Talk:Genetically modified crops
| This is the talk page for discussing improvements to the Genetically modified crops article. This is not a forum for general discussion of the article's subject. |
Article policies
|
| Find sources: Google (books · news · scholar · free images · WP refs) · FENS · JSTOR · TWL |
| Archives: 1, 2, 3, 4, 5, 6, 7Auto-archiving period: 4 months |
| This It is of interest to the following WikiProjects: | |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| |||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 | The Arbitration Committee has authorized discretionary sanctions regarding the language used to summarize the safety and regulation of genetically modified food on this and related articles (including talk pages).
|
Wiki Education assignment: Technology and Culture
[edit]![]() This article was the subject of a Wiki Education Foundation-supported course assignment, between 21 August 2023 and 15 December 2023. Further details are available on the course page. Student editor(s): Kimjammer (article contribs). Peer reviewers: Andretam0027, Straws12!.
This article was the subject of a Wiki Education Foundation-supported course assignment, between 21 August 2023 and 15 December 2023. Further details are available on the course page. Student editor(s): Kimjammer (article contribs). Peer reviewers: Andretam0027, Straws12!.
— Assignment last updated by Thecanyon (talk) 05:33, 12 December 2023 (UTC)
Any of this content useful? Cut from food security.
[edit]I've just cut out this content from the food security article as it was digressing. There might be bits in here that could be added to this article? Or maybe it's all here already and would only be repetitive. Note: most of this content had been added by students several years ago so it's likely not very up to date:
++++++++++
Use of genetically modified (GM) crops:
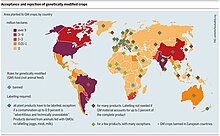
The area sown to genetically engineered crops in developing countries is rapidly catching up with the area sown in industrial nations. According to the International Service for the Acquisition of Agri-biotech Applications (ISAAA), GM crops were grown by approximately 8.5 million farmers in 21 countries in 2005; up from 8.25 million farmers in 17 countries in 2004.[citation needed]
Opposition to GM crops
[edit]Based on evidence from previous attempts, there is a likely lack of transferability of one type of GM crop from one region to another. For example, modified crops that have proven successful in Asia from the Green Revolution have failed when tried in regions of Africa.[1] More research must be done regarding the specific requirements of growing a specific crop in a specific region.[citation needed]
There is also a drastic lack of education given to governments, farmers, and the community about the science behind GM crops, as well as suitable growing practices. In most relief programs, farmers are given seeds with little explanation and little attention is paid to the resources available to them or even laws that prohibit them from distributing produce. Governments are often not advised on the economic and health implications that come with growing GM crops and are then left to make judgments on their own. Because they have so little information regarding these crops, they usually shy away from allowing them or do not take the time and effort required to regulate their use. Members of the community that will then consume the produce from these crops are also left in the dark about what these modifications mean and are often scared off by their 'unnatural' origins. This has resulted in failure to properly grow crops as well as strong opposition to the unknown practices.[2]

A study published in June 2016 evaluated the status of the implementation of Golden Rice, which was first developed in the 1990s to produce higher levels of Vitamin A than its non-GMO counterparts. This strain of rice was designed so that malnourished women and children in third-world countries who were more susceptible to deficiencies could easily improve their Vitamin A intake levels and prevent blindness, which is a common result. Golden Rice production was centralized in the Philippines, yet there have been many hurdles to jump in order to get production moving. The study showed that the project is far behind schedule and is not living up to its expectations. Although research on Golden Rice continues, the country has moved forward with other non-GMO initiatives to address the Vitamin A deficiency problem that is so pervasive in that region.[3][4]
Many anti-GMO activists argue that the use of GM crops decreases biodiversity among plants. Livestock biodiversity is also threatened by the modernization of agriculture and the focus on more productive major breeds. Therefore, efforts have been made by governments and non-governmental organizations to conserve livestock biodiversity through strategies such as cryoconservation of animal genetic resources.[5][6]
Support of GM crops
[edit]Many GM crop success stories exist, primarily in developed nations like the US, China, and various countries in Europe. Common GM crops include cotton, maize, and soybeans, all of which are grown throughout North and South America as well as regions of Asia.[7] Modified cotton crops, for example, have been altered such that they are resistant to pests, can be grown in more extreme heat, cold, or drought, and produce longer, stronger fibers to be used in textile production.[8]
One of the biggest threats to rice, which is a staple food crop especially in India and other countries within Asia, is blast disease, which is a fungal infection that causes lesions to form on all parts of the plant.[9] A genetically engineered strain of rice has been developed so that it is resistant to blast, greatly improving the crop yield of farmers and allowing rice to be more accessible to everyone.[10] Some other crops have been modified such that they produce higher yields per plant or that they require less land for growing. The latter can be helpful in extreme climates with little arable land and also decreases deforestation, as fewer trees need to be cut down in order to make room for crop fields.[11] Others yet have been altered such that they do not require the use of insecticides or fungicides. This addresses various health concerns associated with such pesticides and can also work to improve biodiversity within the area in which these crops are grown.[12]
In a review of Borlaug's 2000 publication entitled Ending world hunger: the promise of biotechnology and the threat of antiscience zealotry,[13] the authors argued that Borlaug's warnings were still true in 2010,[14]
GM crops are as natural and safe as today's bread wheat, opined Dr. Borlaug, who also reminded agricultural scientists of their moral obligation to stand up to the antiscience crowd and warn policy makers that global food insecurity will not disappear without this new technology and ignoring this reality global food insecurity would make future solutions all the more difficult to achieve.
— Rozwadowski and Kagale
Research conducted by the GMO Risk Assessment and Communication of Evidence (GRACE) program through the EU between 2007 and 2013 focused on many uses of GM crops and evaluated many facets of their effects on human, animal, and environmental health.[citation needed]
The body of scientific evidence concluding that GM foods are safe to eat and do not pose environmental risks is wide. Findings from the International Council of Scientists (2003) that analyzed a selection of approximately 50 science-based reviews concluded that "currently available genetically modified foods are safe to eat," and "there is no evidence of any deleterious environmental effects having occurred from the trait/species combinations currently available."[15] The United Nations Food and Agriculture Organization (FAO) supported the same consensus a year later in addition to recommending the extension of biotechnology to the developing world.[16] Similarly, the Royal Society (2003) and British Medical Association (2004) found no adverse health effects of consuming genetically modified foods.[17][18] These findings supported the conclusions of earlier studies by the European Union Research Directorate, a compendium of 81 scientific studies conducted by more than 400 research teams did not show "any new risks to human health or the environment, beyond the usual uncertainties of conventional plant breeding."[19] Likewise, the Organization for Economic Cooperation and Development in Europe (OECD) and the Nuffield Council on Bioethics (1999) did not find that genetically modified foods posed a health risk.[20][21] EMsmile (talk) 22:44, 13 December 2023 (UTC)
References
- ^ Fischer, Klara (2016-07-01). "Why new crop technology is not scale-neutral—A critique of the expectations for a crop-based African Green Revolution". Research Policy. 45 (6): 1185–1194. doi:10.1016/j.respol.2016.03.007.
- ^ Wedding, K. (2013). Pathways to productivity: The role of GMOs for food security in Kenya, Tanzania, and Uganda. Rowman and Littlefield.
- ^ Stone, Glenn Davis; Glover, Dominic (2016-04-16). "Disembedding grain: Golden Rice, the Green Revolution, and heirloom seeds in the Philippines". Agriculture and Human Values. 34: 87–102. doi:10.1007/s10460-016-9696-1. ISSN 0889-048X. S2CID 16474458.
- ^ "Genetically modified Golden Rice falls short on lifesaving promises | The Source | Washington University in St. Louis". 2016-06-02. Archived from the original on 2019-12-12. Retrieved 2016-07-31.
- ^ Global Plan of Action for Animal Genetic Resources and the Interlaken Declaration. Rep. Rome: Food and Agriculture Organization of the United Nations, 2007. FAO. Web.
- ^ Cryoconservation of Animal Genetic Resources.Rep. Rome: Food and Agriculture Organization of the United Nations, 2012. FAO Animal Production and Health Guidelines No. 12. Print.
- ^ www.gmo-compass.org. "GMO Crop Growing: Growing Around the World". www.gmo-compass.org. Archived from the original on 2016-08-06. Retrieved 2016-07-31.
- ^ www.gmo-compass.org. "Cotton – GMO Database". www.gmo-compass.org. Archived from the original on 2016-07-31. Retrieved 2016-07-31.
- ^ TeBeest, D. (2007). "Rice Blast". The Plant Health Instructor. doi:10.1094/phi-i-2007-0313-07. Archived from the original on 2016-06-29.
- ^ Shew, Aaron M.; Nalley, Lawton L.; Danforth, Diana M.; Dixon, Bruce L.; Nayga, Rodolfo M.; Delwaide, Anne-Cecile; Valent, Barbara (2016-01-01). "Are all GMOs the same? Consumer acceptance of cisgenic rice in India" (PDF). Plant Biotechnology Journal. 14 (1): 4–7. doi:10.1111/pbi.12442. hdl:2097/33968. ISSN 1467-7652. PMID 26242818. Archived (PDF) from the original on 2017-09-23. Retrieved 2020-08-19.
- ^ Makinde, D. (2009). "Status of Biotechnology in Africa: Challenges and Opportunities". Asian Biotechnology and Development Review. 11 (3).
- ^ Gerasimova, Ksenia (2015-06-11). "Debates on Genetically Modified Crops in the Context of Sustainable Development". Science and Engineering Ethics. 22 (2): 525–547. doi:10.1007/s11948-015-9656-y. ISSN 1353-3452. PMID 26062746. S2CID 22512421.
- ^ Borlaug, N.E. (2000), "Ending world hunger: the promise of biotechnology and the threat of antiscience zealotry", Plant Physiology, 124 (2): 487–490, doi:10.1104/pp.124.2.487, PMC 1539278, PMID 11027697
- ^ Rozwadowski, Kevin; Kagale, Sateesh, Global Food Security: The Role of Agricultural Biotechnology Commentary (PDF), Saskatoon, Saskatchewan: Saskatoon Research Centre, Agriculture and Agri-Food Canada, archived from the original (PDF) on 24 September 2015, retrieved 12 January 2014
- ^ International Council for Science, "New Genetics, Food and Agriculture: Scientific Discoveries – Societal Dilemmas," 2003.
- ^ Entine, J. (ed), "Let them Eat Precaution: How politics is undermining the genetic revolution in agriculture," The AEI Press: Washington, DC, 2005.
- ^ Royal Society, "Royal Society Submission to the Government's GM Science Review," Royal Society, Policy Document: 14/03, May 2003
- ^ British Medical Association, Board of Science and Education, "Genetically modified foods and health: a second interim statement," British Medical Association, May 2004.
- ^ European Union (EU) Research Directorate, GMOs: Are there any Risks?. EU Commission press briefing, 9 October 2001. Accessed at: http://europa.eu.int/comm/research/index.html Archived 2006-04-25 at the Wayback Machine
- ^ Organization for Economic Cooperation and Development (OECD) "GM Food Safety: Facts, Uncertainties, and Assessment, Rapporteurs' Summary." The OECD Edinburgh Conference on the Scientific and Health Aspects of Genetically Modified Foods, 28 February – 1 March, 2000.
- ^ Millstone, E., and J. Abraham. 1988. Additives: A guide for everyone. London: Penguin. Nuffield Council on Bioethics "Genetically modified crops: the ethical and social issues," 1999.
EMsmile (talk) 22:44, 13 December 2023 (UTC)
Needs updating?
[edit]Much of the information currently given is now 10-20 years old and possibly out of date. I will try to find some newer scientific sources. Mary Mark Ockerbloom (talk) 14:46, 30 May 2024 (UTC)
- Thanks for doing this. If anything needs to be updated in re: WP:GMORFC, I'd be very interested in discussing it. --Tryptofish (talk) 19:25, 30 May 2024 (UTC)
- I'd like to help out a bit too. I wanted to clean it up the article a little but I'm mindful that this is a sensitive page so wanted to proceed carefully. I just wanted to ask should this page mention gm animals as it is a dedicated crops page (eg in the history section)? I wanted to remove mention to animals but perhaps cross reference to Genetically modified food. -- Sporesalad (talk) 00:56, 3 June 2024 (UTC)
- Thanks. That's a good question, about animals. I would take "crops" to mean plants only, but I can see an argument to the contrary, so I'd like to hear more opinions. --Tryptofish (talk) 20:46, 3 June 2024 (UTC)
- I'd go with largely plants only, though there's weird edge cases like algae that aren't really plants. Functionally, I'd say farmed primary producers.
- If there's mention of genetically modified animals, it's probably best only when there's a topic here that covers both, but I wouldn't delve into animals here in significant depth. People may talk about a "crop" of calves in the beef cattle world, but that's more jargon than anything. KoA (talk) 21:03, 3 June 2024 (UTC)
- I agree with that approach. The GM animals page is the right location for such content. I'm fine with including algae here, as, in effect, a non-animal crop. --Tryptofish (talk) 21:14, 3 June 2024 (UTC)
- Thanks. That's a good question, about animals. I would take "crops" to mean plants only, but I can see an argument to the contrary, so I'd like to hear more opinions. --Tryptofish (talk) 20:46, 3 June 2024 (UTC)
- B-Class level-5 vital articles
- Wikipedia level-5 vital articles in Technology
- B-Class vital articles in Technology
- B-Class Molecular Biology articles
- Unknown-importance Molecular Biology articles
- B-Class Genetics articles
- High-importance Genetics articles
- WikiProject Genetics articles
- All WikiProject Molecular Biology pages
- B-Class Food and drink articles
- High-importance Food and drink articles
- WikiProject Food and drink articles
- B-Class Agriculture articles
- High-importance Agriculture articles
- WikiProject Agriculture articles
- B-Class Horticulture and gardening articles
- Low-importance Horticulture and gardening articles
- WikiProject Horticulture and Gardening articles
- B-Class plant articles
- Mid-importance plant articles
- WikiProject Plants articles






