Citric acid
| |||

| |||
| Names | |||
|---|---|---|---|
| IUPAC name
Citric acid[1]
| |||
| Preferred IUPAC name
2-Hydroxypropane-1,2,3-tricarboxylic acid[1] | |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChEBI | |||
| ChEMBL | |||
| ChemSpider | |||
| DrugBank | |||
| ECHA InfoCard | 100.000.973 | ||
| EC Number |
| ||
| E number | E330 (antioxidants, ...) | ||
| KEGG | |||
PubChem CID
|
|||
| RTECS number |
| ||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
| HOC(CO2H)(CH2CO2H)2 | |||
| Molar mass | 192.123 g/mol (anhydrous), 210.14 g/mol (monohydrate)[2] | ||
| Appearance | white solid | ||
| Odor | Odorless | ||
| Density | 1.665 g/cm3 (anhydrous) 1.542 g/cm3 (18 °C, monohydrate) | ||
| Melting point | 156 °C (313 °F; 429 K) | ||
| Boiling point | 310 °C (590 °F; 583 K) decomposes from 175 °C[3] | ||
| 54% w/w (10 °C) 59.2% w/w (20 °C) 64.3% w/w (30 °C) 68.6% w/w (40 °C) 70.9% w/w (50 °C) 73.5% w/w (60 °C) 76.2% w/w (70 °C) 78.8% w/w (80 °C) 81.4% w/w (90 °C) 84% w/w (100 °C)[4] | |||
| Solubility | Soluble in acetone, alcohol, ether, ethyl acetate, DMSO Insoluble in C 6H 6, CHCl3, CS2, toluene[3] | ||
| Solubility in ethanol | 62 g/100 g (25 °C)[3] | ||
| Solubility in amyl acetate | 4.41 g/100 g (25 °C)[3] | ||
| Solubility in diethyl ether | 1.05 g/100 g (25 °C)[3] | ||
| Solubility in 1,4-dioxane | 35.9 g/100 g (25 °C)[3] | ||
| log P | −1.64 | ||
| Acidity (pKa) | pKa1 = 3.13[5] pKa2 = 4.76[5] pKa3 = 6.39,[6] 6.40[7] pKa4 = 14.4[8] | ||
Refractive index (nD)
|
1.493–1.509 (20 °C)[4] 1.46 (150 °C)[3] | ||
| Viscosity | 6.5 cP (50% aq. sol.)[4] | ||
| Structure | |||
| Monoclinic | |||
| Thermochemistry | |||
Heat capacity (C)
|
226.51 J/(mol·K) (26.85 °C)[9] | ||
Std molar
entropy (S⦵298) |
252.1 J/(mol·K)[9] | ||
Std enthalpy of
formation (ΔfH⦵298) |
−1543.8 kJ/mol[4] | ||
| 1985.3 kJ/mol (474.5 kcal/mol, 2.47 kcal/g),[4] 1960.6 kJ/mol[9] 1972.34 kJ/mol (471.4 kcal/mol, 2.24 kcal/g) (monohydrate)[4] | |||
| Pharmacology | |||
| A09AB04 (WHO) | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Skin and eye irritant | ||
| GHS labelling: | |||
  [5] [5]
| |||
| Warning | |||
| H290, H319, H315[5] | |||
| P305+P351+P338[5] | |||
| NFPA 704 (fire diamond) | |||
| Flash point | 155 °C (311 °F; 428 K) | ||
| 345 °C (653 °F; 618 K) | |||
| Explosive limits | 8%[5] | ||
| Lethal dose or concentration (LD, LC): | |||
LD50 (median dose)
|
3000 mg/kg (rats, oral) | ||
| Safety data sheet (SDS) | HMDB | ||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Citric acid is an organic compound with the chemical formula HOC(CO2H)(CH2CO2H)2.[10] It is a colorless weak organic acid.[10] It occurs naturally in citrus fruits. In biochemistry, it is an intermediate in the citric acid cycle, which occurs in the metabolism of all aerobic organisms.[10]
More than two million tons of citric acid are manufactured every year. It is used widely as an acidifier, as a flavoring, and a chelating agent.[11]
A citrate is a derivative of citric acid; that is, the salts, esters, and the polyatomic anion found in solutions and salts of citric acid. An example of the former, a salt is trisodium citrate; an ester is triethyl citrate. When citrate trianion is part of a salt, the formula of the citrate trianion is written as C
6H
5O3−
7 or C
3H
5O(COO)3−
3.
Natural occurrence and industrial production[edit]

Citric acid occurs in a variety of fruits and vegetables, most notably citrus fruits. Lemons and limes have particularly high concentrations of the acid; it can constitute as much as 8% of the dry weight of these fruits (about 47 g/L in the juices[12]).[a] The concentrations of citric acid in citrus fruits range from 0.005 mol/L for oranges and grapefruits to 0.30 mol/L in lemons and limes; these values vary within species depending upon the cultivar and the circumstances under which the fruit was grown.
Citric acid was first isolated in 1784 by the chemist Carl Wilhelm Scheele, who crystallized it from lemon juice.[13][14]
Industrial-scale citric acid production first began in 1890 based on the Italian citrus fruit industry, where the juice was treated with hydrated lime (calcium hydroxide) to precipitate calcium citrate, which was isolated and converted back to the acid using diluted sulfuric acid.[15] In 1893, C. Wehmer discovered Penicillium mold could produce citric acid from sugar.[16] However, microbial production of citric acid did not become industrially important until World War I disrupted Italian citrus exports.
In 1917, American food chemist James Currie discovered that certain strains of the mold Aspergillus niger could be efficient citric acid producers,[17] and the pharmaceutical company Pfizer began industrial-level production using this technique two years later, followed by Citrique Belge in 1929. In this production technique, which is still the major industrial route to citric acid used today, cultures of Aspergillus niger are fed on a sucrose or glucose-containing medium to produce citric acid. The source of sugar is corn steep liquor, molasses, hydrolyzed corn starch, or other inexpensive, carbohydrate solution.[18] After the mold is filtered out of the resulting suspension, citric acid is isolated by precipitating it with calcium hydroxide to yield calcium citrate salt, from which citric acid is regenerated by treatment with sulfuric acid, as in the direct extraction from citrus fruit juice.
In 1977, a patent was granted to Lever Brothers for the chemical synthesis of citric acid starting either from aconitic or isocitrate (also called alloisocitrate) calcium salts under high pressure conditions; this produced citric acid in near quantitative conversion under what appeared to be a reverse, non-enzymatic Krebs cycle reaction.[19]
Global production was in excess of 2,000,000 tons in 2018.[20] More than 50% of this volume was produced in China. More than 50% was used as an acidity regulator in beverages, some 20% in other food applications, 20% for detergent applications, and 10% for applications other than food, such as cosmetics, pharmaceuticals, and in the chemical industry.[15]
Chemical characteristics[edit]
Citric acid can be obtained as an anhydrous (water-free) form or as a monohydrate. The anhydrous form crystallizes from hot water, while the monohydrate forms when citric acid is crystallized from cold water. The monohydrate can be converted to the anhydrous form at about 78 °C. Citric acid also dissolves in absolute (anhydrous) ethanol (76 parts of citric acid per 100 parts of ethanol) at 15 °C. It decomposes with loss of carbon dioxide above about 175 °C.
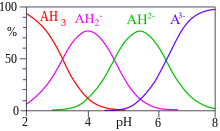
Citric acid is a tribasic acid, with pKa values, extrapolated to zero ionic strength, of 3.128, 4.761, and 6.396 at 25 °C.[21] The pKa of the hydroxyl group has been found, by means of 13C NMR spectroscopy, to be 14.4.[22] The speciation diagram shows that solutions of citric acid are buffer solutions between about pH 2 and pH 8. In biological systems around pH 7, the two species present are the citrate ion and mono-hydrogen citrate ion. The SSC 20X hybridization buffer is an example in common use.[23][24] Tables compiled for biochemical studies are available.[25]
Conversely, the pH of a 1 mM solution of citric acid will be about 3.2. The pH of fruit juices from citrus fruits like oranges and lemons depends on the citric acid concentration, with a higher concentration of citric acid resulting in a lower pH.
Acid salts of citric acid can be prepared by careful adjustment of the pH before crystallizing the compound. See, for example, sodium citrate.
The citrate ion forms complexes with metallic cations. The stability constants for the formation of these complexes are quite large because of the chelate effect. Consequently, it forms complexes even with alkali metal cations. However, when a chelate complex is formed using all three carboxylate groups, the chelate rings have 7 and 8 members, which are generally less stable thermodynamically than smaller chelate rings. In consequence, the hydroxyl group can be deprotonated, forming part of a more stable 5-membered ring, as in ammonium ferric citrate, [NH+4]5Fe3+(C6H4O4−7)2·2H2O.[26]
Citric acid can be esterified at one or more of its three carboxylic acid groups to form any of a variety of mono-, di-, tri-, and mixed esters.[27]
Biochemistry[edit]
Citric acid cycle[edit]
Citrate is an intermediate in the citric acid cycle, also known as the TCA (TriCarboxylic Acid) cycle or the Krebs cycle, a central metabolic pathway for animals, plants, and bacteria. Citrate synthase catalyzes the condensation of oxaloacetate with acetyl CoA to form citrate. Citrate then acts as the substrate for aconitase and is converted into aconitic acid. The cycle ends with regeneration of oxaloacetate. This series of chemical reactions is the source of two-thirds of the food-derived energy in higher organisms. Hans Adolf Krebs received the 1953 Nobel Prize in Physiology or Medicine for the discovery.
Some bacteria (notably E. coli) can produce and consume citrate internally as part of their TCA cycle, but are unable to use it as food, because they lack the enzymes required to import it into the cell. After tens of thousands of evolutions in a minimal glucose medium that also contained citrate during Richard Lenski's Long-Term Evolution Experiment, a variant E. coli evolved with the ability to grow aerobically on citrate. Zachary Blount, a student of Lenski's, and colleagues studied these "Cit+" E. coli[28][29] as a model for how novel traits evolve. They found evidence that, in this case, the innovation was caused by a rare duplication mutation due to the accumulation of several prior "potentiating" mutations, the identity and effects of which are still under study. The evolution of the Cit+ trait has been considered a notable example of the role of historical contingency in evolution.
Other biological roles[edit]
Citrate can be transported out of the mitochondria and into the cytoplasm, then broken down into acetyl-CoA for fatty acid synthesis, and into oxaloacetate. Citrate is a positive modulator of this conversion, and allosterically regulates the enzyme acetyl-CoA carboxylase, which is the regulating enzyme in the conversion of acetyl-CoA into malonyl-CoA (the commitment step in fatty acid synthesis). In short, citrate is transported into the cytoplasm, converted into acetyl-CoA, which is then converted into malonyl-CoA by acetyl-CoA carboxylase, which is allosterically modulated by citrate.
High concentrations of cytosolic citrate can inhibit phosphofructokinase, the catalyst of a rate-limiting step of glycolysis. This effect is advantageous: high concentrations of citrate indicate that there is a large supply of biosynthetic precursor molecules, so there is no need for phosphofructokinase to continue to send molecules of its substrate, fructose 6-phosphate, into glycolysis. Citrate acts by augmenting the inhibitory effect of high concentrations of ATP, another sign that there is no need to carry out glycolysis.[30]
Citrate is a vital component of bone, helping to regulate the size of apatite crystals.[31]
Applications[edit]
Food and drink[edit]

Because it is one of the stronger edible acids, the dominant use of citric acid is as a flavoring and preservative in food and beverages, especially soft drinks and candies.[15] Within the European Union it is denoted by E number E330. Citrate salts of various metals are used to deliver those minerals in a biologically available form in many dietary supplements. Citric acid has 247 kcal per 100 g.[32] In the United States the purity requirements for citric acid as a food additive are defined by the Food Chemicals Codex, which is published by the United States Pharmacopoeia (USP).
Citric acid can be added to ice cream as an emulsifying agent to keep fats from separating, to caramel to prevent sucrose crystallization, or in recipes in place of fresh lemon juice. Citric acid is used with sodium bicarbonate in a wide range of effervescent formulae, both for ingestion (e.g., powders and tablets) and for personal care (e.g., bath salts, bath bombs, and cleaning of grease). Citric acid sold in a dry powdered form is commonly sold in markets and groceries as "sour salt", due to its physical resemblance to table salt. It has use in culinary applications, as an alternative to vinegar or lemon juice, where a pure acid is needed. Citric acid can be used in food coloring to balance the pH level of a normally basic dye.[citation needed]
Cleaning and chelating agent[edit]

Citric acid is an excellent chelating agent, binding metals by making them soluble. It is used to remove and discourage the buildup of limescale from boilers and evaporators.[15] It can be used to treat water, which makes it useful in improving the effectiveness of soaps and laundry detergents. By chelating the metals in hard water, it lets these cleaners produce foam and work better without need for water softening. Citric acid is the active ingredient in some bathroom and kitchen cleaning solutions. A solution with a six percent concentration of citric acid will remove hard water stains from glass without scrubbing. Citric acid can be used in shampoo to wash out wax and coloring from the hair. Illustrative of its chelating abilities, citric acid was the first successful eluant used for total ion-exchange separation of the lanthanides, during the Manhattan Project in the 1940s.[35] In the 1950s, it was replaced by the far more efficient[36] EDTA.
In industry, it is used to dissolve rust from steel, and to passivate stainless steels.[37]
Cosmetics, pharmaceuticals, dietary supplements, and foods[edit]
Citric acid is used as an acidulant in creams, gels, and liquids. Used in foods and dietary supplements, it may be classified as a processing aid if it was added for a technical or functional effect (e.g. acidulent, chelator, viscosifier, etc.). If it is still present in insignificant amounts, and the technical or functional effect is no longer present, it may be exempt from labeling <21 CFR §101.100(c)>.
Citric acid is an alpha hydroxy acid and is an active ingredient in chemical skin peels.[38]
Citric acid is commonly used as a buffer to increase the solubility of brown heroin.[39]
Citric acid is used as one of the active ingredients in the production of facial tissues with antiviral properties.[40]
Other uses[edit]
The buffering properties of citrates are used to control pH in household cleaners and pharmaceuticals.
Citric acid is used as an odorless alternative to white vinegar for fabric dyeing with acid dyes.
Sodium citrate is a component of Benedict's reagent, used for both qualitative and quantitative identification of reducing sugars.[41]
Citric acid can be used as an alternative to nitric acid in passivation of stainless steel.[42]
Citric acid can be used as a lower-odor stop bath as part of the process for developing photographic film. Photographic developers are alkaline, so a mild acid is used to neutralize and stop their action quickly, but commonly used acetic acid leaves a strong vinegar odor in the darkroom.[43]
Citric acid/potassium-sodium citrate can be used as a blood acid regulator. The citric acid is included to improve palatability[44]
Citric acid is an excellent soldering flux,[45] either dry or as a concentrated solution in water. It should be removed after soldering, especially with fine wires, as it is mildly corrosive. It dissolves and rinses quickly in hot water.
Alkali citrate can be used as an inhibitor of kidney stones by increasing urine citrate levels, useful for prevention of calcium stones, and increasing urine pH, useful for preventing uric acid and cystine stones.[46]
Synthesis of other organic compounds[edit]
Citric acid is a versatile precursor to many other organic compounds. Dehydration routes give itaconic acid and its anhydride.[47] Citraconic acid can be produced via thermal isomerization of itaconic acid anhydride.[48] The required itaconic acid anhydride is obtained by dry distillation of citric acid. Aconitic acid can be synthesized by dehydration of citric acid using sulfuric acid:[49]
- (HO2CCH2)2C(OH)CO2H → HO2CCH=C(CO2H)CH2CO2H + H2O
Acetonedicarboxylic acid can also be prepared by decarboxylation of citric acid in fuming sulfuric acid.[50]
Safety[edit]
Although a weak acid, exposure to pure citric acid can cause adverse effects. Inhalation may cause cough, shortness of breath, or sore throat. Over-ingestion may cause abdominal pain and sore throat. Exposure of concentrated solutions to skin and eyes can cause redness and pain.[51] Long-term or repeated consumption may cause erosion of tooth enamel.[51][52][53]
Compendial status[edit]
See also[edit]
- The closely related acids isocitric acid, aconitic acid, and propane-1,2,3-tricarboxylic acid (tricarballylic acid, carballylic acid)
- Acids in wine
Explanatory notes[edit]
- ^ This still does not make the lemon particularly strongly acidic. This is because, as a weak acid, most of the acid molecules are not dissociated so not contributing to acidity inside the lemon or its juice.
References[edit]
- ^ a b International Union of Pure and Applied Chemistry (2014). Nomenclature of Organic Chemistry: IUPAC Recommendations and Preferred Names 2013. The Royal Society of Chemistry. p. 747. doi:10.1039/9781849733069. ISBN 978-0-85404-182-4.
- ^ CID 22230 from PubChem
- ^ a b c d e f g "citric acid". chemister.ru. Archived from the original on November 29, 2014. Retrieved June 1, 2014.
- ^ a b c d e f CID 311 from PubChem
- ^ a b c d e f Fisher Scientific, Citric acid. Retrieved on 2014-06-02.
- ^ "Data for Biochemical Research". ZirChrom Separations, Inc. Retrieved January 11, 2012.
- ^ "Ionization Constants of Organic Acids". Michigan State University. Retrieved January 11, 2012.
- ^ Silva, Andre M. N.; Kong, Xiaole; Hider, Robert C. (2009). "Determination of the pKa value of the hydroxyl group in the α-hydroxycarboxylates citrate, malate and lactate by 13C NMR: implications for metal coordination in biological systems". BioMetals. 22 (5): 771–778. doi:10.1007/s10534-009-9224-5. ISSN 0966-0844.
- ^ a b c Citric acid in Linstrom, Peter J.; Mallard, William G. (eds.); NIST Chemistry WebBook, NIST Standard Reference Database Number 69, National Institute of Standards and Technology, Gaithersburg (MD) (retrieved 2014-06-02)
- ^ a b c "Citric acid | C6H8O7 - PubChem". Archived from the original on January 19, 2022. Retrieved December 19, 2021.
- ^ Apleblat, Alexander (2014). Citric acid. Springer. ISBN 978-3-319-11232-9.
- ^ Penniston KL, Nakada SY, Holmes RP, Assimos DG (2008). "Quantitative Assessment of Citric Acid in Lemon Juice, Lime Juice, and Commercially-Available Fruit Juice Products". Journal of Endourology. 22 (3): 567–570. doi:10.1089/end.2007.0304. PMC 2637791. PMID 18290732.
- ^ Scheele, Carl Wilhelm (1784). "Anmärkning om Citron-saft, samt sätt at crystallisera densamma" [Note about lemon juice, as well as ways to crystallize it]. Kungliga Vetenskaps Academiens Nya Handlingar [New Proceedings of the Royal Academy of Science]. 2nd series (in Swedish). 5: 105–109.
- ^ Graham, Thomas (1842). Elements of chemistry, including the applications of the science in the arts. Hippolyte Baillière, foreign bookseller to the Royal College of Surgeons, and to the Royal Society, 219, Regent Street. p. 944. Retrieved June 4, 2010.
- ^ a b c d Verhoff, Frank H.; Bauweleers, Hugo (2014). "Citric Acid". Ullmann's Encyclopedia of Industrial Chemistry. Weinheim: Wiley-VCH. doi:10.1002/14356007.a07_103.pub3. ISBN 978-3527306732.
- ^ H. Benninga (June 30, 1990). A History of Lactic Acid Making: A Chapter in the History of Biotechnology. Springer Science & Business Media. pp. 140–5. ISBN 978-0-7923-0625-2.
- ^ Currie, James (1917). The Journal of Biological Chemistry. American Society for Biochemistry and Molecular Biology. pp. 15–27.
- ^ Lotfy, Walid A.; Ghanem, Khaled M.; El-Helow, Ehab R. (2007). "Citric acid production by a novel Aspergillus niger isolate: II. Optimization of process parameters through statistical experimental designs". Bioresource Technology. 98 (18): 3470–3477. doi:10.1016/j.biortech.2006.11.032. PMID 17317159.
- ^ US 4056567-V.Lamberti and E.Gutierrez
- ^ "Global Citric Acid Markets Report, 2011-2018 & 2019-2024". prnewswire.com. March 19, 2019. Retrieved October 28, 2019.
- ^ Goldberg, Robert N.; Kishore, Nand; Lennen, Rebecca M. (2002). "Thermodynamic Quantities for the Ionization Reactions of Buffers". J. Phys. Chem. Ref. Data. 31 (1): 231–370. Bibcode:2002JPCRD..31..231G. doi:10.1063/1.1416902. S2CID 94614267.
- ^ Silva, Andre M. N.; Kong, Xiaole; Hider, Robert C. (2009). "Determination of the pKa value of the hydroxyl group in the α-hydroxycarboxylates citrate, malate and lactate by 13C NMR: implications for metal coordination in biological systems". Biometals. 22 (5): 771–778. doi:10.1007/s10534-009-9224-5. PMID 19288211. S2CID 11615864.
- ^ "SSC - OpenWetWare".
- ^ Maniatis, T.; Fritsch, E. F.; Sambrook, J. 1982. Molecular Cloning: A Laboratory Manual. Cold Spring Harbor Laboratory, Cold Spring Harbor, NY.
- ^ Gomori, G. (1955). "16 Preparation of buffers for use in enzyme studies". Methods in Enzymology Volume 1. Vol. 1. pp. 138–146. doi:10.1016/0076-6879(55)01020-3. ISBN 9780121818012.
- ^ Matzapetakis, M.; Raptopoulou, C. P.; Tsohos, A.; Papaefthymiou, V.; Moon, S. N.; Salifoglou, A. (1998). "Synthesis, Spectroscopic and Structural Characterization of the First Mononuclear, Water Soluble Iron−Citrate Complex, (NH4)5Fe(C6H4O7)2·2H2O". J. Am. Chem. Soc. 120 (50): 13266–13267. doi:10.1021/ja9807035.
- ^ Bergeron, Raymond J.; Xin, Meiguo; Smith, Richard E.; Wollenweber, Markus; McManis, James S.; Ludin, Christian; Abboud, Khalil A. (1997). "Total synthesis of rhizoferrin, an iron chelator". Tetrahedron. 53 (2): 427–434. doi:10.1016/S0040-4020(96)01061-7.
- ^ Powell, Alvin (February 14, 2014). "59,000 generations of bacteria, plus freezer, yield startling results". phys.org. Retrieved April 13, 2017.
- ^ Blount, Z. D.; Borland, C. Z.; Lenski, R. E. (June 4, 2008). "Historical contingency and the evolution of a key innovation in an experimental population of Escherichia coli". Proceedings of the National Academy of Sciences. 105 (23): 7899–7906. Bibcode:2008PNAS..105.7899B. doi:10.1073/pnas.0803151105. PMC 2430337. PMID 18524956.
- ^ Stryer, Lubert; Berg, Jeremy; Tymoczko, John (2003). "Section 16.2: The Glycolytic Pathway Is Tightly Controlled". Biochemistry (5. ed., international ed., 3. printing ed.). New York: Freeman. ISBN 978-0716746843.
- ^ Hu, Y.-Y.; Rawal, A.; Schmidt-Rohr, K. (December 2010). "Strongly bound citrate stabilizes the apatite nanocrystals in bone". Proceedings of the National Academy of Sciences. 107 (52): 22425–22429. Bibcode:2010PNAS..10722425H. doi:10.1073/pnas.1009219107. PMC 3012505. PMID 21127269.
- ^ Greenfield, Heather; Southgate, D.A.T. (2003). Food Composition Data: Production, Management and Use. Rome: FAO. p. 146. ISBN 9789251049495.
- ^ Xiang Hao, Yongge Wei, Shiwei Zhang (2001): "Synthesis, crystal structure and magnetic property of a binuclear iron(III) citrate complex". Transition Metal Chemistry, volume 26, issue 4, pages 384–387. doi:10.1023/A:1011055306645
- ^ Shweky, Itzhak; Bino, Avi; Goldberg, David P.; Lippard, Stephen J. (1994). "Syntheses, Structures, and Magnetic Properties of Two Dinuclear Iron(III) Citrate Complexes". Inorganic Chemistry. 33 (23): 5161–5162. doi:10.1021/ic00101a001.
- ^ JOHNSON, WARREN C.; QUILL, LAURENCE L.; DANIELS, FARRINGTON (September 1, 1947). "Rare Earths Separation Developed on Manhattan Project". Chemical & Engineering News Archive. 25 (35): 2494. doi:10.1021/cen-v025n035.p2494. ISSN 0009-2347.
- ^ Saleem, Muhammad Hamzah; Ali, Shafaqat; Rehman, Muzammal; Rizwan, Muhammad; Kamran, Muhammad; Mohamed, Ibrahim A.A.; Khan, Zaid; Bamagoos, Atif A.; Alharby, Hesham F.; Hakeem, Khalid Rehman; Liu, Lijun (August 1, 2020). "Individual and combined application of EDTA and citric acid assisted phytoextraction of copper using jute (Corchorus capsularis L.) seedlings". Environmental Technology & Innovation. 19: 100895. doi:10.1016/j.eti.2020.100895. ISSN 2352-1864. S2CID 219432688.
- ^ "ASTM A967 / A967M - 17 Standard Specification for Chemical Passivation Treatments for Stainless Steel Parts". www.astm.org.
- ^ Tang, Sheau-Chung; Yang, Jen-Hung (April 10, 2018). "Dual Effects of Alpha-Hydroxy Acids on the Skin". Molecules. 23 (4): 863. doi:10.3390/molecules23040863. ISSN 1420-3049. PMC 6017965. PMID 29642579.
- ^ Strang J, Keaney F, Butterworth G, Noble A, Best D (April 2001). "Different forms of heroin and their relationship to cook-up techniques: data on, and explanation of, use of lemon juice and other acids". Subst Use Misuse. 36 (5): 573–88. doi:10.1081/ja-100103561. PMID 11419488. S2CID 8516420.
- ^ "Tissues that fight germs". CNN. July 14, 2004. Retrieved May 8, 2008.
- ^ Chen, Wei; Abramowitz, Matthew K. (February 2014). "Treatment of Metabolic Acidosis in Patients With CKD". American Journal of Kidney Diseases. 63 (2): 311–317. doi:10.1053/j.ajkd.2013.06.017. ISSN 0272-6386. PMC 3946919. PMID 23932089.
- ^ "Pickling and Passivating Stainless Steel" (PDF). Euro-inox.org. Archived from the original (PDF) on September 12, 2012. Retrieved 2013-01-01.
- ^ Anchell, Steve. "The Darkroom Cookbook: 3rd Edition (Paperback)". Focal Press. Retrieved January 1, 2013.
- ^ PubChem. "Sodium citrate". pubchem.ncbi.nlm.nih.gov. Retrieved August 2, 2021.
- ^ "An Investigation of the Chemistry of Citric Acid in Military Soldering Applications" (PDF). June 19, 1995. Archived (PDF) from the original on March 15, 2020.
- ^ Berg, C.; Larsson, L.; Tiselius, H. G. (1992). "The effects of a single evening dose of alkaline citrate on urine composition and calcium stone formation". The Journal of Urology. 148 (3 Pt 2): 979–985. doi:10.1016/s0022-5347(17)36795-2. ISSN 0022-5347. PMID 1507355.
- ^ R. L. Shriner; S. G. Ford; l. J. Roll (1931). "Itaconic anhydride and itaconic acid". Org. Synth. 11: 70. doi:10.15227/orgsyn.011.0070.
- ^ R. L. Shriner; S. G. Ford; l. J. Roll (1931). "Citraconic Anhydride and Citraconic Acid". Org. Synth. 28: 28. doi:10.15227/orgsyn.011.0028.
- ^ Bruce, W. F. (1937). "Aconitic Acid". Organic Syntheses. 17: 1. doi:10.15227/orgsyn.017.0001.
- ^ Roger Adams; H. M. Chiles; C. F. Rassweiler (1925). "Acetonedicarboxylic Acid". Organic Syntheses. 5: 5. doi:10.15227/orgsyn.005.0005.
- ^ a b "Citric acid". International Chemical Safety Cards. NIOSH. September 18, 2018. Archived from the original on July 12, 2018. Retrieved September 9, 2017.
- ^ J. Zheng; F. Xiao; L. M. Qian; Z. R. Zhou (December 2009). "Erosion behavior of human tooth enamel in citric acid solution". Tribology International. 42 (11–12): 1558–1564. doi:10.1016/j.triboint.2008.12.008.
- ^ "Effect of Citric Acid on Tooth Enamel".
- ^ British Pharmacopoeia Commission Secretariat (2009). "Index, BP 2009" (PDF). Archived from the original (PDF) on April 11, 2009. Retrieved February 4, 2010.
- ^ "Japanese Pharmacopoeia, Fifteenth Edition" (PDF). 2006. Archived from the original (PDF) on July 22, 2011. Retrieved 4 February 2010.