Microsporidia
| Microsporidia | |
|---|---|

| |
| Sporoblast of Fibrillanosema crangonycis | |
| Scientific classification | |
| Domain: | Eukaryota |
| (unranked): | Opisthosporidia |
| Phylum: | Microsporidia Balbiani, 1882[1] |
| Classes and orders | |
| Synonyms | |
Microsporidia are a group of spore-forming unicellular parasites. They were once considered protozoans or protists, but are now known to be fungi,[6] or a sister group to fungi.[7] They have recently been discovered to infect on Coleoptera a large scale, in a 2017 Cornell study. Loosely 1500 of the probably more than one million[8] species are named. Microsporidia are restricted to animal hosts, and all major groups of animals host microsporidia. Most infect insects, but they are also responsible for common diseases of crustaceans and fish. The named species of microsporidia usually infect one host species or a group of closely related taxa. Approximately 10 percent of the species are parasites of vertebrates —several species, most of which are opportunistic, can infect humans, in whom they can cause microsporidiosis.
After infection they influence their hosts in various ways and all organs and tissues are invaded, though generally by different species of microsporidia. Some species are lethal, and a few are used in biological control of insect pests. Parasitic castration, gigantism, or change of host sex are all potential effects of microsporidian parasitism (in insects). In the most advanced cases of parasitism the microsporidium rules the host cell completely and controls its metabolism and reproduction, forming a xenoma.[9]
Replication takes place within the host's cells, which are infected by means of unicellular spores. These vary from 1–40 μm, making them some of the smallest eukaryotes.[citation needed] Microsporidia that infect mammals are 1.0–4.0 μm.[10] They also have the smallest eukaryotic genomes.
The terms "microsporidium" (pl. "microsporidia") and "microsporidian" are used as vernacular names for members of the group. The name Microsporidium Balbiani, 1884[11] is also used as a catchall genus for incertae sedis members.[12]

Morphology

Microsporidia lack mitochondria, instead possessing mitosomes. They also lack motile structures, such as flagella.
Microsporidia produce highly resistant spores, capable of surviving outside their host for up to several years. Spore morphology is useful in distinguishing between different species. Spores of most species are oval or pyriform, but rod-shaped or spherical spores are not unusual. A few genera produce spores of unique shape for the genus.
The spore is protected by a wall, consisting of three layers:
- an outer electron-dense exospore
- a median, wide and seemingly structureless endospore, containing chitin
- a thin internal plasma membrane
In most cases there are two closely associated nuclei, forming a diplokaryon, but sometimes there is only one.
The anterior half of the spore contains a harpoon-like apparatus with a long, thread-like polar filament, which is coiled up in the posterior half of the spore. The anterior part of the polar filament is surrounded by a polaroplast, a lamella of membranes. Behind the polar filament, there is a posterior vacuole.[9]
Infection
In the gut of the host the spore germinates, it builds up osmotic pressure until its rigid wall ruptures at its thinnest point at the apex. The posterior vacuole swells, forcing the polar filament to rapidly eject the infectious content into the cytoplasm of the potential host. Simultaneously the material of the filament is rearranged to form a tube which functions as a hypodermic needle and penetrates the gut epithelium.
Once inside the host cell, a sporoplasm grows, dividing or forming a multinucleate plasmodium, before producing new spores. The life cycle varies considerably. Some have a simple asexual life cycle,[14] while others have a complex life cycle involving multiple hosts and both asexual and sexual reproduction. Different types of spores may be produced at different stages, probably with different functions including autoinfection (transmission within a single host).
Medical implications
In animals and humans, microsporidia often cause chronic, debilitating diseases rather than lethal infections. Effects on the host include reduced longevity, fertility, weight, and general vigor. Vertical transmission of microsporidia is frequently reported. In the case of insect hosts, vertical transmission often occurs as transovarial transmission, where the microsporidian parasites pass from the ovaries of the female host into eggs and eventually multiply in the infected larvae. Amblyospora salinaria n. sp. which infects the mosquito Culex salinarius Coquillett, and Amblyospora californica which infects the mosquito Culex tarsalis Coquillett, provide typical examples of transovarial transmission of microsporidia.[15][16][17][18]
Microsporidia, specifically the mosquito-infecting Vavraia culicis, are being explored as a possible 'evolution-proof' malaria-control method.[19] Microsporidian infection of Anopheles gambiae (the principal vector of Plasmodium falciparum malaria) reduces malarial infection within the mosquito, and shortens the mosquito lifespan.[20] As the majority of malaria-infected mosquitoes naturally die before the malaria parasite is mature enough to transmit, any increase in mosquito mortality through microsporidian-infection may reduce malaria transmission to humans. In May 2020, researchers reported that Microsporidia MB, a symbiont in the midgut and ovaries of An. arabiensis, significantly impaired transmisison of P. falciparum, had "no overt effect" on the fitness of host mosquitoes, and was transmitted vertically (through inheritance).[21]
Clinical
This section needs expansion. You can help by adding to it. (November 2013) |
Microsporidian infections of humans sometimes cause a disease called microsporidiosis. At least 14 microsporidian species, spread across eight genera, have been recognized as human pathogens. These include Trachipleistophora hominis.[22]
As hyperparasites
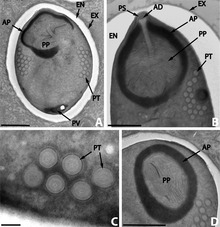
Microsporidia can infect a variety of hosts, including hosts which are themselves parasites. In that case, the microsporidian species is a hyperparasite, i.e. a parasite of a parasite. As an example, more than eighteen species are known which parasitize digeneans (parasitic flatworms). These digeneans are themselves parasites in various vertebrates and molluscs. Eight of these species belong to the genus Nosema.[23]
Genomes
Microsporidia have the smallest known (nuclear) eukaryotic genomes. The parasitic lifestyle of microsporidia has led to a loss of many mitochondrial and Golgi genes, and even their ribosomal RNAs are reduced in size compared with those of most eukaryotes. As a consequence, the genomes of microsporidia are much smaller than those of other eukaryotes. Currently known microsporidial genomes are 2.5 to 11.6 Mb in size, encoding from 1,848 to 3,266 proteins which is in the same range as many bacteria.[24]
Horizontal gene transfer (HGT) seems to have occurred many times in microsporidia. For instance, the genomes of Encephalitozoon romaleae and Trachipleistophora hominis contain genes that derive from animals and bacteria, and some even from fungi.[24]
Classification
The first described microsporidian genus, Nosema, was initially put by Nägeli in the fungal group Schizomycetes together with some bacteria and yeasts.[25][26] For some time microsporidia were considered as very primitive eukaryotes, placed in the protozoan group Cnidospora.[4] Later, especially because of the lack of mitochondria, they were placed along with the other Protozoa such as diplomonads, parabasalids and archamoebae in the protozoan-group Archezoa.[27] More recent research has falsified this theory of early origin (for all of these). Instead, microsporidia are proposed to be highly developed and specialized organisms, which just dispensed functions that are needed no longer, because they are supplied by the host.[28] Furthermore, spore-forming organisms in general do have a complex system of reproduction, both sexual and asexual, which look far from primitive.
Since the mid-2000s microsporidia are placed within the Fungi or as a sister-group of the Fungi with a common ancestor.[29][30][31][32]
Work to identify clades is largely based on habitat and host. Three classes of Microsporidia are proposed by Vossbrinck and Debrunner-Vossbrinck, based on the habitat: Aquasporidia, Marinosporidia and Terresporidia.[33]
Another proposed classification is:[34]
- Class Metchnikovellea Weiser 1977 emend. Cavalier-Smith 1993 [Manubrispora Cavalier-Smith 1998]
- Order Metchnikovellida Vivier 1977
- Family Amphiacanthidae
- Family Metchnikovellidae Caullery & Mesnil 1897
- Order Metchnikovellida Vivier 1977
- Class Microsporea Delphy 1936 ex Levine & Corliss 1963
- Family Cougourdellidae Poisson 1953
- Family Facilisporidae
- Family Heterovesiculidae Lange et al. 1995
- Family Myosporidae
- Family Nadelsporidae
- Family Neonosemoidiidae
- Family Ordosporidae
- Family Pseudonosematidae
- Family Telomyxidae Leger & Hesse 1910
- Family Toxoglugeidae
- Family Tubulinosematidae
- Subclass Haplophasea Sprague, Becnel & Hazard 1992
- Order Chytridiopsida Weiser 1974
- Family Chytridiopsidae Sprague, Ormieres & Manier 1972
- Family Buxtehudiidae Larsson 1980
- Family Enterocytozoonidae
- Family Burkeidae Sprague 1977
- Family Hesseidae Ormierees & Sprague 1973
- Order Glugeida Issi 1986
- Family Glugeidae Thelohan 1892 [Pleistophoridae Stempell 1909]
- Family Gurleyidae Sprague 1977
- Family Encephalitozoonidae
- Family Abelsporidae
- Family Tuzetiidae Sprague, Tuzet & Maurand 1977
- Family Microfilidae
- Family Unikaryonidae Sprague 1977
- Order Chytridiopsida Weiser 1974
- Subclass Dihaplophasea Sprague, Becnel & Hazard 1992
- Order Meiodihaplophasida Sprague, Becnel & Hazard 1992
- Superfamily Thelohanioidea Hazard & Oldacre 1975
- Family Thelohaniidae Hazard & Oldacre 1975 [Coccosporidae]
- Family Duboscqiidae Sprague 1977
- Family Janacekiidae
- Family Pereziidae Loubes et al. 1977
- Family Striatosporidae
- Family Cylindrosporidae
- Superfamily Burenelloidea Jouvenaz & Hazard 1978
- Family Burenellidae Jouvenaz & Hazard 1978
- Superfamily Amblyosporoidea Weiser 1977
- Family Amblyosporidae Weiser 1977
- Superfamily Thelohanioidea Hazard & Oldacre 1975
- Order Dissociodihaplophasida Sprague, Becnel & Hazard 1992
- Superfamily Nosematoidea Labbe
- Family Nosematidae Labbe 1899
- Family Ichthyosporidiidae
- Family Caudosporidae Weiser 1958
- Family Pseudopleistophoridae Sprague 1977
- Family Mrazekiidae Leger & Hesse 1922
- Superfamily Culicosporoidea Weiser 1977
- Family Culicosporidae Weisser 1977
- Family Culicosporellidae
- Family Golbergiidae
- Family Spragueidae Weissenberg 1976
- Superfamily Ovavesiculoidea Sprague, Becnel & Hazard 1992
- Family Ovavesiculidae
- Family Tetramicridae
- Superfamily Nosematoidea Labbe
- Order Meiodihaplophasida Sprague, Becnel & Hazard 1992
A third classification by Cavalier-Smith 1993:[35]
- Subphyla Rudimicrospora Cavalier-Smith 1993
- Class Minisporea Cavalier-Smith 1993
- Order Minisporida Sprague, 1972
- Class Metchnikovellea Weiser, 1977
- Order Metchnikovellida Vivier, 1975
- Class Minisporea Cavalier-Smith 1993
- Subphyla Polaroplasta Cavalier-Smith 1993
- Class Pleistophoridea Cavalier-Smith 1993
- Order Pleistophorida Stempell 1906
- Class Disporea Cavalier-Smith 1993
- Subclass Unikaryotia Cavalier-Smith 1993
- Subclass Diplokaryotia Cavalier-Smith 1993
- Class Pleistophoridea Cavalier-Smith 1993
See also
- List of Microsporidian genera
- Glugea, a genus of microsporidia
- Nosema apis, a microsporidian parasite of bees
References
- ^ Balbiani, G (1882). "Sur les microsporidies ou psorospermies des Articulés". C. R. Acad. Sci. 95: 1168–71.
- ^ Delphy, J. 1936. Sous-règne des Protozoaires. In: Perrier, R. (ed.). La Faune de la France en tableaux synoptiques illustrés, vol 1A. Delagrave: Paris.
- ^ Levine, N. D.; et al. (1980). "A Newly Revised Classification of the Protozoa". The Journal of Protozoology. 27 (1): 37–58. doi:10.1111/j.1550-7408.1980.tb04228.x. PMID 6989987.
- ^ a b Corliss JO, Levine ND (1963). "Establishment of the Microsporidea as a new class in the protozoan subphylum Cnidospora". The Journal of Protozoology. 10 (Suppl): 26–27. doi:10.1111/jeu.1963.10.issue-s3.
- ^ Sprague, V. (1977). Classification and phylogeny of the Microsporidia. In: Comparative pathobiology. vol. 2, Systematics of the Microsporidia. Lee A. Bulla & Thomas C. Cheng (ed.). pp. 1-30. New York: Plenum Press, [1].
- ^ Hibbett, D.S.; et al. (2007). "A higher level phylogenetic classification of the Fungi" (PDF). Mycological Research. 111 (5): 509–47. doi:10.1016/j.mycres.2007.03.004. PMID 17572334.
- ^ Silar, Philippe (2016). Protistes Eucaryotes : Origine, Evolution et Biologie des Microbes Eucaryotes. HAL. p. 462. ISBN 978-2-9555841-0-1.
- ^ Hawksworth, David (2001). "The magnitude of fungal diversity: The 1.5 million spices estimate revisited". Mycological Research. 105 (12): 1422. doi:10.1017/S0953756201004725.
- ^ a b Ronny Larsson, Lund University (Department of Cell and Organism Biology) Cytology and taxonomy of the microsporidia Archived 2009-09-12 at the Wayback Machine 2004.
- ^ Didier, ES. (Apr 2005). "Microsporidiosis: an emerging and opportunistic infection in humans and animals". Acta Trop. 94 (1): 61–76. doi:10.1016/j.actatropica.2005.01.010. PMID 15777637.
- ^ Balbiani, G. 1884. Les Psorospermies des Articulés ou Microsporidies, pp. 150-168, 184. In: Leçons sur les sporozoaires. Paris: Doin, [2].
- ^ Hoffman, G. (1999). Parasites of North American Freshwater Fishes, 2nd edn, University of California Press, Berkeley, California, USA, p. 89, [3].
- ^ Winters, A. D.; Faisal, M. (2014). "Molecular and ultrastructural characterization of Dictyocoela diporeiae n. sp. (Microsporidia), a parasite of Diporeia spp. (Amphipoda, Gammaridea)". Parasite. 21: 26. doi:10.1051/parasite/2014028. PMC 4059264. PMID 24934702.
- ^ Ironside JE (2007). "Multiple losses of sex within a single genus of Microsporidia". BMC Evolutionary Biology. 7: 48. doi:10.1186/1471-2148-7-48. PMC 1853083. PMID 17394631.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Andreadis TG, Hall DW (August 1979). "Development, ultrastructure, and mode of transmission of Amblyospora sp. (Microspora) in the mosquito". The Journal of Protozoology. 26 (3): 444–52. doi:10.1111/j.1550-7408.1979.tb04651.x. PMID 536933.
- ^ Andreadis TG, Hall DW (September 1979). "Significance of transovarial infections of Amblyospora sp. (Microspora:Thelohaniidae) in relation to parasite maintenance in the mosquito Culex salinarius". Journal of Invertebrate Pathology. 34 (2): 152–7. doi:10.1016/0022-2011(79)90095-8. PMID 536610.
- ^ Jahn GC, Hall DW, Zam SG (1986). "A comparison of the life cycles of two Amblyospora (Microspora: Amblyosporidae) in the mosquitoes Culex salinarius and Culex tarsalis Coquillett". Journal of the Florida Anti-Mosquito Association. 57 (1): 24–27.
- ^ Becnel JJ, Andreadis TG (May 1998). "Amblyospora salinaria n. sp. (Microsporidia: amblyosporidae), parasite of Culex salinarius (Diptera: culicidae): its life cycle stages in an intermediate host". Journal of Invertebrate Pathology. 71 (3): 258–62. doi:10.1006/jipa.1998.4729. PMID 9538031.
- ^ Koella, Jacob C.; Lorenz, Lena; Bargielowski, Irka (2009). Chapter 12 Microsporidians as Evolution-Proof Agents of Malaria Control?. Advances in Parasitology. Vol. 68. pp. 315–327. doi:10.1016/S0065-308X(08)00612-X. ISBN 978-0-12-374787-7. PMID 19289199.
- ^ Bargielowski I, Koella JC (2009). Baylis M (ed.). "A Possible Mechanism for the Suppression of Plasmodium berghei Development in the Mosquito Anopheles gambiae by the Microsporidian Vavraia culicis". PLOS ONE. 4 (3): e4676. Bibcode:2009PLoSO...4.4676B. doi:10.1371/journal.pone.0004676. PMC 2651578. PMID 19277119.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Herren, JK; Mbaisi, L; Mararo, E; et al. (2020). "A microsporidian impairs Plasmodium falciparum transmission in Anopheles arabiensis mosquitoes". Nature Communications. 11 (2187): 2187. Bibcode:2020NatCo..11.2187H. doi:10.1038/s41467-020-16121-y. PMC 7198529. PMID 32366903.
- ^ Heinz, E; Williams, TA; Nakjang, S; et al. (Oct 2012). "The genome of the obligate intracellular parasite Trachipleistophora hominis: New insights into microsporidian genome dynamics and reductive evolution". PLOS Pathog. 8 (10): e1002979. doi:10.1371/journal.ppat.1002979. PMC 3486916. PMID 23133373.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ a b Toguebaye, B. S.; Quilichini, Y.; Diagne, P. M.; Marchand, B. (2014). "Ultrastructure and development of Nosema podocotyloidis n. sp. (Microsporidia), a hyperparasite of Podocotyloides magnatestis (Trematoda), a parasite of Parapristipoma octolineatum (Teleostei)". Parasite. 21: 44. doi:10.1051/parasite/2014044. PMC 4150386. PMID 25174849.

- ^ a b Corradi, N.; Selman, M. (2013). "Latest Progress in Microsporidian Genome Research". Journal of Eukaryotic Microbiology. 60 (3): 309–312. doi:10.1111/jeu.12030. PMID 23445243. S2CID 24504235.
- ^ Nägeli, C. von (1857). "Über die neue Krankheit der Seidenraupe und verwandte Organismen. pp. 760–61. In: Caspary, R. (ed.). Bericht über die Verhandlungen der 33. Versammlung deutscher Naturforscher und Aerzte, gehalten in Bonn von 18 bis 24 September 1857". Botanische Zeitung. 15: 749–776.
- ^ Keeling, P. J.; Fast, N. M. (2002). "Microsporidia: biology and evolution of highly reduced intracellular parasites" (PDF). Annual Review of Microbiology. 56 (1): 93–116. doi:10.1146/annurev.micro.56.012302.160854. PMID 12142484.
- ^ Cavalier-Smith, T (1993). "Kingdom protozoa and its 18 phyla" (PDF). Microbiological Reviews. 57 (4): 953–994. doi:10.1128/MR.57.4.953-994.1993. PMC 372943. PMID 8302218.
- ^ Keeling PJ, Slamovits CH (December 2004). "Simplicity and Complexity of Microsporidian Genomes". Eukaryotic Cell. 3 (6): 1363–9. doi:10.1128/EC.3.6.1363-1369.2004. PMC 539024. PMID 15590811.
- ^ Fischer WM, Palmer JD (September 2005). "Evidence from small-subunit ribosomal RNA sequences for a fungal origin of Microsporidia". Molecular Phylogenetics and Evolution. 36 (3): 606–22. doi:10.1016/j.ympev.2005.03.031. PMID 15923129.
- ^ Liu YJ, Hodson MC, Hall BD (2006). "Loss of the flagellum happened only once in the fungal lineage: phylogenetic structure of Kingdom Fungi inferred from RNA polymerase II subunit genes". BMC Evolutionary Biology. 6: 74. doi:10.1186/1471-2148-6-74. PMC 1599754. PMID 17010206.
{{cite journal}}: CS1 maint: unflagged free DOI (link) - ^ Gill EE, Fast NM (June 2006). "Assessing the microsporidia-fungi relationship: Combined phylogenetic analysis of eight genes". Gene. 375: 103–9. doi:10.1016/j.gene.2006.02.023. PMID 16626896.
- ^ Lee SC, Corradi N, Byrnes EJ, et al. (November 2008). "Microsporidia evolved from ancestral sexual fungi". Current Biology. 18 (21): 1675–9. doi:10.1016/j.cub.2008.09.030. PMC 2654606. PMID 18976912.
- ^ Vossbrinck CR, Debrunner-Vossbrinck BA (May 2005). "Molecular phylogeny of the Microsporidia: ecological, ultrastructural and taxonomic considerations". Folia Parasitologica. 52 (1–2): 131–42, discussion 130. doi:10.14411/fp.2005.017. PMID 16004372.
- ^ Alimov, A. F., ed. (May 2007). "Protista 2: Handbook on zoology". Nauka: 1141.
{{cite journal}}: Cite journal requires|journal=(help) - ^ Cavalier-Smith (1993). "Kingdom Protozoa and its 18 phyla" (PDF). Microbiological Reviews. 57 (4): 953–94. doi:10.1128/MR.57.4.953-994.1993. PMC 372943. PMID 8302218.
External links
![]() Data related to Microsporidia at Wikispecies
Data related to Microsporidia at Wikispecies
- BioHealthBase Bioinformatics Resource Center Database of microspordia sequences and related information.
- Microsporidia at the U.S. National Library of Medicine Medical Subject Headings (MeSH)
