Herpes simplex encephalitis
| Herpes simplex encephalitis | |
|---|---|
| Other names | Herpes encephalitis, Herpesviral encephalitis |
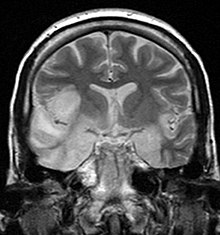 | |
| Coronal T2-weighted MR image shows high signal in the temporal lobes including hippocampal formations and parahippogampal gyrae, insulae, and right inferior frontal gyrus. A brain biopsy was performed and the histology was consistent with encephalitis. PCR was repeated on the biopsy specimen and was positive for HSV | |
| Specialty | Infectious diseases |
Herpes simplex encephalitis (HSE), or simply herpes encephalitis, is encephalitis due to herpes simplex virus. It is estimated to affect at least 1 in 500,000 individuals per year,[1] and some studies suggest an incidence rate of 5.9 cases per 100,000 live births.[2]
About 90% of cases of herpes encephalitis are caused by herpes simplex virus-1 (HSV-1), the same virus that causes cold sores. According to a 2006 estimate, 57% of American adults were infected with HSV-1,[3] which is spread through droplets, casual contact and sometimes sexual contact, though most infected people never have cold sores. The rest of cases are due to HSV-2, which is typically spread through sexual contact and is the cause of genital herpes.
Two-thirds of HSE cases occur in individuals already seropositive for HSV-1, few of whom (only 10%) have history of recurrent orofacial herpes, while about one third of cases results from an initial infection by HSV-1, predominantly occurring in individuals under the age of 18.[1] Approximately half of individuals who develop HSE are over 50 years of age.[4]
The most common cause for encephalitis in children and adults is HSV-1. However, encephalitis found in newborns and immunocompromised individuals is mainly caused by HSV-2.[5]
Signs and symptoms
[edit]Most individuals with HSE show a decrease in their level of consciousness and an altered mental state presenting as confusion, and changes in personality. Increased numbers of white blood cells can be found in patient's cerebrospinal fluid, without the presence of pathogenic bacteria and fungi. Patients typically have a fever[1] and may have seizures. The electrical activity of the brain changes as the disease progresses, first showing abnormalities in one temporal lobe of the brain, which spread to the other temporal lobe 7–10 days later.[1] Imaging by CT or MRI shows characteristic changes in the temporal lobes (see Figure). After the first symptoms appear, patients might lose their sense of smell. This can also be accompanied by the inability to read, write, or speak coherently, and understand verbal speech.[6]
Definite diagnosis requires testing of the cerebrospinal fluid (CSF) by a lumbar puncture (spinal tap) for presence of the virus. The testing takes several days to perform, and patients with suspected Herpes encephalitis should be treated with acyclovir immediately while waiting for test results. Atypical stroke-like presentation of HSV encephalitis has been described as well and the clinicians should be aware that HSV encephalitis can mimic a stroke.[7]
Associated conditions
[edit]Herpesviral encephalitis can serve as a trigger of anti-NMDA receptor encephalitis.[8] About 30% of HSE patients develop this secondary immunologic reaction,[9] which is associated with impaired neurocognitive recovery.[10]
Epidemiology
[edit]The annual incidence of herpesvial encephalitis is from 2 to 4 cases per 1 million population.[11]
Pathophysiology
[edit]HSE is thought to be caused by the transmission of virus from a peripheral site on the face following HSV-1 reactivation, along a nerve axon, to the brain.[1] The virus lies dormant in the ganglion of the trigeminal cranial nerve, but the reason for reactivation, and its pathway to gain access to the brain, remains unclear, though changes in the immune system caused by stress clearly play a role in animal models of the disease. The olfactory nerve may also be involved in HSE,[12] which may explain its predilection for the temporal lobes of the brain, as the olfactory nerve sends branches there. In horses, a single-nucleotide polymorphism is sufficient to allow the virus to cause neurological disease;[13] but no similar mechanism has been found in humans.
Diagnosis
[edit]Brain CT scan (with/without contrast). Complete prior to lumbar puncture to exclude significantly increased ICP, obstructive hydrocephalus, mass effect[14]
Brain MRI—Increased T2 signal intensity in frontotemporal region → viral (HSV) encephalitis[15]
Treatment
[edit]Herpesviral encephalitis can be treated with high-dose intravenous acyclovir, which should be infused 10 mg/kg(adult) over 1 hour to avoid kidney failure. Without treatment, HSE results in rapid death in approximately 70% of cases; survivors suffer severe neurological damage.[1] When treated, HSE is still fatal in one-third of cases, and causes serious long-term neurological damage in over half of survivors. Twenty percent of treated patients recover with minor damage. Only a small population of untreated survivors (2.5%) regain completely normal brain function.[4] Many amnesic cases in the scientific literature have etiologies involving HSE.
Earlier treatment (within 48 hours of symptom onset) improves the chances of a good recovery. Rarely, treated individuals can have relapse of infection weeks to months later. There is evidence that aberrant inflammation triggered by herpes simplex can result in granulomatous inflammation in the brain, which responds to steroids.[16] While the herpes virus can be spread, encephalitis itself is not infectious. Other viruses can cause similar symptoms of encephalitis, though usually milder (Herpesvirus 6, varicella zoster virus, Epstein-Barr, cytomegalovirus, coxsackievirus, etc.).[17][11]
References
[edit]- ^ a b c d e f Whitley RJ (September 2006). "Herpes simplex encephalitis: adolescents and adults". Antiviral Research. 71 (2–3): 141–148. doi:10.1016/j.antiviral.2006.04.002. PMID 16675036.
- ^ Kropp RY, Wong T, Cormier L, Ringrose A, Burton S, Embree JE, Steben M (June 2006). "Neonatal herpes simplex virus infections in Canada: results of a 3-year national prospective study". Pediatrics. 117 (6): 1955–1962. doi:10.1542/peds.2005-1778. PMID 16740836. S2CID 9632498.
- ^ Xu F, Sternberg MR, Kottiri BJ, McQuillan GM, Lee FK, Nahmias AJ, et al. (August 2006). "Trends in herpes simplex virus type 1 and type 2 seroprevalence in the United States". JAMA. 296 (8): 964–973. doi:10.1001/jama.296.8.964. PMID 16926356.
- ^ a b Whitley RJ, Gnann JW (February 2002). "Viral encephalitis: familiar infections and emerging pathogens". Lancet. 359 (9305): 507–513. doi:10.1016/S0140-6736(02)07681-X. PMID 11853816. S2CID 5980017.
- ^ Ak, Ajith Kumar; Mendez, Magda D. (2023), "Herpes Simplex Encephalitis", StatPearls, Treasure Island (FL): StatPearls Publishing, PMID 32491575, retrieved 2023-10-02
- ^ "Encephalitis, Herpes Simplex - Symptoms, Causes, Treatment | NORD". rarediseases.org. Retrieved 2023-10-02.
- ^ Gupta, Harsh; Malhotra, Samira; Batra, Amit (2019-04-15). "An Unusual Stroke-Like Presentation of HSV Encephalitis". The Journal of the Arkansas Medical Society. 115 – via ResearchGate.
- ^ Dalmau J, Armangué T, Planagumà J, Radosevic M, Mannara F, Leypoldt F, et al. (November 2019). "An update on anti-NMDA receptor encephalitis for neurologists and psychiatrists: mechanisms and models". The Lancet. Neurology. 18 (11): 1045–1057. doi:10.1016/S1474-4422(19)30244-3. PMID 31326280. S2CID 197464804.
- ^ Prüss H, Finke C, Höltje M, Hofmann J, Klingbeil C, Probst C, et al. (December 2012). "N-methyl-D-aspartate receptor antibodies in herpes simplex encephalitis". Annals of Neurology. 72 (6): 902–911. doi:10.1002/ana.23689. PMC 3725636. PMID 23280840.
- ^ Westman G, Studahl M, Ahlm C, Eriksson BM, Persson B, Rönnelid J, et al. (November 2016). "N-methyl-d-aspartate receptor autoimmunity affects cognitive performance in herpes simplex encephalitis". Clinical Microbiology and Infection. 22 (11): 934–940. doi:10.1016/j.cmi.2016.07.028. PMID 27497810.
- ^ a b Ajith Kumar AK, Mendez MD (14 March 2022). "Herpes Simplex Encephalitis". StatPearls. PMID 32491575.
- ^ Dinn JJ (November 1980). "Transolfactory spread of virus in herpes simplex encephalitis". British Medical Journal. 281 (6252): 1392. doi:10.1136/bmj.281.6252.1392. PMC 1715042. PMID 7437807.
- ^ Van de Walle GR, Goupil R, Wishon C, Damiani A, Perkins GA, Osterrieder N (July 2009). "A single-nucleotide polymorphism in a herpesvirus DNA polymerase is sufficient to cause lethal neurological disease". The Journal of Infectious Diseases. 200 (1): 20–25. doi:10.1086/599316. PMID 19456260.
- ^ "Herpes Meningoencephalitis". Johns Hopkins Medicine. 19 November 2019. Retrieved 13 July 2021.
- ^ "Encephalitis". The Lecturio Medical Concept Library. Retrieved 13 July 2021.
- ^ Varatharaj A, Nicoll JA, Pelosi E, Pinto AA (April 2017). "Corticosteroid-responsive focal granulomatous herpes simplex type-1 encephalitis in adults". Practical Neurology. 17 (2): 140–144. doi:10.1136/practneurol-2016-001474. PMID 28153849. S2CID 12859405.
- ^ "Encephalitis". Mayo Foundation for Medical Education and Research (MFMER). Retrieved 13 July 2021.
External links
[edit]- Kumar M, Hill JM, Clement C, Varnell ED, Thompson HW, Kaufman HE (December 2009). "A double-blind placebo-controlled study to evaluate valacyclovir alone and with aspirin for asymptomatic HSV-1 DNA shedding in human tears and saliva". Investigative Ophthalmology & Visual Science. 50 (12): 5601–5608. doi:10.1167/iovs.09-3729. PMC 2895993. PMID 19608530.
- Kaufman HE, Azcuy AM, Varnell ED, Sloop GD, Thompson HW, Hill JM (January 2005). "HSV-1 DNA in tears and saliva of normal adults". Investigative Ophthalmology & Visual Science. 46 (1): 241–247. doi:10.1167/iovs.04-0614. PMC 1200985. PMID 15623779.
- Hill JM, Zhao Y, Clement C, Neumann DM, Lukiw WJ (October 2009). "HSV-1 infection of human brain cells induces miRNA-146a and Alzheimer-type inflammatory signaling". NeuroReport. 20 (16): 1500–1505. doi:10.1097/WNR.0b013e3283329c05. PMC 2872932. PMID 19801956.<ref>
