Potassium hexachloroplatinate: Difference between revisions
Appearance
Content deleted Content added
No edit summary |
No edit summary |
||
| Line 23: | Line 23: | ||
| Melting_notes = (decomposes) |
| Melting_notes = (decomposes) |
||
| BoilingPt = |
| BoilingPt = |
||
| Solubility = 0.89 g/100ml H2O <ref name=Grinberg>{{cite journal | author = Grinberg, A. A.; Sibirskaya, V. V. | title = Solubility of hexammine and hexahalo platinum(IV) complexes | journal = Zhurnal Neorganicheskoi Khimii | year = 1967 | volume = 12 | pages = 2069-2071}}</ref> |
|||
| Solubility = 0.89 g/100ml H2O |
|||
}} |
}} |
||
|Section3={{Chembox Hazards |
|Section3={{Chembox Hazards |
||
Revision as of 16:56, 4 May 2015
| |

| |
| Identifiers | |
|---|---|
3D model (JSmol)
|
|
| ECHA InfoCard | 100.037.239 |
PubChem CID
|
|
| RTECS number |
|
CompTox Dashboard (EPA)
|
|
| |
| Properties | |
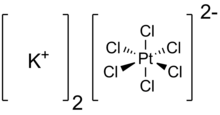
| K2PtCl6 | |
| Molar mass | 485.99 g/mol |
| Appearance | orange to yellow solid |
| Density | 3.344 g/cm3 |
| Melting point | 250 °C (482 °F; 523 K) |
| 0.89 g/100ml H2O [1] | |
| Hazards | |
| Flash point | 250 °C (482 °F; 523 K) |
| Related compounds | |
Other anions
|
Potassium tetrachloroplatinate |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
Potassium hexachloroplatinate, an inorganic compound, is an example of a comparatively insoluble potassium salt.
The precipitation of this compound from solutions of hexachloroplatinic acid was formerly used for the determination of potassium by gravimetric analysis.[2]
Potassium hexachloroplatinate may be reduced by hydrazine dihydrochloride to give the corresponding tetrachloroplatinate salt.[3]
References
- ^ Grinberg, A. A.; Sibirskaya, V. V. (1967). "Solubility of hexammine and hexahalo platinum(IV) complexes". Zhurnal Neorganicheskoi Khimii. 12: 2069–2071.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ G. F. Smith, J. L. Gring (1933). "The Separation and Determination of the Alkali Metals Using Perchloric Acid. V. Perchloric Acid and Chloroplatinic Acid in the Determination of Small Amounts of Potassium in the Presence of Large Amounts of Sodium". J. Am. Chem. Soc. 55 (10): 3957–3961. doi:10.1021/ja01337a007.
- ^ Keller, R. N.; Moeller, T. (1963). "Potassium Tetrachloroplatinate(II)". Inorg. Synth. Inorganic Syntheses. 7: 247–250. doi:10.1002/9780470132333.ch79. ISBN 978-0-470-13233-3.
{{cite journal}}: CS1 maint: multiple names: authors list (link)
