Creatine
 Skeletal formula of neutral form of creatine
| |
 Skeletal formula of one of the zwitterionic forms of creatine
| |
 Ball and stick model of one zwitterionic form of creatine
| |
| Names | |
|---|---|
| Systematic IUPAC name
2-[Carbamimidoyl(methyl)amino]acetic acid | |
| Other names
N-Carbamimidoyl-N-methylglycine; Methylguanidoacetic acid; N-amidinosarcosine
| |
| Identifiers | |
3D model (JSmol)
|
|
| 907175 | |
| ChEBI | |
| ChEMBL | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.000.278 |
| EC Number |
|
| 240513 | |
| KEGG | |
| MeSH | Creatine |
PubChem CID
|
|
| RTECS number |
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C4H9N3O2 | |
| Molar mass | 131.135 g·mol−1 |
| Appearance | White crystals |
| Odor | Odourless |
| Melting point | 255 °C (491 °F; 528 K) |
| 13.3 g L−1 (at 18 °C) | |
| log P | −1.258 |
| Acidity (pKa) | 3.429 |
| Basicity (pKb) | 10.568 |
| Isoelectric point | 8.47 |
| Thermochemistry | |
Heat capacity (C)
|
171.1 J K−1 mol−1 (at 23.2 °C) |
Std molar
entropy (S⦵298) |
189.5 J K−1 mol−1 |
Std enthalpy of
formation (ΔfH⦵298) |
−538.06–−536.30 kJ mol−1 |
Std enthalpy of
combustion (ΔcH⦵298) |
−2.3239–−2.3223 MJ mol−1 |
| Pharmacology | |
| C01EB06 (WHO) | |
| Pharmacokinetics: | |
| 3 hours | |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H315, H319, H335 | |
| P261, P305+P351+P338 | |
| Related compounds | |
Related alkanoic acids
|
|
Related compounds
|
Dimethylacetamide |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Creatine (/ˈkriːətiːn/ or /ˈkriːətɪn/)[1] is an organic compound with the nominal formula (H2N)(HN)CN(CH3)CH2CO2H. It exists in various tautomers in solutions (among which are neutral form and various zwitterionic forms). Creatine is found in vertebrates, where it facilitates recycling of adenosine triphosphate (ATP), primarily in muscle and brain tissue. Recycling is achieved by converting adenosine diphosphate (ADP) back to ATP via donation of phosphate groups. Creatine also acts as a buffer.[2]
History
[edit]Creatine was first identified in 1832 when Michel Eugène Chevreul isolated it from the basified water-extract of skeletal muscle. He later named the crystallized precipitate after the Greek word for meat, κρέας (kreas). In 1928, creatine was shown to exist in equilibrium with creatinine.[3] Studies in the 1920s showed that consumption of large amounts of creatine did not result in its excretion. This result pointed to the ability of the body to store creatine, which in turn suggested its use as a dietary supplement.[4]
In 1912, Harvard University researchers Otto Folin and Willey Glover Denis found evidence that ingesting creatine can dramatically boost the creatine content of the muscle.[5][6] In the late 1920s, after finding that the intramuscular stores of creatine can be increased by ingesting creatine in larger than normal amounts, scientists discovered phosphocreatine (creatine phosphate), and determined that creatine is a key player in the metabolism of skeletal muscle. It is naturally formed in vertebrates.[7]
The discovery of phosphocreatine[8][9] was reported in 1927.[10][11][9] In the 1960s, creatine kinase (CK) was shown to phosphorylate ADP using phosphocreatine (PCr) to generate ATP. It follows that ATP - not PCr - is directly consumed in muscle contraction. CK uses creatine to "buffer" the ATP/ADP ratio.[12]
While creatine's influence on physical performance has been well documented since the early twentieth century, it came into public view following the 1992 Olympics in Barcelona. An August 7, 1992 article in The Times reported that Linford Christie, the gold medal winner at 100 meters, had used creatine before the Olympics (however, it should also be noted that Christie was found guilty of doping later in his career).[13] An article in Bodybuilding Monthly named Sally Gunnell, who was the gold medalist in the 400-meter hurdles, as another creatine user. In addition, The Times also noted that 100 meter hurdler Colin Jackson began taking creatine before the Olympics.[14][15]

At the time, low-potency creatine supplements were available in Britain, but creatine supplements designed for strength enhancement were not commercially available until 1993 when a company called Experimental and Applied Sciences (EAS) introduced the compound to the sports nutrition market under the name Phosphagen.[16] Research performed thereafter demonstrated that the consumption of high glycemic carbohydrates in conjunction with creatine increases creatine muscle stores.[17]
Metabolic role
[edit]Creatine is a naturally occurring non-protein compound and the primary constituent of phosphocreatine, which is used to regenerate ATP within the cell. 95% of the human body's total creatine and phosphocreatine stores are found in skeletal muscle, while the remainder is distributed in the blood, brain, testes, and other tissues.[18][19] The typical creatine content of skeletal muscle (as both creatine and phosphocreatine) is 120 mmol per kilogram of dry muscle mass, but can reach up to 160 mmol/kg through supplementation.[20] Approximately 1–2% of intramuscular creatine is degraded per day and an individual would need about 1–3 grams of creatine per day to maintain average (unsupplemented) creatine storage.[20][21][22] An omnivorous diet provides roughly half of this value, with the remainder synthesized in the liver and kidneys.[18][19][23]
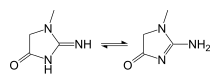
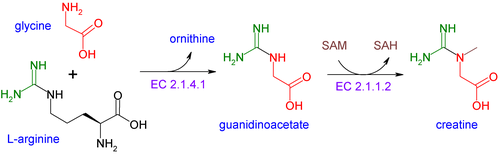
Creatine is not an essential nutrient.[24] It is an amino acid derivative, naturally produced in the human body from the amino acids glycine and arginine, with an additional requirement for S-Adenosyl methionine (a derivative of methionine) to catalyze the transformation of guanidinoacetate to creatine. In the first step of the biosynthesis, the enzyme arginine:glycine amidinotransferase (AGAT, EC:2.1.4.1) mediates the reaction of glycine and arginine to form guanidinoacetate. This product is then methylated by guanidinoacetate N-methyltransferase (GAMT, EC:2.1.1.2), using S-adenosyl methionine as the methyl donor. Creatine itself can be phosphorylated by creatine kinase to form phosphocreatine, which is used as an energy buffer in skeletal muscles and the brain. A cyclic form of creatine, called creatinine, exists in equilibrium with its tautomer and with creatine.
Phosphocreatine system
[edit]
Creatine is transported through the blood and taken up by tissues with high energy demands, such as the brain and skeletal muscle, through an active transport system. The concentration of ATP in skeletal muscle is usually 2–5 mM, which would result in a muscle contraction of only a few seconds.[25] During times of increased energy demands, the phosphagen (or ATP/PCr) system rapidly resynthesizes ATP from ADP with the use of phosphocreatine (PCr) through a reversible reaction catalysed by the enzyme creatine kinase (CK). The phosphate group is attached to an NH center of the creatine. In skeletal muscle, PCr concentrations may reach 20–35 mM or more. Additionally, in most muscles, the ATP regeneration capacity of CK is very high and is therefore not a limiting factor. Although the cellular concentrations of ATP are small, changes are difficult to detect because ATP is continuously and efficiently replenished from the large pools of PCr and CK.[25] A proposed representation has been illustrated by Krieder et al.[26] Creatine has the ability to increase muscle stores of PCr, potentially increasing the muscle's ability to resynthesize ATP from ADP to meet increased energy demands.[27][28][29]
Creatine supplementation appears to increase the number of myonuclei that satellite cells will 'donate' to damaged muscle fibers, which increases the potential for growth of those fibers. This increase in myonuclei probably stems from creatine's ability to increase levels of the myogenic transcription factor MRF4.[30]
Genetic deficiencies
[edit]Genetic deficiencies in the creatine biosynthetic pathway lead to various severe neurological defects.[31] Clinically, there are three distinct disorders of creatine metabolism, termed cerebral creatine deficiencies. Deficiencies in the two synthesis enzymes can cause L-arginine:glycine amidinotransferase deficiency caused by variants in GATM and guanidinoacetate methyltransferase deficiency, caused by variants in GAMT. Both biosynthetic defects are inherited in an autosomal recessive manner. A third defect, creatine transporter defect, is caused by mutations in SLC6A8 and is inherited in a X-linked manner. This condition is related to the transport of creatine into the brain.[32]
Vegans and vegetarians
[edit]Vegan and vegetarian diets are associated with lower levels of muscle creatine, and athletes on these diets may benefit from creatine supplementation.[33]
Pharmacokinetics
[edit]Most of the research to-date on creatine has predominantly focused on the pharmacological properties of creatine, yet there is a lack of research into the pharmacokinetics of creatine. Studies have not established pharmacokinetic parameters for clinical usage of creatine such as volume of distribution, clearance, bioavailability, mean residence time, absorption rate, and half life. A clear pharmacokinetic profile would need to be established prior to optimal clinical dosing.[34]
Dosing
[edit]Loading phase
[edit]
An approximation of 0.3 g/kg/day divided into 4 equal spaced intervals has been suggested since creatine needs may vary based on body weight.[26][20] It has also been shown that taking a lower dose of 3 grams a day for 28 days can also increase total muscle creatine storage to the same amount as the rapid loading dose of 20 g/day for 6 days.[20] However, a 28-day loading phase does not allow for ergogenic benefits of creatine supplementation to be realized until fully saturated muscle storage.
This elevation in muscle creatine storage has been correlated with ergogenic benefits discussed in the research section. However, higher doses for longer periods of time are being studied to offset creatine synthesis deficiencies and mitigating diseases.[35][36][32]
Maintenance phase
[edit]After the 5–7 day loading phase, muscle creatine stores are fully saturated and supplementation only needs to cover the amount of creatine broken down per day. This maintenance dose was originally reported to be around 2–3 g/day (or 0.03 g/kg/day),[20] however, some studies have suggested 3–5 g/day maintenance dose to maintain saturated muscle creatine.[17][22][37][38]
Absorption
[edit]
Endogenous serum or plasma creatine concentrations in healthy adults are normally in a range of 2–12 mg/L. A single 5 gram (5000 mg) oral dose in healthy adults results in a peak plasma creatine level of approximately 120 mg/L at 1–2 hours post-ingestion. Creatine has a fairly short elimination half life, averaging just less than 3 hours, so to maintain an elevated plasma level it would be necessary to take small oral doses every 3–6 hours throughout the day.
Exercise and sport
[edit]Creatine supplements are marketed in ethyl ester, gluconate, monohydrate, and nitrate forms.[40]
Creatine supplementation for sporting performance enhancement is considered safe for short-term use but there is a lack of safety data for long term use, or for use in children and adolescents.[41] Some athletes choose to cycle on and off creatine.[42]
A 2018 review article in the Journal of the International Society of Sports Nutrition said that creatine monohydrate might help with energy availability for high-intensity exercise.[43]
Creatine use can increase maximum power and performance in high-intensity anaerobic repetitive work (periods of work and rest) by 5% to 15%.[44][45][46] Creatine has no significant effect on aerobic endurance, though it will increase power during short sessions of high-intensity aerobic exercise.[47][obsolete source][48][obsolete source]
Creatine is proven to boost the recovery and work capacity of an athlete, and multi-applicable capabilities upon athletes have given it a lot of interest over the course of the past decade. A survey of 21,000 college athletes showed that 14% of athletes take creatine supplements to try to improve performance.[49] Compared to normal athletes, those with creatine supplementation have been shown to produce better athletic performance.[50] Non-athletes report taking creatine supplements to improve appearance.[49]
Research
[edit]Cognitive performance
[edit]Creatine is sometimes reported to have a beneficial effect on brain function and cognitive processing, although the evidence is difficult to interpret systematically and the appropriate dosing is unknown.[51][52] The greatest effect appears to be in individuals who are stressed (due, for instance, to sleep deprivation) or cognitively impaired.[51][52][53]
A 2018 systematic review found that "generally, there was evidence that short term memory and intelligence/reasoning may be improved by creatine administration", whereas for other cognitive domains "the results were conflicting".[54] Another 2023 review initially found evidence of improved memory function.[55] However, it was later determined that faulty statistics lead to the statistical significance and after fixing the "double counting", the effect was only significant in older adults.[56]
A 2023 review study "...supported claims that creatine supplementation can increases [sic] brain creatine content but also demonstrated somewhat equivocal results for effects on cognition. It does, however, provide evidence to suggest that more research is required with stressed populations, as supplementation does appear to significantly affect brain content.[57]
Muscular disease
[edit]A meta-analysis found that creatine treatment increased muscle strength in muscular dystrophies, and potentially improved functional performance.[58] Creatine treatment does not appear to improve muscle strength in people who have metabolic myopathies.[58] High doses of creatine lead to increased muscle pain and an impairment in activities of daily living when taken by people who have McArdle disease.[58]
According to a clinical study focusing on people with various muscular dystrophies, using a pure form of creatine monohydrate can be beneficial in rehabilitation after injuries and immobilization.[59]
Mitochondrial diseases
[edit]Parkinson's disease
[edit]Creatine's impact on mitochondrial function has led to research on its efficacy and safety for slowing Parkinson's disease. As of 2014, the evidence did not provide a reliable foundation for treatment decisions, due to risk of bias, small sample sizes, and the short duration of trials.[60]
Huntington's disease
[edit]Several primary studies[61][62][63] have been completed but no systematic review on Huntington's disease has been completed yet.
ALS
[edit]It is ineffective as a treatment for amyotrophic lateral sclerosis.[64]
Testosterone
[edit]A 2021 systemic review of studies found that "the current body of evidence does not indicate that creatine supplementation increases total testosterone, free testosterone, DHT or causes hair loss/baldness".[65]
Adverse effects
[edit]- Weight gain due to extra water retention to the muscle
- Potential muscle cramps / strains / pulls
- Upset stomach
- Diarrhea
- Dizziness
One well-documented effect of creatine supplementation is weight gain within the first week of the supplement schedule, likely attributable to greater water retention due to the increased muscle creatine concentrations by means of osmosis.[68]
A 2009 systematic review discredited concerns that creatine supplementation could affect hydration status and heat tolerance and lead to muscle cramping and diarrhea.[69][70]
Despite weight gain due to water retention and potential cramps being two seemingly "common" side effects, new research indicates that these side effects are likely not the result of creatine usage. In addition, the initial water retention is attributed to more short-term creatine use (the "loading" phase). Studies have shown that creatine usage does not necessarily affect total body water relative to muscle mass in the long-term.[71]
Renal function
[edit]Long-term creatine supplementation has not been proven safe either in general or for people with kidney conditions.[72]
A 2019 systematic review published by the National Kidney Foundation investigated whether creatine supplementation had adverse effects on renal function.[73] They identified 15 studies from 1997 to 2013 that looked at standard creatine loading and maintenance protocols of 4–20 g/day of creatine versus placebo. They utilized serum creatinine, creatinine clearance, and serum urea levels as a measure of renal damage. While in general creatine supplementation resulted in slightly elevated creatinine levels that remained within normal limits, supplementation did not induce renal damage (P value< 0.001). Special populations included in the 2019 Systematic review included type 2 diabetic patients[74] and post-menopausal women,[75] bodybuilders,[76] athletes,[77] and resistance trained populations.[78][79][80] The study also discussed 3 case studies where there were reports that creatine affected renal function.[81][82][83]
In a joint statement between the American College of Sports Medicine, Academy of Nutrition and Dietetics, and Dietitians in Canada on performance enhancing nutrition strategies, creatine was included in their list of ergogenic aids and they do not list renal function as a concern for use.[84]
The most recent position stand on creatine from the Journal of International Society of Sports Nutrition states that creatine is safe to take in healthy populations from infants to the elderly to performance athletes. They also state that long term (5 years) use of creatine has been considered safe.[26]
It is important to mention that kidneys themselves, for normal physiological function, need phosphocreatine and creatine and indeed kidneys express significant amounts of creatine kinases (BB-CK and u-mtCK isoenzymes).[85] At the same time, the first of two steps for endogenous creatine synthesis takes place in the kidneys themselves. Patients with kidney disease and those undergoing dialysis treatment generally show significantly lower levels of creatine in their organs, since the pathological kidneys are both hampered in creatine synthesis capability and are in back-resorption of creatine from the urine in the distal tubules. In addition, dialysis patients lose creatine due to wash out by the dialysis treatment itself and thus become chronically creatine depleted. This situation is exacerbated by the fact that dialysis patients generally consume less meat and fish, the alimentary sources of creatine. Therefore, to alleviate chronic creatine depletion in these patients and allow organs to replenish their stores of creatine, it was proposed in a 2017 article in Medical Hypotheses to supplement dialysis patients with extra creatine, preferably by intra-dialytic administration. Such a supplementation with creatine in dialysis patients is expected to significantly improve the health and quality of the patients by improving muscle strength, coordination of movement, brain function and to alleviate depression and chronic fatigue that are common in these patients.[86][unreliable medical source?]
Safety
[edit]Contamination
[edit]A 2011 survey of 33 supplements commercially available in Italy found that over 50% of them exceeded the European Food Safety Authority recommendations in at least one contaminant. The most prevalent of these contaminants was creatinine, a breakdown product of creatine also produced by the body.[87] Creatinine was present in higher concentrations than the European Food Safety Authority recommendations in 44% of the samples. About 15% of the samples had detectable levels of dihydro-1,3,5-triazine or a high dicyandiamide concentration. Heavy metals contamination was not found to be a concern, with only minor levels of mercury being detectable. Two studies reviewed in 2007 found no impurities.[88]
Food and cooking
[edit]When creatine is mixed with protein and sugar at high temperatures (above 148 °C), the resulting reaction produces carcinogenic heterocyclic amines (HCAs).[89] Such a reaction happens when grilling or pan-frying meat.[90] Creatine content (as a percentage of crude protein) can be used as an indicator of meat quality.[91]
Dietary considerations
[edit]Creatine-monohydrate is suitable for vegetarians and vegans, as the raw materials used for the production of the supplement have no animal origin.[92]
See also
[edit]References
[edit]- ^ Stout JR, Antonio J, Kalman E, eds. (2008). Essentials of Creatine in Sports and Health. Humana. ISBN 978-1-59745-573-2.
- ^ Barcelos RP, Stefanello ST, Mauriz JL, Gonzalez-Gallego J, Soares FA (2016). "Creatine and the Liver: Metabolism and Possible Interactions". Mini Reviews in Medicinal Chemistry. 16 (1): 12–8. doi:10.2174/1389557515666150722102613. PMID 26202197.
The process of creatine synthesis occurs in two steps, catalyzed by L-arginine:glycine amidinotransferase (AGAT) and guanidinoacetate N-methyltransferase (GAMT), which take place mainly in kidney and liver, respectively. This molecule plays an important energy/pH buffer function in tissues, and to guarantee the maintenance of its total body pool, the lost creatine must be replaced from diet or de novo synthesis.
- ^ Cannan RK, Shore A (1928). "The creatine-creatinine equilibrium. The apparent dissociation constants of creatine and creatinine". The Biochemical Journal. 22 (4): 920–9. doi:10.1042/bj0220920. PMC 1252207. PMID 16744118.
- ^ Volek JS, Ballard KD, Forsythe CE (2008). "Overview of Creatine Metabolism". In Stout JR, Antonio J, Kalman E (eds.). Essentials of Creatine in Sports and Health. Humana. pp. 1–23. ISBN 978-1-59745-573-2.
- ^ Folin O, Denis W (1912). "Protein metabolism from the standpoint of blood and tissue analysis". Journal of Biological Chemistry. 12 (1): 141–61. doi:10.1016/S0021-9258(18)88723-3. Archived from the original on 3 May 2018. Retrieved 8 May 2018.
- ^ Antonio, Jose (8 February 2021). "Common questions and misconceptions about creatine supplementation: what does the scientific evidence really show?". Journal of the International Society of Sports Nutrition. 18 (1): 13. doi:10.1186/s12970-021-00412-w. PMC 7871530. PMID 33557850.
- ^ Brosnan JT, da Silva RP, Brosnan ME (May 2011). "The metabolic burden of creatine synthesis". Amino Acids. 40 (5): 1325–31. doi:10.1007/s00726-011-0853-y. PMID 21387089. S2CID 8293857.
- ^ Saks V (2007). Molecular system bioenergetics: energy for life. Weinheim: Wiley-VCH. p. 2. ISBN 978-3-527-31787-5.
- ^ a b Ochoa S (1989). Sherman EJ, National Academy of Sciences (eds.). David Nachmansohn. Biographical Memoirs. Vol. 58. National Academies Press. pp. 357–404. ISBN 978-0-309-03938-3.
- ^ Eggleton P, Eggleton GP (1927). "The Inorganic Phosphate and a Labile Form of Organic Phosphate in the Gastrocnemius of the Frog". The Biochemical Journal. 21 (1): 190–5. doi:10.1042/bj0210190. PMC 1251888. PMID 16743804.
- ^ Fiske CH, Subbarow Y (April 1927). "The nature of the 'inorganic phosphate' in voluntary muscle". Science. 65 (1686): 401–3. Bibcode:1927Sci....65..401F. doi:10.1126/science.65.1686.401. PMID 17807679.
- ^ Wallimann T (2007). "Introduction – Creatine: Cheap Ergogenic Supplement with Great Potential for Health and Disease". In Salomons GS, Wyss M (eds.). Creatine and Creatine Kinase in Health and Disease. Springer. pp. 1–16. ISBN 978-1-4020-6486-9.
- ^ "Shadow over Christie's reputation". 22 August 2000.
- ^ "Supplement muscles in on the market". National Review of Medicine. 30 July 2004. Archived from the original on 16 November 2006. Retrieved 25 May 2011.
- ^ Passwater RA (2005). Creatine. McGraw Hill Professional. p. 9. ISBN 978-0-87983-868-3. Archived from the original on 19 June 2022. Retrieved 8 May 2018.
- ^ Stoppani J (May 2004). Creatine new and improved: recent high-tech advances have made creatine even more powerful. Here's how you can take full advantage of this super supplement. Muscle & Fitness. Archived from the original on 11 July 2012. Retrieved 29 March 2010.
- ^ a b Green AL, Hultman E, Macdonald IA, Sewell DA, Greenhaff PL (November 1996). "Carbohydrate ingestion augments skeletal muscle creatine accumulation during creatine supplementation in humans". The American Journal of Physiology. 271 (5 Pt 1): E821-6. doi:10.1152/ajpendo.1996.271.5.E821. PMID 8944667.
- ^ a b Cooper R, Naclerio F, Allgrove J, Jimenez A (July 2012). "Creatine supplementation with specific view to exercise/sports performance: an update". Journal of the International Society of Sports Nutrition. 9 (1): 33. doi:10.1186/1550-2783-9-33. PMC 3407788. PMID 22817979.
Creatine is produced endogenously at an amount of about 1 g/d. Synthesis predominately occurs in the liver, kidneys, and to a lesser extent in the pancreas. The remainder of the creatine available to the body is obtained through the diet at about 1 g/d for an omnivorous diet. 95% of the bodies creatine stores are found in the skeletal muscle and the remaining 5% is distributed in the brain, liver, kidney, and testes [1].
- ^ a b Brosnan ME, Brosnan JT (August 2016). "The role of dietary creatine". Amino Acids. 48 (8): 1785–91. doi:10.1007/s00726-016-2188-1. PMID 26874700. S2CID 3700484.
The daily requirement of a 70-kg male for creatine is about 2 g; up to half of this may be obtained from a typical omnivorous diet, with the remainder being synthesized in the body ... More than 90% of the body's creatine and phosphocreatine is present in muscle (Brosnan and Brosnan 2007), with some of the remainder being found in the brain (Braissant et al. 2011). ... Creatine synthesized in liver must be secreted into the bloodstream by an unknown mechanism (Da Silva et al. 2014a)
- ^ a b c d e Hultman E, Söderlund K, Timmons JA, Cederblad G, Greenhaff PL (July 1996). "Muscle creatine loading in men". Journal of Applied Physiology. 81 (1): 232–7. doi:10.1152/jappl.1996.81.1.232. PMID 8828669.
- ^ Balsom PD, Söderlund K, Ekblom B (October 1994). "Creatine in humans with special reference to creatine supplementation". Sports Medicine. 18 (4): 268–80. doi:10.2165/00007256-199418040-00005. PMID 7817065. S2CID 23929060.
- ^ a b Harris RC, Söderlund K, Hultman E (September 1992). "Elevation of creatine in resting and exercised muscle of normal subjects by creatine supplementation". Clinical Science. 83 (3): 367–74. doi:10.1042/cs0830367. PMID 1327657.
- ^ Brosnan JT, da Silva RP, Brosnan ME (May 2011). "The metabolic burden of creatine synthesis". Amino Acids. 40 (5): 1325–31. doi:10.1007/s00726-011-0853-y. PMID 21387089. S2CID 8293857.
Creatinine loss averages approximately 2 g (14.6 mmol) for 70 kg males in the 20- to 39-year age group. ... Table 1 Comparison of rates of creatine synthesis in young adults with dietary intakes of the three precursor amino acids and with the whole body transmethylation flux
Creatine synthesis (mmol/day) 8.3 - ^ "Creatine". Beth Israel Deaconess Medical Center. Archived from the original on 28 January 2011. Retrieved 23 August 2010.
- ^ a b Wallimann T, Wyss M, Brdiczka D, Nicolay K, Eppenberger HM (January 1992). "Intracellular compartmentation, structure and function of creatine kinase isoenzymes in tissues with high and fluctuating energy demands: the 'phosphocreatine circuit' for cellular energy homeostasis". The Biochemical Journal. 281 ( Pt 1) (Pt 1): 21–40. doi:10.1042/bj2810021. PMC 1130636. PMID 1731757.
- ^ a b c Kreider RB, Kalman DS, Antonio J, Ziegenfuss TN, Wildman R, Collins R, et al. (2017). "International Society of Sports Nutrition position stand: safety and efficacy of creatine supplementation in exercise, sport, and medicine". Journal of the International Society of Sports Nutrition. 14: 18. doi:10.1186/s12970-017-0173-z. PMC 5469049. PMID 28615996.
- ^ Spillane M, Schoch R, Cooke M, Harvey T, Greenwood M, Kreider R, Willoughby DS (February 2009). "The effects of creatine ethyl ester supplementation combined with heavy resistance training on body composition, muscle performance, and serum and muscle creatine levels". Journal of the International Society of Sports Nutrition. 6 (1): 6. doi:10.1186/1550-2783-6-6. PMC 2649889. PMID 19228401.
- ^ Wallimann T, Tokarska-Schlattner M, Schlattner U (May 2011). "The creatine kinase system and pleiotropic effects of creatine". Amino Acids. 40 (5): 1271–96. doi:10.1007/s00726-011-0877-3. PMC 3080659. PMID 21448658..
- ^ T. Wallimann, M. Tokarska-Schlattner, D. Neumann u. a.: The Phosphocreatine Circuit: Molecular and Cellular Physiology of Creatine Kinases, Sensitivity to Free Radicals, and Enhancement by Creatine Supplementation. In: Molecular System Bioenergetics: Energy for Life. 22. November 2007. doi:10.1002/9783527621095.ch7C
- ^ Hespel P, Eijnde BO, Derave W, Richter EA (2001). "Creatine supplementation: exploring the role of the creatine kinase/phosphocreatine system in human muscle". Canadian Journal of Applied Physiology. 26 Suppl: S79-102. doi:10.1139/h2001-045. PMID 11897886.
- ^ "L-Arginine:Glycine Amidinotransferase". Archived from the original on 24 August 2013. Retrieved 16 August 2010.
- ^ a b Braissant O, Henry H, Béard E, Uldry J (May 2011). "Creatine deficiency syndromes and the importance of creatine synthesis in the brain" (PDF). Amino Acids. 40 (5): 1315–24. doi:10.1007/s00726-011-0852-z. PMID 21390529. S2CID 13755292. Archived (PDF) from the original on 10 March 2021. Retrieved 8 July 2019.
- ^ Rogerson D (2017). "Vegan diets: practical advice for athletes and exercisers". J Int Soc Sports Nutr. 14: 36. doi:10.1186/s12970-017-0192-9. PMC 5598028. PMID 28924423.
- ^ Persky AM, Brazeau GA (June 2001). "Clinical pharmacology of the dietary supplement creatine monohydrate". Pharmacological Reviews. 53 (2): 161–76. PMID 11356982.
- ^ Hanna-El-Daher L, Braissant O (August 2016). "Creatine synthesis and exchanges between brain cells: What can be learned from human creatine deficiencies and various experimental models?". Amino Acids. 48 (8): 1877–95. doi:10.1007/s00726-016-2189-0. PMID 26861125. S2CID 3675631.
- ^ Bender A, Klopstock T (August 2016). "Creatine for neuroprotection in neurodegenerative disease: end of story?". Amino Acids. 48 (8): 1929–40. doi:10.1007/s00726-015-2165-0. PMID 26748651. S2CID 2349130.
- ^ Kreider RB (February 2003). "Effects of creatine supplementation on performance and training adaptations". Molecular and Cellular Biochemistry. 244 (1–2): 89–94. doi:10.1023/A:1022465203458. PMID 12701815. S2CID 35050122.
- ^ Greenhaff PL, Casey A, Short AH, Harris R, Soderlund K, Hultman E (May 1993). "Influence of oral creatine supplementation of muscle torque during repeated bouts of maximal voluntary exercise in man". Clinical Science. 84 (5): 565–71. doi:10.1042/cs0840565. PMID 8504634.
- ^ Jäger R, Harris RC, Purpura M, Francaux M (November 2007). "Comparison of new forms of creatine in raising plasma creatine levels". Journal of the International Society of Sports Nutrition. 4: 17. doi:10.1186/1550-2783-4-17. PMC 2206055. PMID 17997838.
- ^ Cooper R, Naclerio F, Allgrove J, Jimenez A (July 2012). "Creatine supplementation with specific view to exercise/sports performance: an update". Journal of the International Society of Sports Nutrition. 9 (1): 33. doi:10.1186/1550-2783-9-33. PMC 3407788. PMID 22817979.
- ^ Butts J, Jacobs B, Silvis M (2018). "Creatine Use in Sports". Sports Health. 10 (1): 31–34. doi:10.1177/1941738117737248. PMC 5753968. PMID 29059531.
- ^ "What Happens When You Stop Taking Creatine but Keep Working Out?". Gimme Gummiez. 4 September 2024. Retrieved 24 November 2024.
- ^ Kerksick CM, Wilborn CD, Roberts MD, Smith-Ryan A, Kleiner SM, Jäger R, et al. (August 2018). "ISSN exercise & sports nutrition review update: research & recommendations". Journal of the International Society of Sports Nutrition. 15 (1): 38. doi:10.1186/s12970-018-0242-y. PMC 6090881. PMID 30068354.
- ^ Bemben MG, Lamont HS (2005). "Creatine supplementation and exercise performance: recent findings". Sports Medicine. 35 (2): 107–25. doi:10.2165/00007256-200535020-00002. PMID 15707376. S2CID 57734918.
- ^ Bird SP (December 2003). "Creatine supplementation and exercise performance: a brief review". Journal of Sports Science & Medicine. 2 (4): 123–32. PMC 3963244. PMID 24688272.
- ^ Lanhers C, Pereira B, Naughton G, Trousselard M, Lesage FX, Dutheil F (September 2015). "Creatine Supplementation and Lower Limb Strength Performance: A Systematic Review and Meta-Analyses". Sports Medicine. 45 (9): 1285–1294. doi:10.1007/s40279-015-0337-4. PMID 25946994. S2CID 7372700.
- ^ Engelhardt M, Neumann G, Berbalk A, Reuter I (July 1998). "Creatine supplementation in endurance sports". Medicine and Science in Sports and Exercise. 30 (7): 1123–9. doi:10.1097/00005768-199807000-00016. PMID 9662683.
- ^ Graham AS, Hatton RC (1999). "Creatine: a review of efficacy and safety". Journal of the American Pharmaceutical Association. 39 (6): 803–10, quiz 875–7. doi:10.1016/s1086-5802(15)30371-5. PMID 10609446.
- ^ a b "Office of Dietary Supplements - Dietary Supplements for Exercise and Athletic Performance". Archived from the original on 8 May 2018. Retrieved 5 May 2018.
- ^ Wax, B.; Kerksick, C. M.; Jagim, A. R.; Mayo, J. J.; Lyons, B. C.; Kreider, R. B. (2021). "Creatine for Exercise and Sports Performance, with Recovery Considerations for Healthy Populations". Nutrients. 13 (6): 1915. doi:10.3390/nu13061915. PMC 8228369. PMID 34199588.
- ^ a b Dolan, Eimear; Gualano, Bruno; Rawson, Eric S. (2 January 2019). "Beyond muscle: the effects of creatine supplementation on brain creatine, cognitive processing, and traumatic brain injury". European Journal of Sport Science. 19 (1): 1–14. doi:10.1080/17461391.2018.1500644. ISSN 1746-1391. PMID 30086660. S2CID 51936612. Archived from the original on 29 October 2021. Retrieved 11 October 2021.
- ^ a b Rawson, Eric S.; Venezia, Andrew C. (May 2011). "Use of creatine in the elderly and evidence for effects on cognitive function in young and old". Amino Acids. 40 (5): 1349–1362. doi:10.1007/s00726-011-0855-9. ISSN 0939-4451. PMID 21394604. S2CID 11382225. Archived from the original on 19 June 2022. Retrieved 11 October 2021.
- ^ Gordji-Nejad (2024). "Single dose creatine improves cognitive performance and induces changes in cerebral high energy phosphates during sleep deprivation". Scientific Reports. 14 (1): 4937. Bibcode:2024NatSR..14.4937G. doi:10.1038/s41598-024-54249-9. PMC 10902318. PMID 38418482.
- ^ Avgerinos, K. I.; Spyrou, N.; Bougioukas, K. I.; Kapogiannis, D. (2018). "Effects of creatine supplementation on cognitive function of healthy individuals: A systematic review of randomized controlled trials". Experimental Gerontology. 108: 166–173. doi:10.1016/j.exger.2018.04.013. PMC 6093191. PMID 29704637.
- ^ Prokopidis, Konstantinos; Giannos, Panagiotis; Triantafyllidis, Konstantinos K.; Kechagias, Konstantinos S.; Forbes, Scott C.; Candow, Darren G. (2023). "Effects of creatine supplementation on memory in healthy individuals: a systematic review and meta-analysis of randomized controlled trials". Nutrition Reviews. 81 (4): 416–27. doi:10.1093/nutrit/nuac064. PMC 9999677. PMID 35984306.
- ^ Prokopidis, Konstantinos; Giannos, Panagiotis; Triantafyllidis, Konstantinos K; Kechagias, Konstantinos S; Forbes, Scott C; Candow, Darren G (16 January 2023). "Author's reply: Letter to the Editor: Double counting due to inadequate statistics leads to false-positive findings in "Effects of creatine supplementation on memory in healthy individuals: a systematic review and meta-analysis of randomized controlled trials"". Nutrition Reviews. 81 (11): 1497–1500. doi:10.1093/nutrit/nuac111. PMID 36644912. Retrieved 31 August 2023.
- ^ McMorris, Terry; Hale, Beverley J.; Pine, Beatrice S.; Williams, Thomas B. (4 April 2024). "Creatine supplementation research fails to support the theoretical basis for an effect on cognition: Evidence from a systematic review". Behavioural Brain Research. 466: 114982. doi:10.1016/j.bbr.2024.114982. ISSN 1872-7549. PMID 38582412.
- ^ a b c Kley RA, Tarnopolsky MA, Vorgerd M (June 2013). "Creatine for treating muscle disorders". The Cochrane Database of Systematic Reviews. 2013 (6): CD004760. doi:10.1002/14651858.CD004760.pub4. PMC 6492334. PMID 23740606.
- ^ Walter MC, Lochmüller H, Reilich P, Klopstock T, Huber R, Hartard M, et al. (May 2000). "Creatine monohydrate in muscular dystrophies: A double-blind, placebo-controlled clinical study". Neurology. 54 (9): 1848–50. doi:10.1212/wnl.54.9.1848. PMID 10802796. S2CID 13304657.
- ^ Xiao Y, Luo M, Luo H, Wang J (June 2014). "Creatine for Parkinson's disease". The Cochrane Database of Systematic Reviews. 2014 (6): CD009646. doi:10.1002/14651858.cd009646.pub2. PMC 10196714. PMID 24934384.
- ^ Verbessem P, Lemiere J, Eijnde BO, Swinnen S, Vanhees L, Van Leemputte M, et al. (October 2003). "Creatine supplementation in Huntington's disease: a placebo-controlled pilot trial". Neurology. 61 (7): 925–30. doi:10.1212/01.wnl.0000090629.40891.4b. PMID 14557561. S2CID 43845514.
- ^ Bender A, Auer DP, Merl T, Reilmann R, Saemann P, Yassouridis A, et al. (January 2005). "Creatine supplementation lowers brain glutamate levels in Huntington's disease". Journal of Neurology. 252 (1): 36–41. doi:10.1007/s00415-005-0595-4. PMID 15672208. S2CID 17861207.
- ^ Hersch SM, Schifitto G, Oakes D, Bredlau AL, Meyers CM, Nahin R, Rosas HD (August 2017). "The CREST-E study of creatine for Huntington disease: A randomized controlled trial". Neurology. 89 (6): 594–601. doi:10.1212/WNL.0000000000004209. PMC 5562960. PMID 28701493.
- ^ Pastula DM, Moore DH, Bedlack RS (December 2012). "Creatine for amyotrophic lateral sclerosis/motor neuron disease". The Cochrane Database of Systematic Reviews. 2012 (12): CD005225. doi:10.1002/14651858.CD005225.pub3. PMC 11403570. PMID 23235621.
- ^ Antonio, J.; Candow, D. G.; Forbes, S. C.; Gualano, B.; Jagim, A. R.; Kreider, R. B.; Rawson, E. S.; Smith-Ryan, A. E.; Vandusseldorp, T. A.; Willoughby, D. S.; Ziegenfuss, T. N. (2021). "Common questions and misconceptions about creatine supplementation: what does the scientific evidence really show?". Journal of the International Society of Sports Nutrition. 18 (13): 13. doi:10.1186/s12970-021-00412-w. PMC 7871530. PMID 33557850.
- ^ Francaux M, Poortmans JR (December 2006). "Side effects of creatine supplementation in athletes". International Journal of Sports Physiology and Performance. 1 (4): 311–23. doi:10.1123/ijspp.1.4.311. PMID 19124889. S2CID 21330062.
- ^ Buford TW, Kreider RB, Stout JR, Greenwood M, Campbell B, Spano M, et al. (August 2007). "International Society of Sports Nutrition position stand: creatine supplementation and exercise". Journal of the International Society of Sports Nutrition. 4. jissn: 6. doi:10.1186/1550-2783-4-6. PMC 2048496. PMID 17908288.
- ^ Kreider RB, Kalman DS, Antonio J, Ziegenfuss TN, Wildman R, Collins R, et al. (13 June 2017). "International Society of Sports Nutrition position stand: safety and efficacy of creatine supplementation in exercise, sport, and medicine". Journal of the International Society of Sports Nutrition. 14: 18. doi:10.1186/s12970-017-0173-z. PMC 5469049. PMID 28615996.
- ^ Lopez RM, Casa DJ, McDermott BP, Ganio MS, Armstrong LE, Maresh CM (2009). "Does creatine supplementation hinder exercise heat tolerance or hydration status? A systematic review with meta-analyses". Journal of Athletic Training. 44 (2): 215–23. doi:10.4085/1062-6050-44.2.215. PMC 2657025. PMID 19295968.
- ^ Dalbo VJ, Roberts MD, Stout JR, Kerksick CM (July 2008). "Putting to rest the myth of creatine supplementation leading to muscle cramps and dehydration". British Journal of Sports Medicine. 42 (7): 567–73. doi:10.1136/bjsm.2007.042473. PMID 18184753. S2CID 12920206. Archived from the original on 19 June 2022. Retrieved 27 December 2021.
- ^ Antonio, Jose (2022). "Common questions and misconceptions about creatine supplementation: what does the scientific evidence really show?". Journal of the International Society of Sports Medicine. 18 (1): 13. doi:10.1186/s12970-021-00412-w. PMC 7871530. PMID 33557850.
- ^ Farquhar, William B.; Zambraski, Edward J. (2002). "Effects of creatine use on the athlete's kidney". Current Sports Medicine Reports. 1 (2): 103–106. doi:10.1249/00149619-200204000-00007. PMID 12831718.
- ^ de Souza E, Silva A, Pertille A, Reis Barbosa CG, Aparecida de Oliveira Silva J, de Jesus DV, et al. (November 2019). "Effects of Creatine Supplementation on Renal Function: A Systematic Review and Meta-Analysis". Journal of Renal Nutrition. 29 (6): 480–489. doi:10.1053/j.jrn.2019.05.004. PMID 31375416. S2CID 199388424.
- ^ Gualano B, de Salles Painelli V, Roschel H, Lugaresi R, Dorea E, Artioli GG, et al. (May 2011). "Creatine supplementation does not impair kidney function in type 2 diabetic patients: a randomized, double-blind, placebo-controlled, clinical trial". European Journal of Applied Physiology. 111 (5): 749–56. doi:10.1007/s00421-010-1676-3. PMID 20976468. S2CID 21335546.
- ^ Neves M, Gualano B, Roschel H, Lima FR, Lúcia de Sá-Pinto A, Seguro AC, et al. (June 2011). "Effect of creatine supplementation on measured glomerular filtration rate in postmenopausal women". Applied Physiology, Nutrition, and Metabolism. 36 (3): 419–22. doi:10.1139/h11-014. PMID 21574777.
- ^ Lugaresi R, Leme M, de Salles Painelli V, Murai IH, Roschel H, Sapienza MT, et al. (May 2013). "Does long-term creatine supplementation impair kidney function in resistance-trained individuals consuming a high-protein diet?". Journal of the International Society of Sports Nutrition. 10 (1): 26. doi:10.1186/1550-2783-10-26. PMC 3661339. PMID 23680457.
- ^ Kreider RB, Melton C, Rasmussen CJ, Greenwood M, Lancaster S, Cantler EC, et al. (February 2003). "Long-term creatine supplementation does not significantly affect clinical markers of health in athletes". Molecular and Cellular Biochemistry. 244 (1–2): 95–104. doi:10.1023/A:1022469320296. PMID 12701816. S2CID 25947100.
- ^ Cancela P, Ohanian C, Cuitiño E, Hackney AC (September 2008). "Creatine supplementation does not affect clinical health markers in football players". British Journal of Sports Medicine. 42 (9): 731–5. doi:10.1136/bjsm.2007.030700. PMID 18780799. S2CID 20876433.
- ^ Carvalho AP, Molina GE, Fontana KE (August 2011). "Creatine supplementation associated with resistance training does not alter renal and hepatic functions". Revista Brasileira de Medicina do Esporte. 17 (4): 237–241. doi:10.1590/S1517-86922011000400004. ISSN 1517-8692.
- ^ Mayhew DL, Mayhew JL, Ware JS (December 2002). "Effects of long-term creatine supplementation on liver and kidney functions in American college football players". International Journal of Sport Nutrition and Exercise Metabolism. 12 (4): 453–60. doi:10.1123/ijsnem.12.4.453. PMID 12500988.
- ^ Thorsteinsdottir B, Grande JP, Garovic VD (October 2006). "Acute renal failure in a young weight lifter taking multiple food supplements, including creatine monohydrate". Journal of Renal Nutrition. 16 (4): 341–5. doi:10.1053/j.jrn.2006.04.025. PMID 17046619.
- ^ Taner B, Aysim O, Abdulkadir U (February 2011). "The effects of the recommended dose of creatine monohydrate on kidney function". NDT Plus. 4 (1): 23–4. doi:10.1093/ndtplus/sfq177. PMC 4421632. PMID 25984094.
- ^ Barisic N, Bernert G, Ipsiroglu O, Stromberger C, Müller T, Gruber S, et al. (June 2002). "Effects of oral creatine supplementation in a patient with MELAS phenotype and associated nephropathy". Neuropediatrics. 33 (3): 157–61. doi:10.1055/s-2002-33679. PMID 12200746. S2CID 9250579.
- ^ Rodriguez NR, Di Marco NM, Langley S (March 2009). "American College of Sports Medicine position stand. Nutrition and athletic performance". Medicine and Science in Sports and Exercise. 41 (3): 709–31. doi:10.1249/MSS.0b013e31890eb86. PMID 19225360.
- ^ ML.Guerrero, J.Beron, B.Spindler, P.Grosscurth, T.Wallimann and F.Verrey.Metabolic support of Na+ pump in apically permeabilized A6 kidney cell epithelia: role of creatine kinase.In: Am J Physiol. 1997 Feb;272(2 Pt 1):C697-706. doi:10.1152/ajpcell.1997.272.2.C697, PMID 9124314
- ^ T. Wallimann, U. Riek, M. M. Möddel: Intradialytic creatine supplementation: A scientific rationale for improving the health and quality of life of dialysis patients.In: Medical Hypotheses 2017 Febr;99, S. 1-14. doi:10.1016/j.mehy.2016.12.002, PMID 28110688.
- ^ Moreta S, Prevarin A, Tubaro F (June 2011). "Levels of creatine, organic contaminants and heavy metals in creatine dietary supplements". Food Chemistry. 126 (3): 1232–1238. doi:10.1016/j.foodchem.2010.12.028.
- ^ Persky AM, Rawson ES (2007). "Safety of Creatine Supplementation". Creatine and Creatine Kinase in Health and Disease. Subcellular Biochemistry. Vol. 46. pp. 275–89. doi:10.1007/978-1-4020-6486-9_14. ISBN 978-1-4020-6485-2. PMID 18652082.
- ^ "Heterocyclic Amines in Cooked Meats". National Cancer Institute. 15 September 2004. Archived from the original on 21 December 2010. Retrieved 9 August 2007.
- ^ "Chemicals in Meat Cooked at High Temperatures and Cancer Risk". National Cancer Institute. 2 April 2018. Archived from the original on 6 November 2011. Retrieved 22 February 2015.
- ^ Dahl O (1 July 1963). "Meat Quality Measurement, Creatine Content as an Index of Quality of Meat Products". Journal of Agricultural and Food Chemistry. 11 (4): 350–355. doi:10.1021/jf60128a026.
- ^ Gießing J (20 February 2019). Kreatin: Eine natürliche Substanz und ihre Bedeutung für Muskelaufbau, Fitness und Anti-Aging. BoD – Books on Demand. pp. 135–136, 207. ISBN 9783752803969. Archived from the original on 19 June 2022. Retrieved 27 December 2021.
External links
[edit]- Creatine bound to proteins in the PDB
