Phosphoryl fluoride
Appearance
(Redirected from Phosphorus oxyfluoride)
| |||
| Names | |||
|---|---|---|---|
| IUPAC names
Phosphoryl trifluoride
Phosphorus trifluoride oxide | |||
Other names
| |||
| Identifiers | |||
3D model (JSmol)
|
|||
| ChemSpider | |||
| ECHA InfoCard | 100.033.419 | ||
| EC Number |
| ||
PubChem CID
|
|||
| UNII | |||
CompTox Dashboard (EPA)
|
|||
| |||
| |||
| Properties | |||
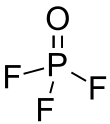
| POF3 | |||
| Molar mass | 103.9684 g/mol | ||
| Appearance | Colourless gas | ||
| Boiling point | −39.7 °C (−39.5 °F; 233.5 K) | ||
| Reacts | |||
| Solubility | Reacts with alcohol and acid, soluble in diethyl ether and hydrocarbons | ||
| 1.76 D[1] | |||
| Structure | |||
| Tetrahedral at the P atom | |||
| Hazards | |||
| Occupational safety and health (OHS/OSH): | |||
Main hazards
|
Poison, corrosive, can form HF on contact with H2O | ||
| GHS labelling: | |||
   
| |||
| Danger | |||
| H302, H314, H330, H372 | |||
| P260, P264, P270, P271, P280, P284, P301+P312, P301+P330+P331, P303+P361+P353, P304+P340, P305+P351+P338, P310, P314, P320, P321, P330, P363, P403+P233, P405, P501 | |||
| NFPA 704 (fire diamond) | |||
| Safety data sheet (SDS) | ICSC 0190 | ||
| Related compounds | |||
Related compounds
|
|||
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |||
Phosphoryl fluoride (commonly called phosphorus oxyfluoride) is a compound with the chemical formula POF3. It is a colorless gas that hydrolyzes rapidly. It has a critical temperature of 73 °C and a critical pressure of 4.25 bars.[1]
Synthesis and reactions
[edit]Phosphorus oxyfluoride is prepared by partial hydrolysis of phosphorus pentafluoride.
Phosphorus oxyfluoride is the progenitor of the simple fluorophosphoric acids by hydrolysis. The sequence starts with difluorophosphoric acid:
- POF3 + H2O → HPO2F2 + HF
The next steps give monofluorophosphoric acid and phosphoric acid:
- HPO2F2 + H2O → H2PO3F + HF
- H2PO3F + H2O → H3PO4 + HF
Phosphoryl fluoride combines with dimethylamine to produce dimethylaminophosphoryl difluoride (H3C−)2N−P(=O)F2 and difluorophosphate and hexafluorophosphate ions.[2]
References
[edit]- ^ a b "Phosphoryl fluoride".
- ^ Cavell, R. G. (1968). "Chemistry of phosphorus fluorides. Part III. The reaction of thiophosphoryl-fluoride with dimethylamine and some properties of the dimethylaminothio- phosphoryl fluorides". Canadian Journal of Chemistry. 46 (4): 613. doi:10.1139/v68-100.
Wikimedia Commons has media related to Phosphoryl fluoride.