From Wikipedia, the free encyclopedia
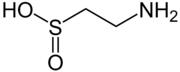
Hypotaurine
Names
IUPAC name
2-Aminoethanesulfinic acid
Identifiers
ChEBI
ChemSpider
ECHA InfoCard 100.155.825
KEGG
UNII
InChI=1S/C2H7NO2S/c3-1-2-6(4)5/h1-3H2,(H,4,5)
Y Key: VVIUBCNYACGLLV-UHFFFAOYSA-N
Y InChI=1/C2H7NO2S/c3-1-2-6(4)5/h1-3H2,(H,4,5)
Key: VVIUBCNYACGLLV-UHFFFAOYAM
Properties
C2 H7 NO2 S
Molar mass
109.15 g/mol
Except where otherwise noted, data are given for materials in their
standard state (at 25 °C [77 °F], 100 kPa).
Chemical compound
Hypotaurine is a sulfinic acid that is an intermediate in the biosynthesis of taurine . Like taurine, it also acts as an endogenous neurotransmitter via action on the glycine receptors .[ 1]
Hypotaurine is derived from cysteine (and homocysteine ). In mammals, the biosynthesis of hypotaurine from cysteine occurs in the pancreas. In the cysteine sulfinic acid pathway, cysteine is first oxidized to its sulfinic acid, catalyzed by the enzyme cysteine dioxygenase . Cysteine sulfinic acid, in turn, is decarboxylated by sulfinoalanine decarboxylase to form hypotaurine . Hypotaurine is enzymatically oxidized to yield taurine by hypotaurine dehydrogenase .[ 2]
Hypotaurine (3) is an intermediate in the conversion of cysteine (1) to taurine (4).
References
^ Kalir, Asher; Kalir, Henry H. "Biological activity of sulfinic acid derivatives" in Chemistry of Sulphinic Acids, Esters Their Derivatives Edited by Patai, Saul. Wiley, New York, 1990, pp. 665.
^ Sumizu K (1962). "Oxidation of hypotaurine in rat liver". Biochim. Biophys. Acta . 63 : 210–212. doi :10.1016/0006-3002(62)90357-8 . PMID 13979247 .
Receptor (ligands )
GlyR Tooltip Glycine receptor
Positive modulators: Alcohols (e.g., brometone , chlorobutanol (chloretone) , ethanol (alcohol) , tert -butanol (2M2P)tribromoethanol , trichloroethanol , trifluoroethanol )Alkylbenzene sulfonate Anandamide Barbiturates (e.g., pentobarbital , sodium thiopental )Chlormethiazole D12-116 Dihydropyridines (e.g., nicardipine )Etomidate Ginseng constituents (e.g., ginsenosides (e.g., ginsenoside-Rf ))Glutamic acid (glutamate) Ivermectin Ketamine Neuroactive steroids (e.g., alfaxolone , pregnenolone (eltanolone) , pregnenolone acetate , minaxolone , ORG-20599 )Nitrous oxide Penicillin G Propofol Tamoxifen Tetrahydrocannabinol Triclofos Tropeines (e.g., atropine , bemesetron , cocaine , LY-278584 , tropisetron , zatosetron )Volatiles /gases (e.g., chloral hydrate , chloroform , desflurane , diethyl ether (ether) , enflurane , halothane , isoflurane , methoxyflurane , sevoflurane , toluene , trichloroethane (methyl chloroform) , trichloroethylene )Xenon Zinc Antagonists: 2-Aminostrychnine 2-Nitrostrychnine 4-Phenyl-4-formyl-N-methylpiperidine αEMBTL Bicuculline Brucine Cacotheline Caffeine Colchicine Colubrine Cyanotriphenylborate Dendrobine Diaboline Endocannabinoids (e.g., 2-AG , anandamide (AEA) )Gaboxadol (THIP) Gelsemine iso-THAZ Isobutyric acid Isonipecotic acid Isostrychnine Laudanosine N-Methylbicuculline N-Methylstrychnine N,N-Dimethylmuscimol Nipecotic acid Pitrazepin Pseudostrychnine Quinolines (e.g., 4-hydroxyquinoline , 4-hydroxyquinoline-3-carboxylic acid , 5,7-CIQA , 7-CIQ , 7-TFQ , 7-TFQA )RU-5135 Sinomenine Strychnine Thiocolchicoside Tutin Negative modulators: Amiloride Benzodiazepines (e.g., bromazepam , clonazepam , diazepam , flunitrazepam , flurazepam )Corymine Cyanotriphenylborate Daidzein Dihydropyridines (e.g., nicardipine , nifedipine , nitrendipine )Furosemide Genistein Ginkgo constituents (e.g., bilobalide , ginkgolides (e.g., ginkgolide A , ginkgolide B , ginkgolide C , ginkgolide J , ginkgolide M ))Imipramine NBQX Neuroactive steroids (e.g., 3α-androsterone sulfate , 3β-androsterone sulfate , deoxycorticosterone , DHEA sulfate , pregnenolone sulfate , progesterone )Opioids (e.g., codeine , dextromethorphan , dextrorphan , levomethadone , levorphanol , morphine , oripavine , pethidine , thebaine )Picrotoxin (i.e., picrotin and picrotoxinin )PMBA Riluzole Tropeines (e.g., bemesetron , LY-278584 , tropisetron , zatosetron )Verapamil Zinc NMDAR Tooltip N-Methyl-D-aspartate receptor
Transporter (blockers )
GlyT1 Tooltip Glycine transporter 1 GlyT2 Tooltip Glycine transporter 2