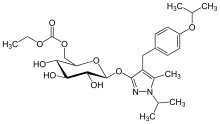
Remogliflozin etabonate
 | |
| Clinical data | |
|---|---|
| Routes of administration | By mouth |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C26H38N2O9 |
| Molar mass | 522.595 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Remogliflozin etabonate (INN/USAN)[1] is a drug of the gliflozin class for the treatment of non-alcoholic steatohepatitis ("NASH") and type 2 diabetes. Remogliflozin was discovered by the Japanese company Kissei Pharmaceutical and is currently being developed by BHV Pharma, a wholly owned subsidiary of North Carolina USA-based Avolynt, and Glenmark Pharmaceuticals through a collaboration with BHV.[2] Remogliflozin was commercially launched first in India by Glenmark in May 2019.
Clinical trials
Remogliflozin etabonate was shown to enhance urinary glucose excretion in rodents and humans. Early studies in diabetics improved plasma glucose levels.[3][4] Remogliflozin etabonate has been studied at doses up to 1000 mg.[5] A pair of 12-week phase 2b randomized clinical trials of diabetics published in 2015, found reductions in glycated hemoglobin and that it was generally well tolerated.[6]
Method of action
Remogliflozin etabonate is a pro-drug of remogliflozin. Remogliflozin inhibits the sodium-glucose transport proteins (SGLT), which are responsible for glucose reabsorption in the kidney. Blocking this transporter causes blood glucose to be eliminated through the urine.[7] Remogliflozin is selective for SGLT2.
See also
References
- ^ Statement on a nonproprietory name adopted by the USAN council
- ^ "Avolynt Announces Completion of Phase 2b BRID Study of SGLT2 Inhibitor Remogliflozin-Etabonate" (Press release). Avolynt, Inc. Retrieved July 24, 2018.
- ^ Mudaliar S, Armstrong DA, Mavian AA, O'Connor-Semmes R, Mydlow PK, Ye J, et al. (November 2012). "Remogliflozin etabonate, a selective inhibitor of the sodium-glucose transporter 2, improves serum glucose profiles in type 1 diabetes". Diabetes Care. 35 (11): 2198–200. doi:10.2337/dc12-0508. PMC 3476920. PMID 23011728.
- ^ Dobbins RL, O'Connor-Semmes R, Kapur A, Kapitza C, Golor G, Mikoshiba I, et al. (January 2012). "Remogliflozin etabonate, a selective inhibitor of the sodium-dependent transporter 2 reduces serum glucose in type 2 diabetes mellitus patients". Diabetes, Obesity & Metabolism. 14 (1): 15–22. doi:10.1111/j.1463-1326.2011.01462.x. PMID 21733056. S2CID 23372554.
- ^ Sykes AP, O'Connor-Semmes R, Dobbins R, Dorey DJ, Lorimer JD, Walker S, et al. (January 2015). "Randomized trial showing efficacy and safety of twice-daily remogliflozin etabonate for the treatment of type 2 diabetes". Diabetes, Obesity & Metabolism. 17 (1): 94–7. doi:10.1111/dom.12391. PMID 25223369. S2CID 6436562.
- ^ Sykes AP, Kemp GL, Dobbins R, O'Connor-Semmes R, Almond SR, Wilkison WO, et al. (January 2015). "Randomized efficacy and safety trial of once-daily remogliflozin etabonate for the treatment of type 2 diabetes". Diabetes, Obesity & Metabolism. 17 (1): 98–101. doi:10.1111/dom.12393. PMID 25238025. S2CID 25280330.
- ^ "Molecule of the Month: Dapagliflozin". Prous Science. November 2007. Archived from the original on January 6, 2008.
