Canavanine
| |
| Names | |
|---|---|
| Preferred IUPAC name
Canavanine | |
| Systematic IUPAC name
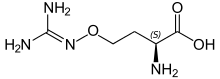
(2S)-2-amino-4-{[(diaminomethylidene)amino]oxy}butanoic acid | |
| Identifiers | |
3D model (JSmol)
|
|
| ChEBI | |
| ChemSpider | |
| DrugBank | |
| ECHA InfoCard | 100.153.281 |
| EC Number |
|
| KEGG | |
| MeSH | Canavanine |
PubChem CID
|
|
| UNII | |
CompTox Dashboard (EPA)
|
|
| |
| |
| Properties | |
| C5H12N4O3 | |
| Molar mass | 176.176 g·mol−1 |
| Density | 1.61 g·cm−3 (predicted) |
| Melting point | 184 °C (363 °F; 457 K) |
| Boiling point | 366 °C (691 °F; 639 K) |
| soluble | |
| Solubility | insoluble in alcohol, ether, benzene |
| log P | -0.91 (predicted) |
| Vapor pressure | 1.61 μPa (predicted) |
| Acidity (pKa) | 2.35 (carboxylic acid), 7.01 (oxoguanidinium), 9.22 (ammonium) |
| Hazards | |
| GHS labelling: | |

| |
| Warning | |
| H302, H312, H332 | |
| Flash point | 214.6 °C (418.3 °F; 487.8 K) (predicted) |
Except where otherwise noted, data are given for materials in their standard state (at 25 °C [77 °F], 100 kPa).
| |
L-(+)-(S)-Canavanine is a non-proteinogenic amino acid found in certain leguminous plants. It is structurally related to the proteinogenic α-amino acid L-arginine, the sole difference being the replacement of a methylene bridge (-CH
2- unit) in arginine with an oxa group (i.e., an oxygen atom) in canavanine. Canavanine is accumulated primarily in the seeds of the organisms which produce it, where it serves both as a highly deleterious defensive compound against herbivores (due to cells mistaking it for arginine) and a vital source of nitrogen for the growing embryo.[citation needed] The related L-canaline is similar to ornithine.
Toxicity[edit]
The mechanism of canavanine's toxicity is that organisms that consume it typically mistakenly incorporate it into their own proteins in place of L-arginine, thereby producing structurally aberrant proteins that may not function properly. Cleavage by arginase also produces canaline, a potent insecticide.
The toxicity of canavanine may be enhanced under conditions of protein starvation,[1] and canavanine toxicity, resulting from consumption of Hedysarum alpinum seeds with a concentration of 1.2% canavanine weight/weight, has been implicated in the death of a malnourished Christopher McCandless.[2] (McCandless was the subject of Jon Krakauer's book (and subsequent movie) Into the Wild).

In mammals[edit]
NZB/W F1, NZB, and DBA/2 mice fed L-canavanine develop a syndrome similar to systemic lupus erythematosus,[1] while BALB/c mice fed a steady diet of protein containing 1% canavanine showed no change in lifespan.[3]
Alfalfa seeds and sprouts contain L-canavanine. The L-canavanine in alfalfa has been linked to lupus-like symptoms in primates, including humans, and other auto-immune diseases. Often stopping consumption reverses the problem.[4][5][6]
Tolerance[edit]
Some specialized herbivores tolerate L-canavanine either because they metabolize it efficiently (cf. L-canaline) or avoid its incorporation into their own nascent proteins.
By metabolic detoxification[edit]
Herbivores may be able to metabolize canavanine efficiently. The beetle Caryedes brasiliensis is able to break canavanine down to canaline, then further detoxifies canaline by reductive deamination to form homoserine and ammonia. As a result, the beetle not only tolerates the chemical, but uses it as a source of nitrogen to synthesize its other amino acids to allow it to develop.[7]
By selectivity[edit]
An example of this ability can be found in the larvae of the tobacco budworm Heliothis virescens, which can tolerate large (lethal concentration 50 or LC50 300 mM) amounts of dietary canavanine.[8] These larvae fastidiously avoid incorporation of L-canavanine into their nascent proteins due to gastrointestinal expression of canavanine hydrolase, an enzyme that cleaves L-canavanine into L-homoserine and hydroxyguanidine, and L-arginine kinase, which phosphorylates L-canavanine.[9] In contrast, larvae of the tobacco hornworm Manduca sexta can only tolerate tiny amounts (1.0 microgram per kilogram of fresh body weight) of dietary canavanine because their arginine-tRNA ligase has little, if any, discriminatory capacity. No one has examined experimentally the arginine-tRNA synthetase of these organisms. But comparative studies of the incorporation of radiolabeled L-arginine and L-canavanine have shown that in Manduca sexta, the ratio of incorporation is about 3 to 1.[10]
Dioclea megacarpa seeds contain high levels of canavanine. The beetle Caryedes brasiliensis is able to tolerate this however as it has the most highly discriminatory arginine-tRNA ligase known (as of 1982). In this insect, the level of radiolabeled L-canavanine incorporated into newly synthesized proteins is barely measurable. Moreover, this beetle uses canavanine as a nitrogen source (see above).[11]
See also[edit]
References[edit]
- ^ a b Akaogi, Jun; Barker, Tolga; Kuroda, Yoshiki; Nacionales, Dina C.; Yamasaki, Yoshioki; Stevens, Bruce R.; Reeves, Westley H.; Satoh, Minoru (2006). "Role of non-protein amino acid l-canavanine in autoimmunity". Autoimmunity Reviews. 5 (6): 429–35. doi:10.1016/j.autrev.2005.12.004. PMID 16890899.
- ^ Krakauer, J., et al. (2015). "Presence of l-canavanine in Hedysarum alpinum seeds and its potential role in the death of Christopher McCandless." Wilderness & Environmental Medicine. doi:10.1016/j.wem.2014.08.014
- ^ Brown, Dan L (2005). "Canavanine-induced longevity in mice may require diets with greater than 15.7% protein". Nutrition & Metabolism. 2 (1): 7. doi:10.1186/1743-7075-2-7. PMC 554090. PMID 15733319.
- ^ Montanaro, A; Bardana Jr, E. J. (1991). "Dietary amino acid-induced systemic lupus erythematosus". Rheumatic Disease Clinics of North America. 17 (2): 323–32. doi:10.1016/S0889-857X(21)00573-1. PMID 1862241.
- ^ Herbert, V; Kasdan, T. S. (1994). "Alfalfa, vitamin E, and autoimmune disorders". The American Journal of Clinical Nutrition. 60 (4): 639–40. doi:10.1093/ajcn/60.4.639. PMID 8092103.[unreliable medical source?]
- ^ http://vegpeace.org/rawfoodtoxins.html[full citation needed][permanent dead link][unreliable medical source?]
- ^ Rosenthal, Gerald A.; Dahlman, D. L.; Janzen, Daniel H. (3 November 1978). "L-Canaline Detoxification: A Seed Predator's Biochemical Mechanism". Science. 202 (4367): 528–529. doi:10.1126/science.202.4367.528.
- ^ Berge MA, Rosenthal GA, Dahlman DL (1986). "Tobacco budworm, Heliothis virescens [Noctuidae] resistance to l-canavanine, a protective allelochemical". Pesticide Biochemistry and Physiology. 25 (3): 319–326. doi:10.1016/0048-3575(86)90005-2.
- ^ Melangeli C, Rosenthal GA, Dalman DL (1997). "The biochemical basis for l-canavanine tolerance by the tobacco budworm Heliothis virescens (Noctuidae)". Proceedings of the National Academy of Sciences of the United States of America. 94 (6): 2255–2260. doi:10.1073/pnas.94.6.2255. PMC 20074. PMID 9122181.
- ^ Rosenthal, G. A.; Dahlman, D. L. (1986). "L-Canavanine and protein synthesis in the tobacco hornworm Manduca sexta". Proceedings of the National Academy of Sciences. 83 (1): 14–8. Bibcode:1986PNAS...83...14R. doi:10.1073/pnas.83.1.14. JSTOR 26787. PMC 322781. PMID 3455753.
- ^ Rosenthal, G. A.; Hughes, C. G.; Janzen, D. H. (1982). "L-Canavanine, a Dietary Nitrogen Source for the Seed Predator Caryedes brasiliensis (Bruchidae)". Science. 217 (4557): 353–5. Bibcode:1982Sci...217..353R. doi:10.1126/science.217.4557.353. PMID 17791516. S2CID 26741233.
This article needs additional citations for verification. (January 2011) |
Bibliography[edit]
- Rosenthal, Gerald A. (1986). "Biochemical insight into insecticidal properties ofl-Canavanine, a higher plant protective allelochemical". Journal of Chemical Ecology. 12 (5): 1145–56. doi:10.1007/BF01639001. PMID 24307052. S2CID 37297952.
- Rosenthal, G. A.; Berge, M. A.; Bleiler, J. A.; Rudd, T. P. (1987). "Aberrant, canavanyl protein formation and the ability to tolerate or utilize L-canavanine". Experientia. 43 (5): 558–61. doi:10.1007/BF02143585. PMID 3582574. S2CID 24239284.
- Boyar, A.; Marsh, R. E. (1982). "L-Canavanine, a paradigm for the structures of substituted guanidines". Journal of the American Chemical Society. 104 (7): 1995–1998. doi:10.1021/ja00371a033.
- Turner BL, Harborne JB (1967). "Distribution of canavanine in the plant kingdom". Phytochemistry. 6 (6): 863–866. Bibcode:1967PChem...6..863T. doi:10.1016/S0031-9422(00)86033-1. and in particularly large amounts in Canavalia gladiata (sword bean).
- Ekanayake S, Skog K, Asp NG (May 2007). "Canavanine content in sword beans (Canavalia gladiata): analysis and effect of processing". Food and Chemical Toxicology. 45 (5): 797–803. doi:10.1016/j.fct.2006.10.030. PMID 17187914.
.........
