Rhodanese
| Rhodanese-like domain | |||||||||
|---|---|---|---|---|---|---|---|---|---|
 | |||||||||
| Identifiers | |||||||||
| Symbol | Rhodanese | ||||||||
| Pfam | PF00581 | ||||||||
| InterPro | IPR001763 | ||||||||
| PROSITE | PDOC00322 | ||||||||
| SCOP2 | 2ora / SCOPe / SUPFAM | ||||||||
| OPM superfamily | 413 | ||||||||
| OPM protein | 2mpn | ||||||||
| CDD | cd00158 | ||||||||
| Membranome | 571 | ||||||||
| |||||||||
| thiosulfate sulfurtransferase | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| Identifiers | |||||||||
| EC no. | 2.8.1.1 | ||||||||
| CAS no. | 9026-04-4 | ||||||||
| Databases | |||||||||
| IntEnz | IntEnz view | ||||||||
| BRENDA | BRENDA entry | ||||||||
| ExPASy | NiceZyme view | ||||||||
| KEGG | KEGG entry | ||||||||
| MetaCyc | metabolic pathway | ||||||||
| PRIAM | profile | ||||||||
| PDB structures | RCSB PDB PDBe PDBsum | ||||||||
| Gene Ontology | AmiGO / QuickGO | ||||||||
| |||||||||
Rhodanese is a mitochondrial enzyme that detoxifies cyanide (CN−) by converting it to thiocyanate (SCN−, also known as "rhodanate").[1] In enzymatology, the common name is listed as thiosulfate sulfurtransferase (EC 2.8.1.1).[2] The diagram on the right shows the crystallographically-determined structure of rhodanese.
It catalyzes the following reaction:
- thiosulfate + cyanide sulfite + thiocyanate
Structure and mechanism
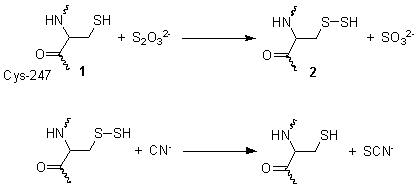
[edit]This reaction takes place in two steps. In the first step, thiosulfate is reduced by the thiol group on cysteine-247 1, to form a persulfide and a sulfite 2. In the second step, the persulfide reacts with cyanide to produce thiocyanate, re-generating the cysteine thiol 1.[3]
Rhodanese shares evolutionary relationship with a large family of proteins, including:
- Cdc25 phosphatase catalytic domain
- non-catalytic domains of eukaryotic dual-specificity MAPK-phosphatases
- non-catalytic domains of yeast PTP-type MAPK-phosphatases
- non-catalytic domains of yeast Ubp4, Ubp5, Ubp7
- non-catalytic domains of mammalian Ubp-Y
- Drosophila heat shock protein HSP-67BB
- several bacterial cold-shock and phage shock proteins
- plant senescence associated proteins
- catalytic and non-catalytic domains of rhodanese[4]
Rhodanese has an internal duplication. This domain is found as a single copy in other proteins, including phosphatases and ubiquitin C-terminal hydrolases.[5]
Clinical relevance
[edit]This reaction is important for the treatment of exposure to cyanide, since the thiocyanate formed is around 1 / 200 as toxic.[6]:p. 15938 The use of thiosulfate solution as an antidote for cyanide poisoning is based on the activation of this enzymatic cycle.
Human proteins
[edit]The human mitochondrial rhodanese gene is TST.
The following other human genes match the "Rhodanese-like" domain on InterPro, but are not the rodanase with its catalytic activity (see also the list of related families in #Structure and mechanism):
- M-phase inducer phosphatase: CDC25A; CDC25B; CDC25C;
- Dual specificity protein phosphatase: DUSP; DUSP1; DUSP2; DUSP4; DUSP5; DUSP6; DUSP7; DUSP10; DUSP16, aka MKP7;
- Thiosulfate:glutathione sulfurtransferase: KAT, now known as "TSTD1";
- Adenylyltransferase and sulfurtransferase: MOCS3;
- 3-mercaptopyruvate sulfurtransferase: MPST, also known as "TSTD2"
- Not an enzyme: TBCK; TSGA14;
- Ubiquitin carboxyl-terminal hydrolase: USP8;
- Unknown activity: TSTD3
Nomenclature
[edit]Although the standard nomenclature rules for enzymes indicate that their names are to end with the letters "-ase", rhodanese was first described in 1933,[7] prior to the 1955 establishment of the Enzyme Commission; as such, the older name had already attained widespread usage.
The systematic name of this enzyme class is "thiosulfate:cyanide sulfurtransferase". Other names in common use include "thiosulfate cyanide transsulfurase", "thiosulfate thiotransferase", "rhodanese", and "rhodanase".
References
[edit]- ^ Cipollone R, Ascenzi P, Tomao P, Imperi F, Visca P (2008). "Enzymatic detoxification of cyanide: clues from Pseudomonas aeruginosa Rhodanese". Journal of Molecular Microbiology and Biotechnology. 15 (2–3): 199–211. doi:10.1159/000121331. PMID 18685272. S2CID 25431686.
- ^ EC 2.8.1.1, at the International Union of Biochemistry and Molecular Biology
- ^ Cipollone R, Ascenzi P, Tomao P, Imperi F, Visca P (2008). "Enzymatic detoxification of cyanide: clues from Pseudomonas aeruginosa Rhodanese". Journal of Molecular Microbiology and Biotechnology. 15 (2–3): 199–211. doi:10.1159/000121331. PMID 18685272. S2CID 25431686.
- ^ "Thiosulphate sulfurtransferase, conserved site (IPR001307)". EMBL-EBI. InterPro.
- ^ Gliubich F, Gazerro M, Zanotti G, Delbono S, Bombieri G, Berni R (August 1996). "Active site structural features for chemically modified forms of rhodanese". The Journal of Biological Chemistry. 271 (35): 21054–61. doi:10.1074/jbc.271.35.21054. PMID 8702871.
- ^ Jaszczak E, Polkowska Ż, Narkowicz S, Namieśnik J (July 2017). "Cyanides in the environment-analysis-problems and challenges". Environmental Science and Pollution Research International. 24 (19): 15929–15948. doi:10.1007/s11356-017-9081-7. PMC 5506515. PMID 28512706.
- ^ Cipollone R, Ascenzi P, Visca P (February 2007). "Common themes and variations in the rhodanese superfamily". IUBMB Life. 59 (2): 51–9. doi:10.1080/15216540701206859. PMID 17454295.
External links
[edit]- Rhodanese at the U.S. National Library of Medicine Medical Subject Headings (MeSH)