Color vision

Color vision, a feature of visual perception, is an ability to perceive differences between light composed of different frequencies independently of light intensity.
Color perception is a part of the larger visual system and is mediated by a complex process between neurons that begins with differential stimulation of different types of photoreceptors by light entering the eye. Those photoreceptors then emit outputs that are propagated through many layers of neurons and then ultimately to the brain. Color vision is found in many animals and is mediated by similar underlying mechanisms with common types of biological molecules and a complex history of evolution in different animal taxa. In primates, color vision may have evolved under selective pressure for a variety of visual tasks including the foraging for nutritious young leaves, ripe fruit, and flowers, as well as detecting predator camouflage and emotional states in other primates.[1][2][3]
Wavelength[edit]
 | |||
|---|---|---|---|
| Color | Wavelength (nm) |
Frequency (THz) |
Photon energy (eV) |
| 380–450 | 670–790 | 2.75–3.26 | |
| 450–485 | 620–670 | 2.56–2.75 | |
| 485–500 | 600–620 | 2.48–2.56 | |
| 500–565 | 530–600 | 2.19–2.48 | |
| 565–590 | 510–530 | 2.10–2.19 | |
| 590–625 | 480–510 | 1.98–2.10 | |
| 625–750 | 400–480 | 1.65–1.98 | |
Isaac Newton discovered that white light after being split into its component colors when passed through a dispersive prism could be recombined to make white light by passing them through a different prism.
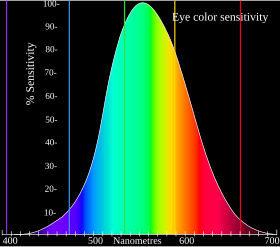
The visible light spectrum ranges from about 380 to 740 nanometers. Spectral colors (colors that are produced by a narrow band of wavelengths) such as red, orange, yellow, green, cyan, blue, and violet can be found in this range. These spectral colors do not refer to a single wavelength, but rather to a set of wavelengths: red, 625–740 nm; orange, 590–625 nm; yellow, 565–590 nm; green, 500–565 nm; cyan, 485–500 nm; blue, 450–485 nm; violet, 380–450 nm.
Wavelengths longer or shorter than this range are called infrared or ultraviolet, respectively. Humans cannot generally see these wavelengths, but other animals may.
Hue detection[edit]
Sufficient differences in wavelength cause a difference in the perceived hue; the just-noticeable difference in wavelength varies from about 1 nm in the blue-green and yellow wavelengths to 10 nm and more in the longer red and shorter blue wavelengths. Although the human eye can distinguish up to a few hundred hues, when those pure spectral colors are mixed together or diluted with white light, the number of distinguishable chromaticities can be much higher.
In very low light levels, vision is scotopic: light is detected by rod cells of the retina. Rods are maximally sensitive to wavelengths near 500 nm and play little, if any, role in color vision. In brighter light, such as daylight, vision is photopic: light is detected by cone cells which are responsible for color vision. Cones are sensitive to a range of wavelengths, but are most sensitive to wavelengths near 555 nm. Between these regions, mesopic vision comes into play and both rods and cones provide signals to the retinal ganglion cells. The shift in color perception from dim light to daylight gives rise to differences known as the Purkinje effect.
The perception of "white" is formed by the entire spectrum of visible light, or by mixing colors of just a few wavelengths in animals with few types of color receptors. In humans, white light can be perceived by combining wavelengths such as red, green, and blue, or just a pair of complementary colors such as blue and yellow.[4]
Non-spectral colors[edit]
There are a variety of colors in addition to spectral colors and their hues. These include grayscale colors, shades of colors obtained by mixing grayscale colors with spectral colors, violet-red colors, impossible colors, and metallic colors.
Grayscale colors include white, gray, and black. Rods contain rhodopsin, which reacts to light intensity, providing grayscale coloring.
Shades include colors such as pink or brown. Pink is obtained from mixing red and white. Brown may be obtained from mixing orange with gray or black. Navy is obtained from mixing blue and black.
Violet-red colors include hues and shades of magenta. The light spectrum is a line on which violet is one end and the other is red, and yet we see hues of purple that connect those two colors.
Impossible colors are a combination of cone responses that cannot be naturally produced. For example, medium cones cannot be activated completely on their own; if they were, we would see a 'hyper-green' color.
Dimensionality[edit]
Color vision is categorized foremost according to the dimensionality of the color gamut, which is defined by the number of primaries required to represent the color vision. This is generally equal to the number of photopsins expressed: a correlation that holds for vertebrates but not invertebrates. The common vertebrate ancestor possessed four photopsins (expressed in cones) plus rhodopsin (expressed in rods), so was tetrachromatic. However, many vertebrate lineages have lost one or many photopsin genes, leading to lower-dimension color vision. The dimensions of color vision range from 1-dimensional and up:
| Dimension | Characteristic | Occurrence |
|---|---|---|
| Monochromacy | 1D color vision lack of any color perception |
Some mammals, including Pinnipeds, Cetaceans, and Xenarthra[5][6] |
| Dichromacy | 2D color vision | most mammals and a quarter of color blind humans |
| Trichromacy | 3D color vision | most humans |
| Tetrachromacy | 4D color vision | most birds, reptiles and fish |
| Pentachromacy and higher | 5D+ color vision | rare in vertebrates |
Physiology of color perception[edit]


Perception of color begins with specialized retinal cells known as cone cells. Cone cells contain different forms of opsin – a pigment protein – that have different spectral sensitivities. Humans contain three types, resulting in trichromatic color vision.
Each individual cone contains pigments composed of opsin apoprotein covalently linked to a light-absorbing prosthetic group: either 11-cis-hydroretinal or, more rarely, 11-cis-dehydroretinal.[7]
The cones are conventionally labeled according to the ordering of the wavelengths of the peaks of their spectral sensitivities: short (S), medium (M), and long (L) cone types. These three types do not correspond well to particular colors as we know them. Rather, the perception of color is achieved by a complex process that starts with the differential output of these cells in the retina and which is finalized in the visual cortex and associative areas of the brain.
For example, while the L cones have been referred to simply as red receptors, microspectrophotometry has shown that their peak sensitivity is in the greenish-yellow region of the spectrum. Similarly, the S cones and M cones do not directly correspond to blue and green, although they are often described as such. The RGB color model, therefore, is a convenient means for representing color but is not directly based on the types of cones in the human eye.
The peak response of human cone cells varies, even among individuals with so-called normal color vision;[8] in some non-human species this polymorphic variation is even greater, and it may well be adaptive.[jargon][9]
Theories[edit]

Two complementary theories of color vision are the trichromatic theory and the opponent process theory. The trichromatic theory, or Young–Helmholtz theory, proposed in the 19th century by Thomas Young and Hermann von Helmholtz, posits three types of cones preferentially sensitive to blue, green, and red, respectively. Others have suggested that the trichromatic theory is not specifically a theory of color vision but a theory of receptors for all vision, including color but not specific or limited to it.[10] Equally, it has been suggested that the relationship between the phenomenal opponency described by Hering and the physiological opponent processes are not straightforward (see below), making of physiological opponency a mechanism that is relevant to the whole of vision, and not just to color vision alone.[10] Ewald Hering proposed the opponent process theory in 1872.[11] It states that the visual system interprets color in an antagonistic way: red vs. green, blue vs. yellow, black vs. white. Both theories are generally accepted as valid, describing different stages in visual physiology, visualized in the adjacent diagram.[12]: 168
Green–magenta and blue—yellow are scales with mutually exclusive boundaries. In the same way that there cannot exist a "slightly negative" positive number, a single eye cannot perceive a bluish-yellow or a reddish-green. Although these two theories are both currently widely accepted theories, past and more recent work has led to criticism of the opponent process theory, stemming from a number of what are presented as discrepancies in the standard opponent process theory. For example, the phenomenon of an after-image of complementary color can be induced by fatiguing the cells responsible for color perception, by staring at a vibrant color for a length of time, and then looking at a white surface. This phenomenon of complementary colors demonstrates cyan, rather than green, to be the complement of red and magenta, rather than red, to be the complement of green, as well as demonstrating, as a consequence, that the reddish-green color proposed to be impossible by opponent process theory is, in fact, the color yellow. Although this phenomenon is more readily explained by the trichromatic theory, explanations for the discrepancy may include alterations to the opponent process theory, such as redefining the opponent colors as red vs. cyan, to reflect this effect. Despite such criticisms, both theories remain in use.
A recent demonstration, using the Color Mondrian, has shown that, just as the color of a surface that is part of a complex 'natural' scene is independent of the wavelength-energy composition of the light reflected from it alone but depends upon the composition of the light reflected from its surrounds as well, so the after image produced by looking at a given part of a complex scene is also independent of the wavelength energy-composition of the light reflected from it alone. Thus, while the color of the after-image produced by looking at a green surface that is reflecting more "green" (middle-wave) than "red" (long-wave) light is magenta, so is the after image of the same surface when it reflects more "red" than "green" light (when it is still perceived as green). This would seem to rule out an explanation of color opponency based on retinal cone adaptation.[13]
Cone cells in the human eye[edit]

A range of wavelengths of light stimulates each of these receptor types to varying degrees. The brain combines the information from each type of receptor to give rise to different perceptions of different wavelengths of light.
| Cone type | Name | Range | Peak wavelength[14][15] |
|---|---|---|---|
| S | β | 400–500 nm | 420–440 nm |
| M | γ | 450–630 nm | 534–555 nm |
| L | ρ | 500–700 nm | 564–580 nm |
Cones and rods are not evenly distributed in the human eye. Cones have a high density at the fovea and a low density in the rest of the retina.[16] Thus color information is mostly taken in at the fovea. Humans have poor color perception in their peripheral vision, and much of the color we see in our periphery may be filled in by what our brains expect to be there on the basis of context and memories. However, our accuracy of color perception in the periphery increases with the size of stimulus.[17]
The opsins (photopigments) present in the L and M cones are encoded on the X chromosome; defective encoding of these leads to the two most common forms of color blindness. The OPN1LW gene, which encodes the opsin present in the L cones, is highly polymorphic; one study found 85 variants in a sample of 236 men.[18] A small percentage of women may have an extra type of color receptor because they have different alleles for the gene for the L opsin on each X chromosome. X chromosome inactivation means that while only one opsin is expressed in each cone cell, both types may occur overall, and some women may therefore show a degree of tetrachromatic color vision.[19] Variations in OPN1MW, which encodes the opsin expressed in M cones, appear to be rare, and the observed variants have no effect on spectral sensitivity.
Color in the primate brain[edit]

Color processing begins at a very early level in the visual system (even within the retina) through initial color opponent mechanisms. Both Helmholtz's trichromatic theory and Hering's opponent-process theory are therefore correct, but trichromacy arises at the level of the receptors, and opponent processes arise at the level of retinal ganglion cells and beyond. In Hering's theory, opponent mechanisms refer to the opposing color effect of red–green, blue–yellow, and light-dark. However, in the visual system, it is the activity of the different receptor types that are opposed. Some midget retinal ganglion cells oppose L and M cone activity, which corresponds loosely to red–green opponency, but actually runs along an axis from blue-green to magenta. Small bistratified retinal ganglion cells oppose input from the S cones to input from the L and M cones. This is often thought to correspond to blue–yellow opponency but actually runs along a color axis from yellow-green to violet.
Visual information is then sent to the brain from retinal ganglion cells via the optic nerve to the optic chiasma: a point where the two optic nerves meet and information from the temporal (contralateral) visual field crosses to the other side of the brain. After the optic chiasma, the visual tracts are referred to as the optic tracts, which enter the thalamus to synapse at the lateral geniculate nucleus (LGN).
The lateral geniculate nucleus is divided into laminae (zones), of which there are three types: the M-laminae, consisting primarily of M-cells, the P-laminae, consisting primarily of P-cells, and the koniocellular laminae. M- and P-cells receive relatively balanced input from both L- and M-cones throughout most of the retina, although this seems to not be the case at the fovea, with midget cells synapsing in the P-laminae. The koniocellular laminae receives axons from the small bistratified ganglion cells.[20][21]
After synapsing at the LGN, the visual tract continues on back to the primary visual cortex (V1) located at the back of the brain within the occipital lobe. Within V1 there is a distinct band (striation). This is also referred to as "striate cortex", with other cortical visual regions referred to collectively as "extrastriate cortex". It is at this stage that color processing becomes much more complicated.
In V1 the simple three-color segregation begins to break down. Many cells in V1 respond to some parts of the spectrum better than others, but this "color tuning" is often different depending on the adaptation state of the visual system. A given cell that might respond best to long-wavelength light if the light is relatively bright might then become responsive to all wavelengths if the stimulus is relatively dim. Because the color tuning of these cells is not stable, some believe that a different, relatively small, population of neurons in V1 is responsible for color vision. These specialized "color cells" often have receptive fields that can compute local cone ratios. Such "double-opponent" cells were initially described in the goldfish retina by Nigel Daw;[22][23] their existence in primates was suggested by David H. Hubel and Torsten Wiesel, first demonstrated by C.R. Michael[24] and subsequently confirmed by Bevil Conway.[25] As Margaret Livingstone and David Hubel showed, double opponent cells are clustered within localized regions of V1 called blobs, and are thought to come in two flavors, red–green and blue-yellow.[26] Red–green cells compare the relative amounts of red–green in one part of a scene with the amount of red–green in an adjacent part of the scene, responding best to local color contrast (red next to green). Modeling studies have shown that double-opponent cells are ideal candidates for the neural machinery of color constancy explained by Edwin H. Land in his retinex theory.[27]

From the V1 blobs, color information is sent to cells in the second visual area, V2. The cells in V2 that are most strongly color tuned are clustered in the "thin stripes" that, like the blobs in V1, stain for the enzyme cytochrome oxidase (separating the thin stripes are interstripes and thick stripes, which seem to be concerned with other visual information like motion and high-resolution form). Neurons in V2 then synapse onto cells in the extended V4. This area includes not only V4, but two other areas in the posterior inferior temporal cortex, anterior to area V3, the dorsal posterior inferior temporal cortex, and posterior TEO.[29][30] Area V4 was initially suggested by Semir Zeki to be exclusively dedicated to color,[31] and he later showed that V4 can be subdivided into subregions with very high concentrations of color cells separated from each other by zones with lower concentration of such cells though even the latter cells respond better to some wavelengths than to others,[32] a finding confirmed by subsequent studies.[29][33][34] The presence in V4 of orientation-selective cells led to the view that V4 is involved in processing both color and form associated with color[35] but it is worth noting that the orientation selective cells within V4 are more broadly tuned than their counterparts in V1, V2 and V3.[32] Color processing in the extended V4 occurs in millimeter-sized color modules called globs.[29][30] This is the part of the brain in which color is first processed into the full range of hues found in color space.[36][29][30]
Anatomical studies have shown that neurons in extended V4 provide input to the inferior temporal lobe. "IT" cortex is thought to integrate color information with shape and form, although it has been difficult to define the appropriate criteria for this claim. Despite this murkiness, it has been useful to characterize this pathway (V1 > V2 > V4 > IT) as the ventral stream or the "what pathway", distinguished from the dorsal stream ("where pathway") that is thought to analyze motion, among other features.
Subjectivity of color perception [edit]
Color is a feature of visual perception by an observer. There is a complex relationship between the wavelengths of light in the visual spectrum and human experiences of color. Although most people are assumed to have the same mapping, the philosopher John Locke recognized that alternatives are possible, and described one such hypothetical case with the "inverted spectrum" thought experiment. For example, someone with an inverted spectrum might experience green while seeing 'red' (700 nm) light, and experience red while seeing 'green' (530 nm) light. This inversion has never been demonstrated in experiment, though.
Synesthesia (or ideasthesia) provides some atypical but illuminating examples of subjective color experience triggered by input that is not even light, such as sounds or shapes. The possibility of a clean dissociation between color experience from properties of the world reveals that color is a subjective psychological phenomenon.
The Himba people have been found to categorize colors differently from most Westerners and are able to easily distinguish close shades of green, barely discernible for most people.[37] The Himba have created a very different color scheme which divides the spectrum to dark shades (zuzu in Himba), very light (vapa), vivid blue and green (buru) and dry colors as an adaptation to their specific way of life.
The perception of color depends heavily on the context in which the perceived object is presented.[38]
Psychophysical experiments have shown that color is perceived before the orientation of lines and directional motion by as much as 40ms and 80 ms respectively, thus leading to a perceptual asynchrony that is demonstrable with brief presentation times.
Chromatic adaptation[edit]
In color vision, chromatic adaptation refers to color constancy; the ability of the visual system to preserve the appearance of an object under a wide range of light sources.[39] For example, a white page under blue, pink, or purple light will reflect mostly blue, pink, or purple light to the eye, respectively; the brain, however, compensates for the effect of lighting (based on the color shift of surrounding objects) and is more likely to interpret the page as white under all three conditions, a phenomenon known as color constancy.
In color science, chromatic adaptation is the estimation of the representation of an object under a different light source from the one in which it was recorded. A common application is to find a chromatic adaptation transform (CAT) that will make the recording of a neutral object appear neutral (color balance), while keeping other colors also looking realistic.[40] For example, chromatic adaptation transforms are used when converting images between ICC profiles with different white points. Adobe Photoshop, for example, uses the Bradford CAT.[41]
Color vision in nonhumans[edit]
Many species can see light with frequencies outside the human "visible spectrum". Bees and many other insects can detect ultraviolet light, which helps them to find nectar in flowers. Plant species that depend on insect pollination may owe reproductive success to ultraviolet "colors" and patterns rather than how colorful they appear to humans. Birds, too, can see into the ultraviolet (300–400 nm), and some have sex-dependent markings on their plumage that are visible only in the ultraviolet range.[42][43] Many animals that can see into the ultraviolet range, however, cannot see red light or any other reddish wavelengths. For example, bees' visible spectrum ends at about 590 nm, just before the orange wavelengths start. Birds, however, can see some red wavelengths, although not as far into the light spectrum as humans.[44] It is a myth that the common goldfish is the only animal that can see both infrared and ultraviolet light;[45] their color vision extends into the ultraviolet but not the infrared.[46]
The basis for this variation is the number of cone types that differ between species. Mammals, in general, have a color vision of a limited type, and usually have red–green color blindness, with only two types of cones. Humans, some primates, and some marsupials see an extended range of colors, but only by comparison with other mammals. Most non-mammalian vertebrate species distinguish different colors at least as well as humans, and many species of birds, fish, reptiles, and amphibians, and some invertebrates, have more than three cone types and probably superior color vision to humans.
In most Catarrhini (Old World monkeys and apes—primates closely related to humans), there are three types of color receptors (known as cone cells), resulting in trichromatic color vision. These primates, like humans, are known as trichromats. Many other primates (including New World monkeys) and other mammals are dichromats, which is the general color vision state for mammals that are active during the day (i.e., felines, canines, ungulates). Nocturnal mammals may have little or no color vision. Trichromat non-primate mammals are rare.[12]: 174–175 [47]
Many invertebrates have color vision. Honeybees and bumblebees have trichromatic color vision which is insensitive to red but sensitive to ultraviolet. Osmia rufa, for example, possess a trichromatic color system, which they use in foraging for pollen from flowers.[48] In view of the importance of color vision to bees one might expect these receptor sensitivities to reflect their specific visual ecology; for example the types of flowers that they visit. However, the main groups of hymenopteran insects excluding ants (i.e., bees, wasps and sawflies) mostly have three types of photoreceptor, with spectral sensitivities similar to the honeybee's.[49] Papilio butterflies possess six types of photoreceptors and may have pentachromatic vision.[50] The most complex color vision system in the animal kingdom has been found in stomatopods (such as the mantis shrimp) having between 12 and 16 spectral receptor types thought to work as multiple dichromatic units.[51]
Vertebrate animals such as tropical fish and birds sometimes have more complex color vision systems than humans; thus the many subtle colors they exhibit generally serve as direct signals for other fish or birds, and not to signal mammals.[52] In bird vision, tetrachromacy is achieved through up to four cone types, depending on species. Each single cone contains one of the four main types of vertebrate cone photopigment (LWS/ MWS, RH2, SWS2 and SWS1) and has a colored oil droplet in its inner segment.[49] Brightly colored oil droplets inside the cones shift or narrow the spectral sensitivity of the cell. Pigeons may be pentachromats.[53]
Reptiles and amphibians also have four cone types (occasionally five), and probably see at least the same number of colors that humans do, or perhaps more. In addition, some nocturnal geckos and frogs have the capability of seeing color in dim light.[54][55] At least some color-guided behaviors in amphibians have also been shown to be wholly innate, developing even in visually deprived animals.[56]
In the evolution of mammals, segments of color vision were lost, then for a few species of primates, regained by gene duplication. Eutherian mammals other than primates (for example, dogs, mammalian farm animals) generally have less-effective two-receptor (dichromatic) color perception systems, which distinguish blue, green, and yellow—but cannot distinguish oranges and reds. There is some evidence that a few mammals, such as cats, have redeveloped the ability to distinguish longer wavelength colors, in at least a limited way, via one-amino-acid mutations in opsin genes.[57] The adaptation to see reds is particularly important for primate mammals, since it leads to the identification of fruits, and also newly sprouting reddish leaves, which are particularly nutritious.
However, even among primates, full color vision differs between New World and Old World monkeys. Old World primates, including monkeys and all apes, have vision similar to humans. New World monkeys may or may not have color sensitivity at this level: in most species, males are dichromats, and about 60% of females are trichromats, but the owl monkeys are cone monochromats, and both sexes of howler monkeys are trichromats.[58][59][60][61] Visual sensitivity differences between males and females in a single species is due to the gene for yellow-green sensitive opsin protein (which confers ability to differentiate red from green) residing on the X sex chromosome.
Several marsupials, such as the fat-tailed dunnart (Sminthopsis crassicaudata), have trichromatic color vision.[62]
Marine mammals, adapted for low-light vision, have only a single cone type and are thus monochromats.[citation needed]
Evolution[edit]
Color perception mechanisms are highly dependent on evolutionary factors, of which the most prominent is thought to be satisfactory recognition of food sources. In herbivorous primates, color perception is essential for finding proper (immature) leaves. In hummingbirds, particular flower types are often recognized by color as well. On the other hand, nocturnal mammals have less-developed color vision since adequate light is needed for cones to function properly. There is evidence that ultraviolet light plays a part in color perception in many branches of the animal kingdom, especially insects. In general, the optical spectrum encompasses the most common electronic transitions in the matter and is therefore the most useful for collecting information about the environment.
The evolution of trichromatic color vision in primates occurred as the ancestors of modern monkeys, apes, and humans switched to diurnal (daytime) activity and began consuming fruits and leaves from flowering plants.[63] Color vision, with UV discrimination, is also present in a number of arthropods—the only terrestrial animals besides the vertebrates to possess this trait.[64]
Some animals can distinguish colors in the ultraviolet spectrum. The UV spectrum falls outside the human visible range, except for some cataract surgery patients.[65] Birds, turtles, lizards, many fish and some rodents have UV receptors in their retinas.[66] These animals can see the UV patterns found on flowers and other wildlife that are otherwise invisible to the human eye.
Ultraviolet vision is an especially important adaptation in birds. It allows birds to spot small prey from a distance, navigate, avoid predators, and forage while flying at high speeds. Birds also utilize their broad spectrum vision to recognize other birds, and in sexual selection.[67][68]
Mathematics of color perception[edit]
A "physical color" is a combination of pure spectral colors (in the visible range). In principle there exist infinitely many distinct spectral colors, and so the set of all physical colors may be thought of as an infinite-dimensional vector space (a Hilbert space). This space is typically notated Hcolor. More technically, the space of physical colors may be considered to be the topological cone over the simplex whose vertices are the spectral colors, with white at the centroid of the simplex, black at the apex of the cone, and the monochromatic color associated with any given vertex somewhere along the line from that vertex to the apex depending on its brightness.
An element C of Hcolor is a function from the range of visible wavelengths—considered as an interval of real numbers [Wmin,Wmax]—to the real numbers, assigning to each wavelength w in [Wmin,Wmax] its intensity C(w).
A humanly perceived color may be modeled as three numbers: the extents to which each of the 3 types of cones is stimulated. Thus a humanly perceived color may be thought of as a point in 3-dimensional Euclidean space. We call this space R3color.
Since each wavelength w stimulates each of the 3 types of cone cells to a known extent, these extents may be represented by 3 functions s(w), m(w), l(w) corresponding to the response of the S, M, and L cone cells, respectively.
Finally, since a beam of light can be composed of many different wavelengths, to determine the extent to which a physical color C in Hcolor stimulates each cone cell, we must calculate the integral (with respect to w), over the interval [Wmin,Wmax], of C(w)·s(w), of C(w)·m(w), and of C(w)·l(w). The triple of resulting numbers associates with each physical color C (which is an element in Hcolor) a particular perceived color (which is a single point in R3color). This association is easily seen to be linear. It may also easily be seen that many different elements in the "physical" space Hcolor can all result in the same single perceived color in R3color, so a perceived color is not unique to one physical color.
Thus human color perception is determined by a specific, non-unique linear mapping from the infinite-dimensional Hilbert space Hcolor to the 3-dimensional Euclidean space R3color.
Technically, the image of the (mathematical) cone over the simplex whose vertices are the spectral colors, by this linear mapping, is also a (mathematical) cone in R3color. Moving directly away from the vertex of this cone represents maintaining the same chromaticity while increasing its intensity. Taking a cross-section of this cone yields a 2D chromaticity space. Both the 3D cone and its projection or cross-section are convex sets; that is, any mixture of spectral colors is also a color.
In practice, it would be quite difficult to physiologically measure an individual's three cone responses to various physical color stimuli. Instead, a psychophysical approach is taken.[69] Three specific benchmark test lights are typically used; let us call them S, M, and L. To calibrate human perceptual space, scientists allowed human subjects to try to match any physical color by turning dials to create specific combinations of intensities (IS, IM, IL) for the S, M, and L lights, resp., until a match was found. This needed only to be done for physical colors that are spectral, since a linear combination of spectral colors will be matched by the same linear combination of their (IS, IM, IL) matches. Note that in practice, often at least one of S, M, L would have to be added with some intensity to the physical test color, and that combination matched by a linear combination of the remaining 2 lights. Across different individuals (without color blindness), the matchings turned out to be nearly identical.
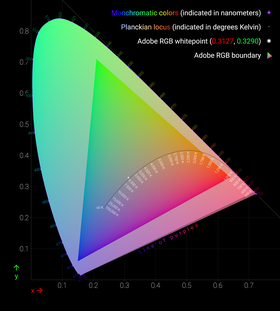
By considering all the resulting combinations of intensities (IS, IM, IL) as a subset of 3-space, a model for human perceptual color space is formed. (Note that when one of S, M, L had to be added to the test color, its intensity was counted as negative.) Again, this turns out to be a (mathematical) cone, not a quadric, but rather all rays through the origin in 3-space passing through a certain convex set. Again, this cone has the property that moving directly away from the origin corresponds to increasing the intensity of the S, M, L lights proportionately. Again, a cross-section of this cone is a planar shape that is (by definition) the space of "chromaticities" (informally: distinct colors); one particular such cross-section, corresponding to constant X+Y+Z of the CIE 1931 color space, gives the CIE chromaticity diagram.
This system implies that for any hue or non-spectral color not on the boundary of the chromaticity diagram, there are infinitely many distinct physical spectra that are all perceived as that hue or color. So, in general, there is no such thing as the combination of spectral colors that we perceive as (say) a specific version of tan; instead, there are infinitely many possibilities that produce that exact color. The boundary colors that are pure spectral colors can be perceived only in response to light that is purely at the associated wavelength, while the boundary colors on the "line of purples" can each only be generated by a specific ratio of the pure violet and the pure red at the ends of the visible spectral colors.
The CIE chromaticity diagram is horseshoe-shaped, with its curved edge corresponding to all spectral colors (the spectral locus), and the remaining straight edge corresponding to the most saturated purples, mixtures of red and violet.
See also[edit]
References[edit]
- ^ Vorobyev M (July 2004). "Ecology and evolution of primate colour vision". Clinical & Experimental Optometry. 87 (4–5): 230–8. doi:10.1111/j.1444-0938.2004.tb05053.x. PMID 15312027. S2CID 40234800.
- ^ Carvalho LS, Pessoa D, Mountford JK, Davies WI, Hunt DM (26 April 2017). "The Genetic and Evolutionary Drives behind Primate Color Vision". Frontiers in Ecology and Evolution. 5. doi:10.3389/fevo.2017.00034.
- ^ Hiramatsu C, Melin AD, Allen WL, Dubuc C, Higham JP (June 2017). "Experimental evidence that primate trichromacy is well suited for detecting primate social colour signals". Proceedings. Biological Sciences. 284 (1856): 20162458. doi:10.1098/rspb.2016.2458. PMC 5474062. PMID 28615496.
- ^ Davson H, Perkins ES (7 August 2020). "Human eye". Encyclopedia Britannica.
- ^ Peichl, Leo; Behrmann, Gunther; Kroger, Ronald H. H. (April 2001). "For whales and seals the ocean is not blue: a visual pigment loss in marine mammals". European Journal of Neuroscience. 13 (8): 1520–1528. CiteSeerX 10.1.1.486.616. doi:10.1046/j.0953-816x.2001.01533.x. PMID 11328346. S2CID 16062564.
- ^ Emerling, Christopher A.; Springer, Mark S. (2015-02-07). "Genomic evidence for rod monochromacy in sloths and armadillos suggests early subterranean history for Xenarthra". Proceedings of the Royal Society B: Biological Sciences. 282 (1800): 20142192. doi:10.1098/rspb.2014.2192. ISSN 0962-8452. PMC 4298209. PMID 25540280.
- ^ Nathans J, Thomas D, Hogness DS (April 1986). "Molecular genetics of human color vision: the genes encoding blue, green, and red pigments". Science. 232 (4747): 193–202. Bibcode:1986Sci...232..193N. doi:10.1126/science.2937147. JSTOR 169687. PMID 2937147. S2CID 34321827.
- ^ Neitz J, Jacobs GH (1986). "Polymorphism of the long-wavelength cone in normal human colour vision". Nature. 323 (6089): 623–5. Bibcode:1986Natur.323..623N. doi:10.1038/323623a0. PMID 3773989. S2CID 4316301.
- ^ Jacobs GH (January 1996). "Primate photopigments and primate color vision". Proceedings of the National Academy of Sciences of the United States of America. 93 (2): 577–81. Bibcode:1996PNAS...93..577J. doi:10.1073/pnas.93.2.577. PMC 40094. PMID 8570598.
- ^ a b Zeki, Semir (2022-10-09). "The Paton prize lecture 2021: A colourful experience leading to a reassessment of colour vision and its theories". Experimental Physiology. 107 (11): 1189–1208. doi:10.1113/ep089760. ISSN 0958-0670. PMID 36114718. S2CID 252335063.
- ^ Hering E (1872). "Zur Lehre vom Lichtsinne". Sitzungsberichte der Mathematisch–Naturwissenschaftliche Classe der Kaiserlichen Akademie der Wissenschaften. LXVI. Band (III Abtheilung). K.-K. Hof- und Staatsdruckerei in Commission bei C. Gerold's Sohn.
- ^ a b Ali MA, Klyne MA (1985). Vision in Vertebrates. New York: Plenum Press. ISBN 978-0-306-42065-8.
- ^ Zeki S, Cheadle S, Pepper J, Mylonas D (2017). "The Constancy of Colored After-Images". Frontiers in Human Neuroscience. 11: 229. doi:10.3389/fnhum.2017.00229. PMC 5423953. PMID 28539878.
 Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
Text was copied from this source, which is available under a Creative Commons Attribution 4.0 International License.
- ^ Wyszecki G, Stiles WS (1982). Color Science: Concepts and Methods, Quantitative Data and Formulae (2nd ed.). New York: Wiley Series in Pure and Applied Optics. ISBN 978-0-471-02106-3.
- ^ Hunt RW (2004). The Reproduction of Colour (6th ed.). Chichester UK: Wiley–IS&T Series in Imaging Science and Technology. pp. 11–2. ISBN 978-0-470-02425-6.
- ^ Purves D, Augustine GJ, Fitzpatrick D, Katz LC, LaMantia AS, McNamara JO, Williams SM (2001). "Anatomical Distribution of Rods and Cones". Neuroscience. 2nd Edition.
- ^ Johnson MA (February 1986). "Color vision in the peripheral retina". American Journal of Optometry and Physiological Optics. 63 (2): 97–103. doi:10.1097/00006324-198602000-00003. PMID 3953765.
- ^ Verrelli BC, Tishkoff SA (September 2004). "Signatures of selection and gene conversion associated with human color vision variation". American Journal of Human Genetics. 75 (3): 363–75. doi:10.1086/423287. PMC 1182016. PMID 15252758.
- ^ Roth M (2006). "Some women may see 100 million colors, thanks to their genes". Post-Gazette.com. Archived from the original on 2006-11-08.
- ^ Rodieck RW (1998). The First Steps in Seeing. Sunderland, Massachusetts, USA: Sinauer Associates, Inc. ISBN 978-0-87893-757-8.
- ^ Hendry SH, Reid RC (1970-01-01). "The koniocellular pathway in primate vision". Annual Review of Neuroscience. 23: 127–53. doi:10.1146/annurev.neuro.23.1.127. PMID 10845061.
- ^ Daw NW (November 1967). "Goldfish retina: organization for simultaneous color contrast". Science. 158 (3803): 942–4. Bibcode:1967Sci...158..942D. doi:10.1126/science.158.3803.942. PMID 6054169. S2CID 1108881.
- ^ Conway BR (2002). Neural Mechanisms of Color Vision: Double-Opponent Cells in the Visual Cortex. Springer. ISBN 978-1-4020-7092-1.
- ^ Michael, C. R. (1978-05-01). "Color vision mechanisms in monkey striate cortex: dual-opponent cells with concentric receptive fields". Journal of Neurophysiology. 41 (3): 572–588. doi:10.1152/jn.1978.41.3.572. ISSN 0022-3077. PMID 96222.
- ^ Conway BR (April 2001). "Spatial structure of cone inputs to color cells in alert macaque primary visual cortex (V-1)". The Journal of Neuroscience. 21 (8): 2768–83. doi:10.1523/JNEUROSCI.21-08-02768.2001. PMC 6762533. PMID 11306629.
- ^ Dowling JE (2001). Neurons, and Networks: An Introduction to Behavioral Neuroscience. Harvard University Press. ISBN 978-0-674-00462-7.
- ^ McCann M, ed. (1993). Edwin H. Land's Essays. Springfield, Va.: Society for Imaging Science and Technology.
- ^ Judd DB, Wyszecki G (1975). Color in Business, Science and Industry. Wiley Series in Pure and Applied Optics (third ed.). New York: Wiley-Interscience. p. 388. ISBN 978-0-471-45212-6.
- ^ a b c d Conway BR, Moeller S, Tsao DY (November 2007). "Specialized color modules in macaque extrastriate cortex". Neuron. 56 (3): 560–73. doi:10.1016/j.neuron.2007.10.008. PMC 8162777. PMID 17988638. S2CID 11724926.
- ^ a b c Conway BR, Tsao DY (October 2009). "Color-tuned neurons are spatially clustered according to color preference within alert macaque posterior inferior temporal cortex". Proceedings of the National Academy of Sciences of the United States of America. 106 (42): 18034–9. Bibcode:2009PNAS..10618034C. doi:10.1073/pnas.0810943106. PMC 2764907. PMID 19805195.
- ^ Zeki SM (April 1973). "Colour coding in rhesus monkey prestriate cortex". Brain Research. 53 (2): 422–7. doi:10.1016/0006-8993(73)90227-8. PMID 4196224.
- ^ a b Zeki S (March 1983). "The distribution of wavelength and orientation selective cells in different areas of monkey visual cortex". Proceedings of the Royal Society of London. Series B, Biological Sciences. 217 (1209): 449–70. Bibcode:1983RSPSB.217..449Z. doi:10.1098/rspb.1983.0020. PMID 6134287. S2CID 39700958.
- ^ Bushnell BN, Harding PJ, Kosai Y, Bair W, Pasupathy A (August 2011). "Equiluminance cells in visual cortical area v4". The Journal of Neuroscience. 31 (35): 12398–412. doi:10.1523/JNEUROSCI.1890-11.2011. PMC 3171995. PMID 21880901.
- ^ Tanigawa H, Lu HD, Roe AW (December 2010). "Functional organization for color and orientation in macaque V4". Nature Neuroscience. 13 (12): 1542–8. doi:10.1038/nn.2676. PMC 3005205. PMID 21076422.
- ^ Zeki S (June 2005). "The Ferrier Lecture 1995 behind the seen: the functional specialization of the brain in space and time". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 360 (1458): 1145–83. doi:10.1098/rstb.2005.1666. PMC 1609195. PMID 16147515.
- ^ Zeki, S. (1980). "The representation of colours in the cerebral cortex". Nature. 284 (5755): 412–418. Bibcode:1980Natur.284..412Z. doi:10.1038/284412a0. ISSN 1476-4687. PMID 6767195. S2CID 4310049.
- ^ Roberson D, Davidoff J, Davies IR, Shapiro LR (November 2006). "Colour categories and category acquisition in Himba and English" (PDF). In Pitchford N, Biggam CP (eds.). Progress in Colour Studies. Vol. II. Psychological aspects. John Benjamins Publishing. pp. 159–72. ISBN 978-90-272-9302-2.
- ^ Nänni, Jürg (2008). Visual perception - an interactive journey of discovery through our visual system (in German and English). Sulgen Zurich, Switzerland: Niggli AG. ISBN 978-3-7212-0618-0.
- ^ Fairchild MD (2005). "8. Chromatic Adaptation". Color Appearance Models. Wiley. p. 146. ISBN 978-0-470-01216-1.
- ^ Süsstrunk S. "Chromatic Adaptation". The Image and Visual Representation Lab (IVRL). Archived from the original on 2011-08-18.
- ^ Lindbloom B. "Chromatic Adaptation". Lindbloom.com. Archived from the original on 2011-09-26.
- ^ Cuthill IC (1997). "Ultraviolet vision in birds". In Slater PJ (ed.). Advances in the Study of Behavior. Vol. 29. Oxford, England: Academic Press. p. 161. ISBN 978-0-12-004529-7.
- ^ Jamieson BG (2007). Reproductive Biology and Phylogeny of Birds. Charlottesville VA: University of Virginia. p. 128. ISBN 978-1-57808-386-2.
- ^ Varela FJ, Palacios AG, Goldsmith TH (1993). "Color vision of birds". In Zeigler HP, Bischof HJ (eds.). Vision, Brain, and Behavior in Birds. MIT Press. pp. 77–94. ISBN 978-0-262-24036-9.
- ^ "True or False? The common goldfish is the only animal that can see both infrared and ultra-violet light". Skeptive. Archived from the original on December 24, 2013. Retrieved September 28, 2013.
- ^ Neumeyer C (2012). "Chapter 2: Color Vision in Goldfish and Other Vertebrates". In Lazareva O, Shimizu T, Wasserman E (eds.). How Animals See the World: Comparative Behavior, Biology, and Evolution of Vision. Oxford Scholarship Online. ISBN 978-0-195-33465-4.
- ^ Jacobs GH (August 1993). "The distribution and nature of colour vision among the mammals". Biological Reviews of the Cambridge Philosophical Society. 68 (3): 413–71. doi:10.1111/j.1469-185X.1993.tb00738.x. PMID 8347768. S2CID 24172719.
- ^ Menzel R, Steinmann E, De Souza J, Backhaus W (1988-05-01). "Spectral Sensitivity of Photoreceptors and Colour Vision in the Solitary Bee, Osmia Rufa". Journal of Experimental Biology. 136 (1): 35–52. doi:10.1242/jeb.136.1.35. ISSN 0022-0949. Archived from the original on 2016-03-04.
- ^ a b Osorio D, Vorobyev M (September 2008). "A review of the evolution of animal colour vision and visual communication signals". Vision Research. 48 (20): 2042–51. doi:10.1016/j.visres.2008.06.018. PMID 18627773. S2CID 12025276.
- ^ Arikawa K (November 2003). "Spectral organization of the eye of a butterfly, Papilio". Journal of Comparative Physiology A: Neuroethology, Sensory, Neural & Behavioral Physiology. 189 (11): 791–800. doi:10.1007/s00359-003-0454-7. PMID 14520495. S2CID 25685593.
- ^ Cronin TW, Marshall NJ (1989). "A retina with at least ten spectral types of photoreceptors in a mantis shrimp". Nature. 339 (6220): 137–40. Bibcode:1989Natur.339..137C. doi:10.1038/339137a0. S2CID 4367079.
- ^ Kelber A, Vorobyev M, Osorio D (February 2003). "Animal colour vision--behavioural tests and physiological concepts". Biological Reviews of the Cambridge Philosophical Society. 78 (1): 81–118. doi:10.1017/S1464793102005985. PMID 12620062. S2CID 7610125.
- ^ Thompson E (1995). "Introducing Comparative Colour Vision". Colour vision : a study in cognitive science and the philosophy of perception. London: Routledge. p. 149. ISBN 978-0-203-41767-6.
- ^ Roth LS, Lundström L, Kelber A, Kröger RH, Unsbo P (March 2009). "The pupils and optical systems of gecko eyes". Journal of Vision. 9 (3): 27.1–11. doi:10.1167/9.3.27. PMID 19757966.
- ^ Yovanovich CA, Koskela SM, Nevala N, Kondrashev SL, Kelber A, Donner K (April 2017). "The dual rod system of amphibians supports colour discrimination at the absolute visual threshold". Philosophical Transactions of the Royal Society of London. Series B, Biological Sciences. 372 (1717). doi:10.1098/rstb.2016.0066. PMC 5312016. PMID 28193811.
- ^ Hunt JE, Bruno JR, Pratt KG (May 12, 2020). "An Innate Color Preference Displayed by Xenopus Tadpoles Is Persistent and Requires the Tegmentum". Frontiers in Behavioral Neuroscience. 14 (71): 71. doi:10.3389/fnbeh.2020.00071. PMC 7235192. PMID 32477078.
- ^ Shozo Yokoyama and F. Bernhard Radlwimmera, "The Molecular Genetics of Red and Green Color Vision in Mammals", Genetics, Vol. 153, 919–932, October 1999.
- ^ Jacobs GH, Deegan JF (April 2001). "Photopigments and colour vision in New World monkeys from the family Atelidae". Proceedings. Biological Sciences. 268 (1468): 695–702. doi:10.1098/rspb.2000.1421. PMC 1088658. PMID 11321057.
- ^ Jacobs GH, Deegan JF, Neitz J, Crognale MA, Neitz M (September 1993). "Photopigments and color vision in the nocturnal monkey, Aotus". Vision Research. 33 (13): 1773–83. CiteSeerX 10.1.1.568.1560. doi:10.1016/0042-6989(93)90168-V. PMID 8266633. S2CID 3745725.
- ^ Mollon JD, Bowmaker JK, Jacobs GH (September 1984). "Variations of colour vision in a New World primate can be explained by polymorphism of retinal photopigments". Proceedings of the Royal Society of London. Series B, Biological Sciences. 222 (1228): 373–99. Bibcode:1984RSPSB.222..373M. doi:10.1098/rspb.1984.0071. PMID 6149558. S2CID 24416536.
- ^ Sternberg RJ (2006). Cognitive Psychology (4th ed.). Thomson Wadsworth.
- ^ Arrese CA, Beazley LD, Neumeyer C (March 2006). "Behavioural evidence for marsupial trichromacy". Current Biology. 16 (6): R193-4. doi:10.1016/j.cub.2006.02.036. PMID 16546067.
- ^ Steven P (1997). How the Mind Works. New York: Norton. p. 191. ISBN 978-0-393-04535-2.
- ^ Koyanagi M, Nagata T, Katoh K, Yamashita S, Tokunaga F (February 2008). "Molecular evolution of arthropod color vision deduced from multiple opsin genes of jumping spiders". Journal of Molecular Evolution. 66 (2): 130–7. Bibcode:2008JMolE..66..130K. doi:10.1007/s00239-008-9065-9. PMID 18217181. S2CID 23837628.
- ^ Hambling D (May 30, 2002). "Let the light shine in: You don't have to come from another planet to see ultraviolet light". EducationGuardian.co.uk. Archived from the original on November 23, 2014.
- ^ Jacobs GH, Neitz J, Deegan JF (October 1991). "Retinal receptors in rodents maximally sensitive to ultraviolet light". Nature. 353 (6345): 655–6. Bibcode:1991Natur.353..655J. doi:10.1038/353655a0. PMID 1922382. S2CID 4283145.
- ^ Varela FJ, Palacios AG, Goldsmith TM (1993). Bischof HJ, Zeigler HP (eds.). Vision, brain, and behavior in birds. Cambridge, Mass: MIT Press. pp. 77–94. ISBN 978-0-262-24036-9.
- ^ Cuthill IC, Partridge JC, Bennett AT, Church SC, Hart NS, Hunt S (2000). "Ultraviolet Vision in Birds". Advances in the Study of Behavior. Vol. 29. pp. 159–214.
- ^ Jacobs DE, Gallo O, Cooper EA, Pulli K, Levoy M (May 2015). "Simulating the Visual Experience of Very Bright and Very Dark Scenes". ACM Trans. Graph. 34 (3): 15. doi:10.1145/2714573. S2CID 14960893.
Further reading[edit]
- Biggs T, McPhail S, Nassau K, Patankar H, Stenerson M, Maulana F, Douma M. Smith SE (ed.). "What colors do animals see?". Web Exhibits. Institute for Dynamic Educational Advancement (IDEA).
- Feynman RP (2015). "Color Vision". In Gottlieb MA, Pfeiffer R (eds.). Feynman lectures on physics. Volume, Mainly mechanics, radiation, and heat (New millennium ed.). New York: Basic Books. ISBN 978-0-465-04085-8 – via California Institute of Technology.
- Gouras P (May 2009). "Color Vision". Webvision. University of Utah School of Medicine. PMID 21413395.
- McEvoy B (2008). "Color vision". Retrieved 2012-03-30.
- Rogers A (26 February 2015). "The Science of Why No One Agrees on the Color of This Dress". Wired.