Rhodium: Difference between revisions
| Line 3: | Line 3: | ||
'''Rhodium''' ({{pron-en|ˈroʊdiəm}} {{respell|ROH|dee-əm}}) is a [[chemical element]] that is a rare, silvery-white, hard [[transition metal]] and a member of the [[platinum group]]. Rhodium is found in platinum ores and is used in [[alloy]]s with platinum and as a [[catalyst]]. It is abbreviated to '''Rh''' and has [[atomic number]] 45. It is one of the most expensive [[precious metal]]s. |
'''Rhodium''' ({{pron-en|ˈroʊdiəm}} {{respell|ROH|dee-əm}}) is a [[chemical element]] that is a rare, silvery-white, hard [[transition metal]] and a member of the [[platinum group]]. Rhodium is found in platinum ores and is used in [[alloy]]s with platinum and as a [[catalyst]]. It is abbreviated to '''Rh''' and has [[atomic number]] 45. It is one of the most expensive [[precious metal]]s. |
||
== Characteristics == |
== Characteristics ==Epp balls hard! A-Bent is the bomb! |
||
Rhodium is a hard silvery white and durable metal that has a high [[reflectance]]. Rhodium metal does not normally form an [[oxide]], even when heated.<ref name="ASM13B">{{cite book | last = Cramer | first = Stephen; S., Jr Covino, Bernard | authorlink= | coauthors= | title = ASM handbook | date = 1990 | publisher = ASM International | location = Materials Park, OH | isbn=0-87170-707-1 | pages = 393–396 | url = http://books.google.com/books?id=QV0sWU2qF5oC&pg=PA396}}</ref> [[Oxygen]] is absorbed from the [[atmosphere]] at the [[melting point]] of rhodium, but on solidification, the oxygen is released.<ref> {{cite book | last = Emsley | first = John | title = Nature's Building Blocks | edition = (Hardcover, First Edition) | publisher = [[Oxford University Press]] | date = 2001 | pages = 363 | isbn = 0198503407 }}</ref> Rhodium has both a higher melting point and lower [[density]] than [[platinum]]. It is not attacked by [[acid]]s: it is completely insoluble in [[nitric acid]] and dissolves slightly in [[aqua regia]]. |
Rhodium is a hard silvery white and durable metal that has a high [[reflectance]]. Rhodium metal does not normally form an [[oxide]], even when heated.<ref name="ASM13B">{{cite book | last = Cramer | first = Stephen; S., Jr Covino, Bernard | authorlink= | coauthors= | title = ASM handbook | date = 1990 | publisher = ASM International | location = Materials Park, OH | isbn=0-87170-707-1 | pages = 393–396 | url = http://books.google.com/books?id=QV0sWU2qF5oC&pg=PA396}}</ref> [[Oxygen]] is absorbed from the [[atmosphere]] at the [[melting point]] of rhodium, but on solidification, the oxygen is released.<ref> {{cite book | last = Emsley | first = John | title = Nature's Building Blocks | edition = (Hardcover, First Edition) | publisher = [[Oxford University Press]] | date = 2001 | pages = 363 | isbn = 0198503407 }}</ref> Rhodium has both a higher melting point and lower [[density]] than [[platinum]]. It is not attacked by [[acid]]s: it is completely insoluble in [[nitric acid]] and dissolves slightly in [[aqua regia]]. |
||
Revision as of 14:13, 14 January 2010
 | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rhodium | ||||||||||||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Pronunciation | /ˈroʊdiəm/ | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Appearance | Silvery white metallic | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Standard atomic weight Ar°(Rh) | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Rhodium in the periodic table | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic number (Z) | 45 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Group | group 9 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Period | period 5 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Block | d-block | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Electron configuration | [Kr] 4d8 5s1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrons per shell | 2, 8, 18, 16, 1 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Physical properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Phase at STP | solid | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Melting point | 2237 K (1964 °C, 3567 °F) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Boiling point | 3968 K (3695 °C, 6683 °F) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Density (at 20° C) | 12.423 g/cm3[3] | |||||||||||||||||||||||||||||||||||||||||||||||||||
| when liquid (at m.p.) | 10.7 g/cm3 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of fusion | 26.59 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Heat of vaporization | 493 kJ/mol | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar heat capacity | 24.98 J/(mol·K) | |||||||||||||||||||||||||||||||||||||||||||||||||||
Vapor pressure
| ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Oxidation states | −3[4], −1, 0, +1, +2, +3, +4, +5, +6, +7[5] (an amphoteric oxide) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Electronegativity | Pauling scale: 2.28 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Ionization energies |
| |||||||||||||||||||||||||||||||||||||||||||||||||||
| Atomic radius | empirical: 134 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Covalent radius | 142±7 pm | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Other properties | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Natural occurrence | primordial | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Crystal structure | face-centered cubic (fcc) (cF4) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Lattice constant | a = 380.34 pm (at 20 °C)[3] | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal expansion | 8.46×10−6/K (at 20 °C)[3] | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Thermal conductivity | 150 W/(m⋅K) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Electrical resistivity | 43.3 nΩ⋅m (at 0 °C) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Magnetic ordering | paramagnetic[6] | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Molar magnetic susceptibility | +111.0×10−6 cm3/mol (298 K)[7] | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Young's modulus | 380 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Shear modulus | 150 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Bulk modulus | 275 GPa | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Speed of sound thin rod | 4700 m/s (at 20 °C) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Poisson ratio | 0.26 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Mohs hardness | 6.0 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Vickers hardness | 1100–8000 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Brinell hardness | 980–1350 MPa | |||||||||||||||||||||||||||||||||||||||||||||||||||
| CAS Number | 7440-16-6 | |||||||||||||||||||||||||||||||||||||||||||||||||||
| History | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| Discovery and first isolation | William Hyde Wollaston (1804) | |||||||||||||||||||||||||||||||||||||||||||||||||||
| Isotopes of rhodium | ||||||||||||||||||||||||||||||||||||||||||||||||||||
| ||||||||||||||||||||||||||||||||||||||||||||||||||||
Rhodium (Template:Pron-en ROH-dee-əm) is a chemical element that is a rare, silvery-white, hard transition metal and a member of the platinum group. Rhodium is found in platinum ores and is used in alloys with platinum and as a catalyst. It is abbreviated to Rh and has atomic number 45. It is one of the most expensive precious metals.
== Characteristics ==Epp balls hard! A-Bent is the bomb! Rhodium is a hard silvery white and durable metal that has a high reflectance. Rhodium metal does not normally form an oxide, even when heated.[9] Oxygen is absorbed from the atmosphere at the melting point of rhodium, but on solidification, the oxygen is released.[10] Rhodium has both a higher melting point and lower density than platinum. It is not attacked by acids: it is completely insoluble in nitric acid and dissolves slightly in aqua regia.
Chemical properties
Rhodium belongs to group 9 in the periodic table,
| Z | Element | No. of electrons/shell |
|---|---|---|
| 27 | cobalt | 2, 8, 15, 2 |
| 45 | rhodium | 2, 8, 18, 16, 1 |
| 77 | iridium | 2, 8, 18, 32, 15, 2 |
| 109 | meitnerium | 2, 8, 18, 32, 32, 15, 2 |
but has an atypical configuration in its outermost electron shells compared to the rest of the members. (This can also be observed in the neighborhood of niobium (41), ruthenium (44), rhodium (45), and palladium (46).)
| Oxidation states of rhodium | |
|---|---|
| +0 | Rh 4(CO) 12 |
| +1 | RhCl(PH 3) 2 |
| +2 | Rh 2(O 2CCH 3) |
| +3 | RhCl 3, Rh 2O 3 |
| +4 | RhF 4, RhO 2 |
| +5 | RhF 5, Sr 3LiRhO 6 |
| +6 | RhF 6 |
Common oxidation states of rhodium is +3, but oxidation states from +0 to +6 are observed.[11]
Unlike ruthenium and osmium, rhodium forms no volatile oxygen compounds. The known stable oxides include Rh
2O
3, RhO
2, RhO
2·xH
2O, Na
2RhO
3, Sr
3LiRhO
6 and Sr
3NaRhO
6[12]. Halogen compounds are known in nearly the full range of possible oxidation states. Rhodium(III) chloride, rhodium(IV) fluoride, rhodium(V) fluoride and rhodium(VI) fluoride are some examples. The lower oxidation states are only stable if ligands are present.[13]

The best known example is the Wilkinson's catalyst chlorotris(triphenylphosphine)rhodium(I). The catalyst is for example used for the hydrogenation of alkenes.[14]
Isotopes
Naturally occurring rhodium is composed of only one isotope, 103Rh. The most stable radioisotopes are 101Rh with a half-life of 3.3 years, 102Rh with a half-life of 207 days, 102mRh with a half-life of 2.9 years, and 99Rh with a half-life of 16.1 days. Twenty other radioisotopes have been characterized with atomic weights ranging from 92.926 u (93Rh) to 116.925 u (117Rh). Most of these have half-lives that are less than an hour except 100Rh (half-life: 20.8 hours) and 105Rh (half-life: 35.36 hours). There are also numerous meta states with the most stable being 102mRh (0.141 MeV) with a half-life of about 2.9 years and 101mRh (0.157 MeV) with a half-life of 4.34 days. See isotopes of rhodium.[15]
The primary decay mode before the only stable isotope, 103Rh, is electron capture and the primary mode after is beta emission. The primary decay product before 103Rh is ruthenium and the primary product after is palladium.[16]
History
Rhodium (Greek rhodon (ῥόδον) meaning "rose") was discovered in 1803 by William Hyde Wollaston,[17][18] soon after his discovery of palladium.[19][20] He made this discovery in England using crude platinum ore that he presumably obtained from South America.[21]
His procedure involved dissolving the ore in aqua regia and neutralizing the acid with sodium hydroxide (NaOH). He then precipitated the platinum by adding ammonium chloride, NH
4Cl, as ammonium chloroplatinate. All other metals like copper, lead, palladium and rhodium were precipitated with zinc. Diluted nitric acid dissolved all but palladium and rhodium, which were dissolved in aqua regia and the rhodium was precipitated by the addition of sodium chloride as Na
3[RhCl
6]·nH
2O. After washing with ethanol, the rose red precipitate was reacted with zinc forming rhodium metal.[22]
Applications

The primary use of this element is in automobiles as a catalytic converter, which converts harmful emissions from the engine into less harmful gases.[23][24]
Catalytic converter

In 2007 81%[23] of the world production of rhodium was consumed to produce three-way catalytic converters.[23] Rhodium shows some advantages over the other platinum metals in the reduction of nitrogen oxides to nitrogen and oxygen:[25]
- 2 NO
x → x O
2 + N
2
The recycling of catalytic converters also became a valuable source for rhodium. In 2007 5.7 t were extracted from this source. Compared to the 22 t which had been mined, this is a relatively high recycling rate.[23]
Other uses
Rhodium is used as an alloying agent for hardening and improving the corrosion resistance[9] of platinum and palladium. These alloys are used in furnace windings, bushings for glass fiber production, thermocouple elements, electrodes for aircraft spark plugs, and laboratory crucibles.[26] Other uses include:
- An electrical contact material due to its low electrical resistance, low and stable contact resistance, and high corrosion resistance.[27]
- Plated rhodium, made by electroplating or evaporation, is extremely hard and is used for optical instruments.[28]

- This metal finds use in jewelry and for decorations. It is electroplated on white gold and platinum to give it a reflective white surface. This is known as rhodium flashing in the jewelry business. It also may be used in coating sterling silver in order to strengthen the metal from tarnish (silver sulfide, Ag2S--caused by hydrogen sulfide, H2S in the atmosphere). Solid (pure) rhodium jewelry is very rare, because the metal has both high melting point and poor malleability (making such jewelry very hard to fabricate) rather than due to its high price.[29]
- It is also a highly useful catalyst in a number of industrial processes. Notably, it is used in the automobile catalytic converters and for catalytic carbonylation of methanol to produce acetic acid by the Monsanto process)[30] It is also used to catalyze addition of hydrosilanes to molecular double bonds, a process important in manufacture of certain silicone rubbers.[31]
- Rhodium catalysts can be used to reduce benzene to cyclohexane.[32]
- The complex of a rhodium ion with BINAP gives a widely used chiral catalyst for chiral synthesis, as in the synthesis of menthol.[33]
- It is also used as a filter in mammography systems because of the characteristic X-rays it produces.[34]
- It is also used in high quality pen surfaces due to its high chemical and mechanical resistance. These pens include Graf von Faber-Castell[35] and Caran D'ache[36].
- Rhodium neutron detectors are used in Combustion Engineering Nuclear Reactors to measure neutron flux levels - a method that requires a digital filter to determine the current neutron flux level, as there are three signals generated: immediate, a few seconds later, and a minute later, each with its own signal level, and all three are combined in the rhodium detector signals. The three Palo Verde nuclear reactors each have 305 rhodium neutron detectors, 61 detectors on each of 5 vertical levels, providing an accurate 3-D "picture" of reactivity, allowing fine tuning to most economically burn the nuclear fuel.[37]
Occurrence
Normal mining
The industrial extraction of rhodium is complex as the metal occurs in ores mixed with other metals such as palladium, silver, platinum, and gold. It is found in platinum ores and obtained free as a white inert metal which is very difficult to fuse. Principal sources of this element are located in South Africa, in river sands of the Ural Mountains, and in North America, including the copper-nickel sulfide mining area of the Sudbury, Ontario region. Although the quantity at Sudbury is very small, the large amount of processed nickel ore makes rhodium recovery cost effective. The main exporter of rhodium is South Africa (>80%) followed by Russia.[38] The annual world production of this element is only about 25 tons and there are very few rhodium-bearing minerals. As of October 2007, rhodium costed approximately eight times more than gold, 450 times more than silver, and 27,250 times more than copper by weight. Rhodium's typical historical price is about $1,000/troy oz,[39] but in recent years, it has increased to about $4500/troy oz.[40] In 2008 the price briefly rose above $10,000 per ounce.[40] The 3rd quarter 2008 economic slowdown pushed rhodium prices sharply back below $1,000 per ounce, however, bouncing up to $2,750 by early 2010 (over twice the gold price).[40]
Fission product
It is also possible to extract rhodium from used nuclear fuel, which contains rhodium (1 kg of the fission products of 235U contains 13.3 grams of 103Rh). As a typical used fuel has 3% fission products by weight, it will contain about 400 grams of rhodium per ton of used fuel. The longest lived radioisotope of rhodium is 102mRh which has a half life of 2.9 years, whereas the ground state (102Rh) has a half life of 207 days.
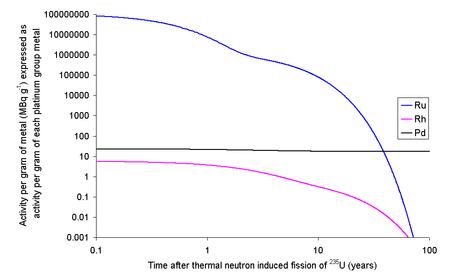
One kilogram of fission rhodium will contain 6.62 ng of 102Rh and 3.68 ng of 102mRh. As 102Rh decays by beta decay to either 102Ru (80%) (some positron emission will occur) or 102Pd (20%) (gamma ray photons with about 500 keV are generated) and the excited state decays by beta decay (electron capture) to 102Ru (gamma ray photons with about 1 MeV are generated). If the fission occurs in an instant then 13.3 grams of rhodium will contain 67.1 MBq (1.81 mCi) of 102Rh and 10.8 MBq (291 μCi) of 102mRh. As it is normal to allow used nuclear fuel to rest for about five years before reprocessing, much of this activity will decay leaving 4.7 MBq of 102Rh and 5.0 MBq of 102mRh. If the rhodium metal was then left for 20 years after fission, then the 13.3 grams of rhodium metal would contain 1.3 kBq of 102Rh and 500 kBq of 102mRh. At first glance, the rhodium might be adding to the resource value of reprocessed fission waste, but the cost of the separation of rhodium from other metals needs to be considered.[41]
Precautions
Rhodium metal is, as a noble metal, inert. However, chemical complexes of rhodium can be reactive. Lethal intake (LD50) for rats is 12.6 mg/kg of rhodium chloride (RhCl
3).[42] Rhodium compounds can strongly stain human skin. The element plays no biological role in humans. If used in elemental form rather than as compounds, the metal is harmless.[43]
Ornamental uses
Rhodium has been used for honours, or to symbolize wealth, when more commonly used metals such as silver, gold, or platinum are deemed insufficient. In 1979 the Guinness Book of World Records gave Paul McCartney a rhodium-plated disc for being history's all-time best-selling songwriter and recording artist.[44]
See also
References
- ^ "Standard Atomic Weights: Rhodium". CIAAW. 2017.
- ^ Prohaska, Thomas; Irrgeher, Johanna; Benefield, Jacqueline; Böhlke, John K.; Chesson, Lesley A.; Coplen, Tyler B.; Ding, Tiping; Dunn, Philip J. H.; Gröning, Manfred; Holden, Norman E.; Meijer, Harro A. J. (2022-05-04). "Standard atomic weights of the elements 2021 (IUPAC Technical Report)". Pure and Applied Chemistry. doi:10.1515/pac-2019-0603. ISSN 1365-3075.
- ^ a b c Arblaster, John W. (2018). Selected Values of the Crystallographic Properties of Elements. Materials Park, Ohio: ASM International. ISBN 978-1-62708-155-9.
- ^ Ellis J E. Highly Reduced Metal Carbonyl Anions: Synthesis, Characterization, and Chemical Properties. Adv. Organomet. Chem, 1990, 31: 1-51.
- ^ Rh(VII) is known in the RhO3+ cation, see Da Silva Santos, Mayara; Stüker, Tony; Flach, Max; Ablyasova, Olesya S.; Timm, Martin; von Issendorff, Bernd; Hirsch, Konstantin; Zamudio‐Bayer, Vicente; Riedel, Sebastian; Lau, J. Tobias (2022). "The Highest Oxidation State of Rhodium: Rhodium(VII) in [RhO3]+". Angew. Chem. Int. Ed. 61 (38): e202207688. doi:10.1002/anie.202207688. PMC 9544489. PMID 35818987.
- ^ Lide, D. R., ed. (2005). "Magnetic susceptibility of the elements and inorganic compounds". CRC Handbook of Chemistry and Physics (PDF) (86th ed.). Boca Raton (FL): CRC Press. ISBN 0-8493-0486-5.
- ^ Weast, Robert (1984). CRC, Handbook of Chemistry and Physics. Boca Raton, Florida: Chemical Rubber Company Publishing. pp. E110. ISBN 0-8493-0464-4.
- ^ Kondev, F. G.; Wang, M.; Huang, W. J.; Naimi, S.; Audi, G. (2021). "The NUBASE2020 evaluation of nuclear properties" (PDF). Chinese Physics C. 45 (3): 030001. doi:10.1088/1674-1137/abddae.
- ^ a b Cramer, Stephen; S., Jr Covino, Bernard (1990). ASM handbook. Materials Park, OH: ASM International. pp. 393–396. ISBN 0-87170-707-1.
{{cite book}}: Cite has empty unknown parameter:|coauthors=(help)CS1 maint: multiple names: authors list (link) - ^ Emsley, John (2001). Nature's Building Blocks ((Hardcover, First Edition) ed.). Oxford University Press. p. 363. ISBN 0198503407.
- ^ Holleman, Arnold F. (1985). Lehrbuch der Anorganischen Chemie (91–100 ed.). Walter de Gruyter. pp. 1056–1057. ISBN 3-11-007511-3.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help)CS1 maint: extra punctuation (link) - ^ Reisner, B. A. (1998). "Sr3ARhO6(A = Li, Na): Crystallization of a Rhodium(V) Oxide from Molten Hydroxide". Of the American Chemical Society. 120: 9682–9989. doi:10.1021/ja974231q.
{{cite journal}}: Unknown parameter|Srignored (help); Unknown parameter
3ARhO
6 (A=|coauthor=ignored (|author=suggested) (help); no-break space character in|title=at position 22 (help) - ^ Griffith, W. P. The Rarer Platinum Metals; John Wiley and Sons: New York, 1976; p 313.
- ^ Osborn, J. A.; Jardine, F. H.; Young, J. F.; Wilkinson, G. (1966). "The Preparation and Properties of Tris(triphenylphosphine)halogenorhodium(I) and Some Reactions Thereof Including Catalytic Homogeneous Hydrogenation of Olefins and Acetylenes and Their Derivatives". Journal of the Chemical Society A: 1711–1732. doi:10.1039/J19660001711.
- ^ Audi, G. (2003). "The NUBASE Evaluation of Nuclear and Decay Properties". Nuclear Physics A. 729. Atomic Mass Data Center: 3–128. doi:10.1016/j.nuclphysa.2003.11.001.
- ^ David R. Lide (ed.), Norman E. Holden in CRC Handbook of Chemistry and Physics, 85th Edition CRC Press. Boca Raton, Florida (2005). Section 11, Table of the Isotopes.
- ^ "WebElements - The History of Rhodium". Retrieved 2009-02-06.
{{cite web}}: Text "webelements" ignored (help) - ^ Wollaston, W. H. (1805). "On the Discovery of Palladium; With Observations on Other Substances Found with Platina". Philosophical Transactions of the Royal Society of London. 95: 316–330. doi:10.1098/rstl.1805.0024.
- ^ W. P. Griffith (2003). "Rhodium and Palladium - Events Surrounding Its Discovery". Platinum Metals Review. 47 (4): 175–183.
- ^ Wollaston, W. H. (1804). "On a New Metal, Found in Crude Platina". Philosophical Transactions of the Royal Society of London. 94: 419–430. doi:10.1098/rstl.1804.0019.
- ^ Lide, David R (2004). CRC handbook of chemistry and physics: a ready-reference book of chemical and physical data. Boca Raton: CRC Press. pp. 4–26. ISBN 0-8493-0485-7.
{{cite book}}: Cite has empty unknown parameter:|coauthors=(help) - ^ Griffith, W. P. (2003). "Bicentenary of Four Platinum Group Metals: Osmium and iridium – events surrounding their discoveries". Platinum Metals Review. 47 (4): 175–183.
- ^ a b c d George, Micheal W. "Commodity Report: Platinum-Group Metals" (PDF). United States Geological Survey USGS. Retrieved 2008-09-16.
- ^ George, Micheal W. "2006 Minerals Yearbook: Platinum-Group Metals" (PDF). United States Geological Survey USGS. Retrieved 2008-09-16.
- ^ Shelef, M. (1994). "Why Rhodium in Automotive Three-Way Catalysts?". Catalysis Reviews. 36 (3): 433–457. doi:10.1080/01614949408009468.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Lide, David R (2004). CRC handbook of chemistry and physics: a ready-reference book of chemical and physical data. Boca Raton: CRC Press. pp. 4–26. ISBN 0-8493-0485-7.
{{cite book}}: Cite has empty unknown parameter:|coauthors=(help) - ^ Weisberg, Alfred M. (1999). "Rhodium plating". Metal Finishing. 97 (1): 296–299. doi:10.1016/S0026-0576(00)83088-3.
- ^ Smith, Warren J. (2007). "Reflectors". Modern optical engineering: the design of optical systems. McGraw-Hill. pp. 247–248. ISBN 9780071476874.
- ^ Fischer, Torkel; Fregert, S; Gruvberger, B; Rystedt, I (1984). "Contact sensitivity to nickel in white gold". Contact Dermatitis. 10 (1): 23–24. doi:10.1111/j.1600-0536.1984.tb00056.x. PMID 6705515.
- ^ Roth, James F. (1975). "Rhodium Catalysed Carbonylation of Methanol" (PDF). Platinum Metals Review. 19 (1 January): 12–14.
- ^ M. Heidingsfeldova and M. Capka "Rhodium complexes as catalysts for hydrosilylation crosslinking of silicone rubber" Journal of Applied Polymer Science 30 (2003) 1837
- ^ S. B. Halligudi et al. "Hydrogenation of benzene to cyclohexane catalyzed by rhodium(I) complex supported on montmorillonite clay" Reaction Kinetics and Catalysis Letters 48 (1992) 547
- ^ S. Akutagawa "Asymmetric synthesis by metal BINAP catalysts" Applied Catalysis A: 128 (1995) 171
- ^ C P McDonagh, J L Leake and S A Beaman "Optimum x-ray spectra for mammography: choice of K-edge filters for tungsten anode tubes" Phys. Med. Biol. 29 (1984) 249
- ^ Guilloche luxury pen range by Graf von Faber-Castell
- ^ Caran D'Ache Ecridor Type 55 Rhodium Fountain Pen
- ^ Sokolov Pochivalin, G. P.; Shipovskikh, Yu. M.; Garusov, Yu. V.; Chernikov O. G.; Shevchenko V. G., A. P. (1993 pages = 365–367). "Rhodium self-powered detector for monitoring neutron fluence, energy production, and isotopic composition of fuel". Atomic Energy. 74. doi:10.1007/BF00844622.
{{cite journal}}: Check date values in:|year=(help); Missing pipe in:|year=(help)CS1 maint: multiple names: authors list (link) CS1 maint: year (link) - ^ Chevalier, Patrick (?). "Mineral Yearbook: Platinum Group Metals" (PDF). Natural Resources Canada. Retrieved 2008-10-17.
{{cite web}}: Check date values in:|date=(help) - ^ http://periodic.lanl.gov/elements/45.html
- ^ a b c KITCO Rhodium Price Charts
- ^ Bush, R. P. (1991). "Recovery of Platinum Group Metals from High Level Radioactive Waste". Platinum Metals Review. 35 (4): 202–208.
- ^ Landolt, Robert R. (1972). "Studies on the toxicity of rhodium trichloride in rats and rabbits". Toxicology and Applied Pharmacology. 21 (4): 589–590. doi:10.1016/0041-008X(72)90016-6. PMID 5047055.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Leikin, Jerrold B. (2008). Poisoning and Toxicology Handbook. Informa Health Care. p. 846. ISBN 9781420044799.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ "Hit & Run: Ring the changes". The Independent. Retrieved 2009-06-06.
External links
- WebElements.com – Rhodium
- Current Rhodium price
- Rhodium Technical and Safety Data
- Los Alamos National Laboratory – Rhodium


