User:LossIsNotMore/Uranium trioxide
| Uranium trioxide | |
|---|---|

|
solid γ-UO3 (gamma polymorph) oxygen diameters sharply reduced for visibility |
| General | |
| Systematic name | Uranium trioxide Uranium(VI) oxide |
| Other names | Uranyl oxide Uranic oxide |
| Molecular formula | UO3 (or O3U[1]) |
| CAS number | [1344-58-7] |
| Properties | |
| Molar mass | 286.2873 g/mol Commercial samples may have undergone isotope fractionation, and their molecular mass may be significantly different |
| Density and phase | 5.5 – 8.7 g/cm3 |
| Solubility (water) | Partially soluble |
| Solubility (dog lung fluid) | < 5 days[2] |
| Melting point | ~ 200 – 650 °C decomp. (s) |
| Structure | |
| Coordination geometry |
γ-UO3: [UO2]2+[UO4]2- |
| Crystal structure | Space group I41/amd (γ-UO3) |
| Hazards | |
| MSDS | UO3-MSDS |
| Main hazards | highly toxic: teratogen, immunotoxin, neurotoxin, genotoxin, nephrotoxin |
| Related compounds | |
| Other uranyl compounds | Uranyl nitrate Uranyl hydroxide Uranyl acetate |
| Related trioxides | Tungsten trioxide Molybdenum trioxide Chromium trioxide |
| Other uranium oxides | Uranium dioxide Triuranium octaoxide |
| Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox disclaimer and references | |
Uranium trioxide (UO3), also called uranyl oxide, uranium(VI) oxide, and uranic oxide, is the hexavalent oxide of uranium. The solid may be obtained by heating uranyl nitrate to 400 °C. Its most commonly encountered polymorph, γ-UO3, is a yellow-orange powder.
Uranium trioxide is a product of uranium metal combustion and corrosion. It is a poisonous genotoxin and teratogen in all its forms.
Production and use
[edit]There are three methods to generate uranium trioxide. As noted below, two are used industrially in the reprocessing of nuclear fuel and uranium enrichment.
- U3O8 can be oxidized at 500°C with oxygen.[3] Note that above 750°C even in 5 Atm O2 UO3 decomposes into U3O8.[4]
- Uranyl nitrate, (UO2(NO3)2·6H2O) can be heated to yield UO3. This occurs during the reprocessing of nuclear fuel. Fuel rods are dissolved in HNO3 to separate uranyl nitrate from plutonium and the fission products (the PUREX method). The pure uranyl nitrate is converted to solid UO3 by heating at 400 °C. After reduction with hydrogen (with other inert gas present) to uranium dioxide, the uranium can be used in new MOX fuel rods.
- Ammonium diuranate or sodium diuranate (Na2U2O7·6H2O) may be decomposed. Sodium diuranate, also known as yellowcake, is converted to uranium trioxide in the enrichment of uranium. Uranium dioxide and uranium tetrafluoride are intermediates in the process which ends in uranium hexafluoride.[5]
Uranium trioxide is shipped between processing facilities in the form of a gel.
Cameco Corporation, which operates at the world's largest uranium refinery at Blind River, Ontario, produces high-purity uranium trioxide.
Health and safety hazards
[edit]Like all hexavalent uranium compounds, UO3 is hazardous by inhalation, ingestion, and through skin contact. It is a poisonous, radioactive substance, which may cause shortness of breath, coughing, acute arterial lesions, and changes in the chromosomes of white blood cells and gonads leading to congenital malformations if inhaled.[2][6]
During nuclear fuel fabrication or reprocessing stages of a nuclear fuel cycle, it is possible for small particles of uranium oxides including UO3 to escape into the environment.[7] The extent of immediate inhalation intake of uranium oxides is inversely proportional to the size of particles inhaled; uranium oxide gases are absorbed immediately into the bloodstream.[2][8] Urine assay for UO3 exposure can be useful, provided that measurements are made soon after a known acute intake.[9][10] Treatment for UO3 inhalation primarily involves decorporation therapy.[11]
Chemistry and structure
[edit]Solid state structure
[edit]The only well characterized binary trioxide of any actinide is UO3, of which several polymorphs are known. Solid UO3 loses O2 on heating to give green-colored U3O8: reports of the decomposition temperature in air vary from 200–650 °C. Heating at 700 °C under H2 gives dark brown uranium dioxide (UO2), which is used in MOX nuclear fuel rods.
Alpha
[edit]
 |
The α (alpha) form: a layered solid where the 2D layers are linked by oxygen atoms (shown in red) |
Hydrated uranyl peroxide formed by the addition of hydrogen peroxide to an aqueous solution of uranyl nitrate when heated to 200-225 °C forms an amorphous uranium trioxide which on heating to 400-450 °C will form alpha-uranium trioxide.[4] It has been stated that the presence of nitrate will lower the temperature at which the exothermic change from the amorphous form to the alpha form occurs.[12] |
Beta
[edit]
 |
β (beta) UO3. This solid has a structure which defeats most attempts to describe it. |
This form can be formed by heating ammonium diuranate, while P.C. Debets and B.O. Loopstra, found four solid phases in the UO3-H2O-NH3 system that they could all be considered as being UO2(OH)2.H2O where some of the water has been replaced with ammonia.[13][14] No matter what the exact stiochiometry or structure, it was found that calcination at 500°C in air forms the beta form of uranium trioxide.[4] |
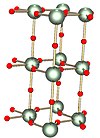
Gamma
[edit]
 |
The γ (gamma) form, with the different uranium environments in green and yellow |
The most frequently encountered polymorph is γ-UO3, whose x-ray structure has been solved from powder diffraction data. The compound crystallizes in the space group I41/amd with two uranium atoms in the asymmetric unit. Both are surrounded by somewhat distorted octahedra of oxygen atoms. One uranium atom has two closer and four more distant oxygen atoms whereas the other has four close and two more distant oxygen atoms as neighbors. Thus it is not incorrect to describe the structure as [UO2]2+[UO4]2- , that is uranyl uranate.[15] |
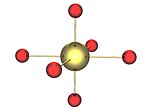 |
The environment of the uranium atoms shown as yellow in the gamma form |
 |
The chains of U2O2 rings in the gamma form in layers, alternate layers running at 90 degrees to each other. These chains are shown as containing the yellow uranium atoms, in an octahedral environment which are distorted towards square planar by an elongation of the axial oxygen-uranium bonds. |
Delta
[edit]
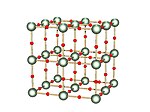 |
The delta (δ) form is a cubic solid where the oxygen atoms are arranged between the uranium atoms.[16] |
High pressure form
[edit]There is a high-pressure solid form with U2O2 and U3O3 rings in it.[17][18]
Hydrates
[edit]Several hydrates of uranium trioxide are known, e.g., UO3•6H2O.[4]
Molecular forms
[edit]While uranium trioxide is mostly encountered as a polymeric solid, work has been done on molecular forms in inert gas matrices and in the vapor phase, too.
Gas phase
[edit]Uranium trioxide is produced when uranium burns.[19][20][21] Uranyl ion contamination in uranium oxides has been detected in the residue of depleted uranium munitions fires.[22]
At elevated temperatures gaseous UO3 and O2 are in equilibrium with solid U3O8.[23][24][25][26]
- 1/3 U3O8(s) + 1/6 O2(g) UO3(g)
With increasing temperature the equilibrium is shifted to the right. This system has been studied at temperatures between 900 °C and 2200 Kelvin. The vapor pressure of monomeric UO3 is low but appreciable, about 10−5 mbar (1 mPa) at 980 °C, rising to 0.1 mbar (10 Pa) at 1400 °C, 0.34 mbar (34 Pa) at 1800 K, 1.9 mbar (193 Pa) at 2000 K, and 8.1 mbar (809 Pa) at 2200 K.[27][28] Small pieces of uranium burn at temperatures exceeding 2500 Kelvin.[29]
Matrix isolation
[edit]
Infrared spectroscopy of molecular UO3 isolated in an argon matrix indicates a T-shaped structure (point group C2v) for the molecule. This is in contrast to the commonly encountered D3h symmetry exhibited by most trioxides. From the force constants the authors deduct the U-O bond lengths to be between 1.76 and 1.79 angstroms (176 to 179 picometers).[30]
Calculations indicate that the point group of gaseous UO3 is C2v, with an axial bond length of 1.75 Å, an equatorial bond length of 1.83 Å and an angle of 161 ° between the axial oxygens. The more symmetrical D3h species is a saddle point, 49 kJ/mol above the C2v minimum. The authors invoke a second-order Jahn-Teller effect as explanation.[31]
Reactivity
[edit]Uranium trioxide reacts at 400 °C with freon-12 to form chlorine, phosgene, carbon dioxide and uranium(IV) fluoride. The freon-12 can be replaced with freon-11 which forms carbon tetrachloride instead of carbon dioxide. This is a case of a hard perhalogenated freon which is normally considered to be inert being converted chemically at a moderate temperature.[32]
2 CF2Cl2 + UO3 → UF4 + CO2 + COCl2 + Cl2
4 CF2Cl2 + UO3 → UF4 + 3COCl2 + CCl4 + Cl2
Uranium trioxide can be dissolved in a mixture of tributyl phosphate and thenoyltrifluoroacetone in supercritical carbon dioxide, ultrasound was employed during the dissolution.[33]
Corrosion of uranium metal
[edit]It has been reported that the corrosion of uranium in a silica rich aqueous solution forms both uranium dioxide and uranium trioxide.[34] Reports on the corrosion of uranium metal have been published by the Royal Society.[35][36]
Electrochemistry
[edit]The reversible insertion of magnesium cations into the lattice of uranium trioxide by cyclic voltammetry using a graphite electrode modifed with microscopic particles of the uranium oxide has been investigated. This experiment has also been done for U3O8. This is an example of electrochemistry of a solid modifed electrode, the experiment which used for uranium trioxide is related to a carbon paste electrode experiment. It is also possible to reduce uranium trioxide with sodium metal to form sodium uranium oxides.[37] The Journal of Solid State Electrochemistry is devoted to this type of electrochemistry.
It has been the case that it is possible to insert lithium ions and protons into the uranium trioxide lattice by electrochemical means, this is similar to the way that some rechargeable lithium ion batteries work. In these rechargeable cells one of the electrodes is a metal oxide which contains a metal such as cobalt which can be reduced, to maintain the electroneutrality for each electron which is added to the electrode material a lithium ion enters the lattice of this oxide electrode. [38][39](Li+) [40](H+)
Uranium oxides in ceramics
[edit]UO3-based ceramics become green or black when fired in a reducing atmosphere and yellow to orange when fired with oxygen. Orange-coloured Fiestaware is a well-known example of a product with a uranium-based glaze. UO3-has also been used in formulations of enamel, uranium glass, and porcelain.
Prior to 1960, UO3 was used as an agent of crystallization in crystalline coloured glazes. It is possible to determine with a Geiger counter if a glaze or glass was made from UO3.
Related anions and cations
[edit]Uranium oxide is amphoteric and reacts as acid and as a base, depending on the conditions.
- As an acid
- UO3 + H2O → UO42− + H+
Dissolving uranium oxide in a strong base like sodium hydroxide forms the doubly negatively charged uranate anion (UO42−). Uranates tend to agglomerate, forming diuranate, U2O72−</sup,> or other poly-uranates. Important diuranates include ammonium diuranate ((NH4)2U2O7), sodium diuranate (Na2U2O7) and magnesium diuranate (MgU2O7), which forms part of some yellowcakes. It is worth noting that uranates of the form M2UO4 do not contain UO42− ions, but rather flattened UO6 octahedra, containing a uranyl group and bridging oxygens.[41]
- As a base
- UO3 + H2O → UO22+ + OH−
Dissolving uranium oxide in a strong acid like sulfuric or nitric acid forms the double positive charged uranyl cation. The uranyl nitrate formed (UO2(NO3)2ˑ6H2O) is soluble in ethers, alcohols, ketones and esters; for example, tributylphosphate. This solubilty is used to separate uranium from other elements in nuclear reprocessing, which begins with the dissolution of nuclear fuel rods in nitric acid. The uranyl nitrate is then converted to uranium trioxide by heating.
From nitric acid one obtains uranyl nitrate, trans-UO2(NO3)2·2H2O, consisting of eight-coordinated uranium with two bidentate nitrato ligands and two water ligands as well as the familiar O=U=O core.
References
[edit]- ^ National Institute of Standards and Technology (2005) "Uranium trioxide" chemical data from NIST Standard Reference Database 69, NIST Chemistry WebBook
- ^ a b c Morrow, PE, Gibb FR, Beiter HD (1972). "Inhalation studies of uranium trioxide". Health Physics. 23: 273–280.
{{cite journal}}: CS1 maint: multiple names: authors list (link) abstract - ^ Sheft I, Fried S, Davidson N (1950). "Preparation of Uranium Trioxide". Journal of the American Chemical Society. 72: 2172–2173. doi:10.1021/ja01161a082.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c d Wheeler VJ, Dell RM, Wait E (1964). "Uranium trioxide and the UO3 hydrates". J. Inorganic Nuclear Chemistry. 26: 1829–1845. doi:10.1016/0022-1902(64)80007-5.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Dell RM, Wheeler VJ (1962). "Chemical Reactivity of Uranium Trioxide Part 1. — Conversion to U3O8, UO2 and UF4". Transactions of the Faraday Society. 58: 1590–1607. doi:10.1039/TF9625801590.
- ^ Sutton M, Burastero SR (2004). "Uranium(VI) solubility and speciation in simulated elemental human biological fluids". Chemical Research in Toxicology. 17: 1468–1480. doi:10.1021/tx049878k. full text
- ^ Schuenemana RA, Khaskelisa AI, Eastwooda D, van Ooijb WJ, Burggraf LW (2003). "Uranium oxide weathering: spectroscopy and kinetics". Journal of Nuclear Materials. 323: 8–17. doi:10.1016/j.jnucmat.2003.07.003.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Chazel V (1998). "Effect of U3O8 specific surface area on in vitro dissolution, biokinetics, and dose coefficients". Radiation Protection Dosimetry. 79: 39–42. full text
- ^ Ansoborlo E (1998). "Exposure implications for uranium aerosols formed at a new laser enrichment facility: application of the ICRP respiratory tract and systemic model". Radiation Protection Dosimetry. 79: 23–27. full text
- ^ Stradling N (2003). "Optimising monitoring regimens for inhaled uranium oxides". Radiation Protection Dosimetry. 105: 109–114. full text
- ^ Stradling GN (2000). "Treatment for Actinide-bearing Industrial Dusts and Aerosols". Radiation Protection Dosimetry. 87: 41–50. abstract, full text
- ^ Sato T (1963). Journal of Applied Chemistry. 13: 361.
{{cite journal}}: Missing or empty|title=(help) - ^ Debets PC, Loopstra BO (1963). Journal of Inorganic Nuclear Chemistry. 25: 945.
{{cite journal}}: Missing or empty|title=(help) - ^ Debets PC (1966). "The Structure of β-UO3". Acta Crystallographica. 21: 589.
- ^ Engmann R, de Wolff PM (1963). "The Crystal Structure of γ-UO3". Acta Crystallographica. 16: 993. doi:10.1107/S0365110X63002656.
- ^ Weller et al. (1988) Polyhedron, 7, 243-244.
- ^ Siegel S, Hoekstra HR, Sherry E (1966) Acta Crystallographica 20, 292-295.
- ^ Gmelin Handbuch (1982) U-C1, 129-135.
- ^ Rostker, B. (2000) "Depleted Uranium in the Gulf (II)" Environmental Exposure Reports Tech. Rep. No. 2000179-2 (Washington, DC: Special Assistant for Gulf War Illnesses, Department of Defense)
- ^ Army Environmental Policy Institute (June, 1995) Health and Environmental Consequences of Depleted Uranium Use in the US Army, Champaign, Illinois, June 1995.
- ^ U.S. Army Center for Health Promotion and Preventive Medicine (1998) Interim Summary, Total Uranium and Isotope Uranium Results, Operation Southern Watch, CHPPM Project No. 47-EM-8111-98.
- ^ Salbu, B. et al. (2005) "Oxidation states of uranium in depleted uranium particles from Kuwait," Journal of Environmental Radioactivity, 78, 125–135.
- ^ Ackermann RJ, Thorn RJ, Alexander C, Tetenbaum M (1960). "Free Energies of Formation of Gaseous Uranium, Molybdenum, and Tungsten Trioxides". Journal of Physical Chemistry. 64: 350–355. doi:10.1021/j100832a016.
{{cite journal}}: CS1 maint: multiple names: authors list (link) (page 350, 351, 352, 353, 354, 355) - ^ Wilson WB (1961). "High-Pressure High-Temperature Investigation of the Uranium-Oxygen System". Journal Inorganic Nuclear Chemistry. 19: 212–222. doi:10.1016/0022-1902(61)80109-7. full text
- ^ Chatillion C, Defoort F, Froment K (2005). "Mass spectrometric critical assessment of thermodynamic data for UO3(g)". Journal of Physics and Chemistry of Solids. 66: 379–383. doi:10.1016/j.jpcs.2004.06.077.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ H. Wanner and I. Forest, eds. (2004) "Gaseous uranium oxides" Chemical Thermodynamics of Uranium (Paris: OECD and French Nuclear Energy Agency) pp. 97-98
- ^ Ackermann RJ, Gilles PW, Thorn RJ (1956). "High-Temperature Thermodynamic Properties of Uranium Dioxide". Journal of Chemical Physics. 25: 1089. doi:10.1063/1.1743156.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Alexander CA (2005). "Volatilization of urania under strongly oxidizing conditions". Journal of Nuclear Materials. 346: 312–318. doi:10.1016/j.jnucmat.2005.07.013. full text
- ^ Mouradian and Baker (1963). "Burning Temperatures of Uranium and Zirconium in Air". Nuclear Science and Engineering. 15: 388–394.
- ^ Gabelnick SD, Reedy GT, Chasanov MG (1973). "Infrared spectra of matrix-isolated uranium oxide species. II: Spectral interpretation and structure of UO3". Journal of Chemical Physics. 59: 6397–6404. doi:10.1063/1.1680018.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pyykkö P, Li J (1994). "Quasirelativistic pseudopotential study of species isoelectronic to uranyl and the equatorial coordination of uranyl". Journal of Physical Chemistry. 98: 4809–4813.
- ^ Booth HS, Krasny-Ergen W, Heath RE (1946). "Uranium Tetrafluoride". Journal of the American Chemical Society. 68: 1969–1970. doi:10.1021/ja01214a028.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Trofimov TI, Samsonov MD, Lee SC, Myasoedov BF, Wai CM (2001). "Dissolution of uranium oxides in supercritical carbon dioxide containing tri-n-butyl phosphate and thenoyltrifluoroacetone". Mendeleev Communications. 11: 129–130. doi:10.1070/MC2001v011n04ABEH001468.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Trueman ER, Black S, Read D, Hodson ME (2003) "Alteration of Depleted Uranium Metal" Goldschmidt Conference Abstracts, p. A493 abstract
- ^ Ander L, Smith B (2002) "Annexe F: Groundwater transport modelling" The health hazards of depleted uranium munitions, part II (London: The Royal Society)
- ^ Smith B (2002) "Annexe G: Corrosion of DU and DU alloys: a brief discussion and review" The health hazards of depleted uranium munitions, part II (London: The Royal Society)
- ^ Dueber RE, Bond AM, Dickens PG (1992) Journal of the Electrochemical Society 139, 2363-2371.
- ^ Dickens PG, Lawrence SD, Penny DJ, Powell AV (1989) Solid State Ionics 32/33, 77-83.
- ^ P.G. Dickens, S.D Lawrence and M.T. Weller (1985) Mat. Res. Bull. 20, 635.
- ^ P.G. Dickens, S.V. Hawke and M.T. Weller (1984) Mat. Res. Bull. 19, 543.
- ^ Cotton, Simon (1991). Lanthanides and Actinides. New York: Oxford University Press. p. 128. (page 126, 127, 128)
- Rauh EG, Ackermann RJ (1974). "First ionization potentials of some refractory oxide vapors". J Chem Phys. 60: 1396.


