002-7863. PMID 23790049.
- ^ Zhang, Wencui; Liao, Xiaobin; Pan, Xuelei; Yan, Mengyu; Li, Yanxi; Tian, Xiaocong; Zhao, Yan; Xu, Lin; Mai, Liqiang (2019). "Superior Hydrogen Evolution Reaction Performance in 2H-MoS 2 to that of 1T Phase". Small. 15 (31): 1900964. doi:10.1002/smll.201900964. ISSN 1613-6810. PMID 31211511. S2CID 190523589.
- ^ a b c Pumera, Martin; Wong, Colin Hong An (2013). "Graphane and hydrogenated graphene". Chemical Society Reviews. 42 (14): 5987–5995. doi:10.1039/c3cs60132c. ISSN 0306-0012. PMID 23686139.
- ^ Sluiter, Marcel H. F.; Kawazoe, Yoshiyuki (2003-08-21). "Cluster expansion method for adsorption: Application to hydrogen chemisorption on graphene". Physical Review B. 68 (8): 085410. Bibcode:2003PhRvB..68h5410S. doi:10.1103/PhysRevB.68.085410. ISSN 0163-1829.
- ^ Sofo, Jorge O.; Chaudhari, Ajay S.; Barber, Greg D. (2007-04-10). "Graphane: A two-dimensional hydrocarbon". Physical Review B. 75 (15): 153401. arXiv:cond-mat/0606704. Bibcode:2007PhRvB..75o3401S. doi:10.1103/PhysRevB.75.153401. ISSN 1098-0121. S2CID 101537520.
- ^ Elias, D. C.; Nair, R. R.; Mohiuddin, T. M. G.; Morozov, S. V.; Blake, P.; Halsall, M. P.; Ferrari, A. C.; Boukhvalov, D. W.; Katsnelson, M. I.; Geim, A. K.; Novoselov, K. S. (2009-01-30). "Control of Graphene's Properties by Reversible Hydrogenation: Evidence for Graphane". Science. 323 (5914): 610–613. arXiv:0810.4706. Bibcode:2009Sci...323..610E. doi:10.1126/science.1167130. ISSN 0036-8075. PMID 19179524. S2CID 3536592.
- ^ Zhou, Chao; Chen, Sihao; Lou, Jianzhong; Wang, Jihu; Yang, Qiujie; Liu, Chuanrong; Huang, Dapeng; Zhu, Tonghe (2014-01-13). "Graphene's cousin: the present and future of graphane". Nanoscale Research Letters. 9 (1): 26. Bibcode:2014NRL.....9...26Z. doi:10.1186/1556-276X-9-26. ISSN 1556-276X. PMC 3896693. PMID 24417937.
- ^ Garcia, J. C.; de Lima, D. B.; Assali, L. V. C.; Justo, J. F. (2011). "Group IV Graphene- and Graphane-Like Nanosheets". J. Phys. Chem. C. 115 (27): 13242. arXiv:1204.2875. doi:10.1021/jp203657w.
- ^ Bianco, E.; Butler, S.; Jiang, S.; Restrepo, O. D.; Windl, W.; Goldberger, J. E. (2013). "Stability and Exfoliation of Germanane: A Germanium Graphane Analogue". ACS Nano. 7 (5): 4414–21. doi:10.1021/nn4009406. hdl:1811/54792. PMID 23506286.
- ^ "'Germanane' may replace silicon for lighter, faster electronics". KurzweilAI. Retrieved 2013-04-12.
- ^ Shekaari, Ashkan; Jafari, Mahmoud (July 2020). "Unveiling the first post-graphene member of silicon nitrides: A novel 2D material". Computational Materials Science. 180: 109693. doi:10.1016/j.commatsci.2020.109693. S2CID 216464099.
- ^ Sheberla, Dennis; Sun, Lei; Blood-Forsythe, Martin A.; Er, Süleyman; Wade, Casey R.; Brozek, Carl K.; Aspuru-Guzik, Alán; Dincă, Mircea (2014). "High Electrical Conductivity in Ni3(2,3,6,7,10,11-hexaiminotriphenylene)2, a Semiconducting Metal–Organic Graphene Analogue". Journal of the American Chemical Society. 136 (25): 8859–8862. doi:10.1021/ja502765n. PMID 24750124. S2CID 5714037.
- ^ "A new self-assembling graphene-like material for flat semiconductors". KurzweilAI. 2014-05-01. Retrieved 2014-08-24.
- ^ Irving, Michael (2022-02-03). ""Impossible" 2D material is light as plastic and stronger than steel". New Atlas. Retrieved 2022-02-03.
- ^ Zeng, Yuwen; Gordiichuk, Pavlo; Ichihara, Takeo; Zhang, Ge; Sandoz-Rosado, Emil; Wetzel, Eric D.; Tresback, Jason; Yang, Jing; Kozawa, Daichi; Yang, Zhongyue; Kuehne, Matthias (2022-02-03). "Irreversible synthesis of an ultrastrong two-dimensional polymeric material". Nature. 602 (7895): 91–95. Bibcode:2022Natur.602...91Z. doi:10.1038/s41586-021-04296-3. ISSN 0028-0836. PMID 35110762. S2CID 246487991.
- ^ a b c d e Butler, Sheneve Z.; Hollen, Shawna M.; Cao, Linyou; Cui, Yi; Gupta, Jay A.; Gutiérrez, Humberto R.; Heinz, Tony F.; Hong, Seung Sae; Huang, Jiaxing (2013). "Progress, Challenges, and Opportunities in Two-Dimensional Materials Beyond Graphene". ACS Nano. 7 (4): 2898–2926. doi:10.1021/nn400280c. PMID 23464873.
- ^ Bhimanapati, Ganesh R.; Lin, Zhong; Meunier, Vincent; Jung, Yeonwoong; Cha, Judy; Das, Saptarshi; Xiao, Di; Son, Youngwoo; Strano, Michael S. (2015). "Recent Advances in Two-Dimensional Materials beyond Graphene". ACS Nano. 9 (12): 11509–11539. doi:10.1021/acsnano.5b05556. PMID 26544756.
- ^ a b c d Rao, C. N. R.; Nag, Angshuman (2010-09-01). "Inorganic Analogues of Graphene". European Journal of Inorganic Chemistry. 2010 (27): 4244–4250. doi:10.1002/ejic.201000408.
- ^ Sung, S.H.; Schnitzer, N.; Brown, L.; Park, J.; Hovden, R. (2019-06-25). "Stacking, strain, and twist in 2D materials quantified by 3D electron diffraction". Physical Review Materials. 3 (6): 064003. arXiv:1905.11354. Bibcode:2019PhRvM...3f4003S. doi:10.1103/PhysRevMaterials.3.064003. S2CID 166228311.
- ^ a b c Rao, C. N. R.; Ramakrishna Matte, H. S. S.; Maitra, Urmimala (2013-12-09). "Graphene Analogues of Inorganic Layered Materials". Angewandte Chemie International Edition. 52 (50): 13162–13185. doi:10.1002/anie.201301548. PMID 24127325.
- ^ Rao, C. N. R; Maitra, Urmimala (2015-01-01). "Inorganic Graphene Analogs". Annual Review of Materials Research. 45 (1): 29–62. Bibcode:2015AnRMS..45...29R. doi:10.1146/annurev-matsci-070214-021141.
- ^ Androulidakis, Charalampos; Zhang, Kaihao; Robertson, Matthew; Tawfick, Sameh (2018-06-13). "Tailoring the mechanical properties of 2D materials and heterostructures". 2D Materials. 5 (3): 032005. Bibcode:2018TDM.....5c2005A. doi:10.1088/2053-1583/aac764. ISSN 2053-1583. S2CID 139728037.
- ^ a b Lee, Changgu; Wei, Xiaoding; Kysar, Jeffrey W.; Hone, James (2008-07-18). "Measurement of the Elastic Properties and Intrinsic Strength of Monolayer Graphene". Science. 321 (5887): 385–388. Bibcode:2008Sci...321..385L. doi:10.1126/science.1157996. ISSN 0036-8075. PMID 18635798. S2CID 206512830.
- ^ Jiang, Jin-Wu; Park, Harold S. (2014-08-18). "Negative poisson's ratio in single-layer black phosphorus". Nature Communications. 5 (1): 4727. arXiv:1403.4326. Bibcode:2014NatCo...5.4727J. doi:10.1038/ncomms5727. ISSN 2041-1723. PMID 25131569. S2CID 9132961.
- ^ Jiang, Jin-Wu; Chang, Tienchong; Guo, Xingming; Park, Harold S. (2016-08-10). "Intrinsic Negative Poisson's Ratio for Single-Layer Graphene". Nano Letters. 16 (8): 5286–5290. arXiv:1605.01827. Bibcode:2016NanoL..16.5286J. doi:10.1021/acs.nanolett.6b02538. ISSN 1530-6984. PMID 27408994. S2CID 33516006.
- ^ Zhang, Zhuhua; Yang, Yang; Penev, Evgeni S.; Yakobson, Boris I. (March 2017). "Elasticity, Flexibility, and Ideal Strength of Borophenes". Advanced Functional Materials. 27 (9): 1605059. arXiv:1609.07533. doi:10.1002/adfm.201605059. ISSN 1616-301X. S2CID 119199830.
- ^ Liu, Xiao; Metcalf, Thomas H.; Robinson, Jeremy T.; Houston, Brian H.; Scarpa, Fabrizio (2012-02-08). "Shear Modulus of Monolayer Graphene Prepared by Chemical Vapor Deposition". Nano Letters. 12 (2): 1013–1017. Bibcode:2012NanoL..12.1013L. doi:10.1021/nl204196v. ISSN 1530-6984. PMID 22214257.
- ^ Min, K.; Aluru, N. R. (2011-01-03). "Mechanical properties of graphene under shear deformation". Applied Physics Letters. 98 (1): 013113. Bibcode:2011ApPhL..98a3113M. doi:10.1063/1.3534787. ISSN 0003-6951.
- ^ Zhang, Peng; Ma, Lulu; Fan, Feifei; Zeng, Zhi; Peng, Cheng; Loya, Phillip E.; Liu, Zheng; Gong, Yongji; Zhang, Jiangnan; Zhang, Xingxiang; Ajayan, Pulickel M. (2014-04-29). "Fracture toughness of graphene". Nature Communications. 5 (1): 3782. Bibcode:2014NatCo...5.3782Z. doi:10.1038/ncomms4782. ISSN 2041-1723. PMID 24777167.
- ^ Liu, Ning; Hong, Jiawang; Pidaparti, Ramana; Wang, Xianqiao (2016-03-03). "Fracture patterns and the energy release rate of phosphorene". Nanoscale. 8 (10): 5728–5736. Bibcode:2016Nanos...8.5728L. doi:10.1039/C5NR08682E. ISSN 2040-3372. PMID 26902970.
- ^ Kerativitayanan, P; Carrow, JK; Gaharwar, AK (26 May 2015). "Nanomaterials for Engineering Stem Cell Responses". Advanced Healthcare Materials. 4 (11): 1600–27. doi:10.1002/adhm.201500272. PMID 26010739. S2CID 21582516.
- ^ Huang, X; Tan, C; Yin, Z; Zhang, H (9 April 2014). "25th anniversary article: hybrid nanostructures based on two-dimensional nanomaterials". Advanced Materials. 26 (14): 2185–204. Bibcode:2014AdM....26.2185H. doi:10.1002/adma.201304964. PMID 24615947. S2CID 196924648.
- ^ Carrow, James K.; Gaharwar, Akhilesh K. (February 2015). "Bioinspired Polymeric Nanocomposites for Regenerative Medicine". Macromolecular Chemistry and Physics. 216 (3): 248–264. doi:10.1002/macp.201400427.
- ^ Nandwana, Dinkar; Ertekin, Elif (21 June 2015). "Lattice mismatch induced ripples and wrinkles in planar graphene/boron nitride superlattices". Journal of Applied Physics. 117 (234304): 234304. arXiv:1504.02929. Bibcode:2015JAP...117w4304N. doi:10.1063/1.4922504. S2CID 119251606.
- ^ Gaharwar, AK; Peppas, NA; Khademhosseini, A (March 2014). "Nanocomposite hydrogels for biomedical applications". Biotechnology and Bioengineering. 111 (3): 441–53. doi:10.1002/bit.25160. PMC 3924876. PMID 24264728.
- ^ Filipovic, L; Selberherr, S (October 2022). "Application of Two-Dimensional Materials towards CMOS-Integrated Gas Sensors". Nanomaterials. 12 (20): 3651(1–60). doi:10.3390/nano12203651. PMC 9611560. PMID 36296844.
- ^ Goenka, S; Sant, V; Sant, S (10 January 2014). "Graphene-based nanomaterials for drug delivery and tissue engineering". Journal of Controlled Release. 173: 75–88. doi:10.1016/j.jconrel.2013.10.017. PMID 24161530.
- ^ Gaharwar, A.K.; et al. (2013). Nanomaterials in tissue engineering : fabrication and applications. Oxford: Woodhead Publishing. ISBN 978-0-85709-596-1.
External links
[edit]- "What Are 2D Materials, and Why Do They Interest Scientists?" in Columbia News (March 6, 2024)
- "Twenty years of 2D materials" in Nature Physics (January 16, 2024)
Additional reading
[edit]- Xu, Yang; Cheng, Cheng; Du, Sichao; Yang, Jianyi; Yu, Bin; Luo, Jack; Yin, Wenyan; Li, Erping; Dong, Shurong; Ye, Peide; Duan, Xiangfeng (2016). "Contacts between Two- and Three-Dimensional Materials: Ohmic, Schottky, and p–n Heterojunctions". ACS Nano. 10 (5): 4895–4919. doi:10.1021/acsnano.6b01842. PMID 27132492.
- Briggs, Natalie; Subramanian, Shruti; Lin, Zhong; Li, Xufan; Zhang, Xiaotian; Zhang, Kehao; Xiao, Kai; Geohegan, David; Wallace, Robert; Chen, Long-Qing; Terrones, Mauricio; Ebrahimi, Aida; Das, Saptarshi; Redwing, Joan; Hinkle, Christopher; Momeni, Kasra; van Duin, Adri; Crespi, Vin; Kar, Swastik; Robinson, Joshua A. (2019). "A roadmap for electronic grade 2D materials". 2D Materials. 6 (2): 022001. Bibcode:2019TDM.....6b2001B. doi:10.1088/2053-1583/aaf836. OSTI 1503991. S2CID 188118830.
- Shahzad, F.; Alhabeb, M.; Hatter, C. B.; Anasori, B.; Man Hong, S.; Koo, C. M.; Gogotsi, Y. (2016). "Electromagnetic interference shielding with 2D transition metal carbides (MXenes)". Science. 353 (6304): 1137–1140. Bibcode:2016Sci...353.1137S. doi:10.1126/science.aag2421. PMID 27609888.
- "Graphene Uses & Applications". Graphenea. Retrieved 2014-04-13.
- cao, yameng; Robson, Alexander J.; Alharbi, Abdullah; Roberts, Jonathan; Woodhead, Christopher Stephen; Noori, Yasir Jamal; Gavito, Ramon Bernardo; Shahrjerdi, Davood; Roedig, Utz (2017). "Optical identification using imperfections in 2D materials". 2D Materials. 4 (4): 045021. arXiv:1706.07949. Bibcode:2017TDM.....4d5021C. doi:10.1088/2053-1583/aa8b4d. S2CID 35147364.
- Kolesnichenko, Pavel; Zhang, Qianhui; Zheng, Changxi; Fuhrer, Michael; Davis, Jeffrey (2021). "Multidimensional analysis of excitonic spectra of monolayers of tungsten disulphide: toward computer-aided identification of structural and environmental perturbations of 2D materials". Machine Learning: Science and Technology. 2 (2): 025021. arXiv:2003.01904. doi:10.1088/2632-2153/abd87c.
