4-Aminosalicylic acid
 | |
 | |
| Clinical data | |
|---|---|
| Trade names | Paser, Granupas, others |
| AHFS/Drugs.com | Monograph |
| License data | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Pharmacokinetic data | |
| Protein binding | 50–60% |
| Metabolism | liver |
| Excretion | kidney |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| NIAID ChemDB | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.000.557 |
| Chemical and physical data | |
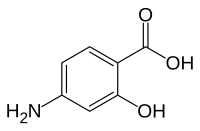
| Formula | C7H7NO3 |
| Molar mass | 153.137 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 150.5 °C (302.9 °F) |
| |
| |
| (verify) | |
4-Aminosalicylic acid, also known as para-aminosalicylic acid (PAS) and sold under the brand name Paser among others, is an antibiotic primarily used to treat tuberculosis.[2] Specifically it is used to treat active drug resistant tuberculosis together with other antituberculosis medications.[3] It has also been used as a second line agent to sulfasalazine in people with inflammatory bowel disease such as ulcerative colitis and Crohn's disease.[3] It is typically taken by mouth.[3]
Common side effects include nausea, abdominal pain, and diarrhea.[3] Other side effects may include liver inflammation and allergic reactions.[3] It is not recommended in people with end stage kidney disease.[3] While there does not appear to be harm with use during pregnancy it has not been well studied in this population.[3] 4-Aminosalicylic acid is believed to work by blocking the ability of bacteria to make folic acid.[3]
4-Aminosalicylic acid was first made in 1902, and came into medical use in 1943.[4] It is on the World Health Organization's List of Essential Medicines.[5]
Medical uses
[edit]The main use for 4-aminosalicylic acid is for the treatment of tuberculosis infections.[1][6]
In the United States, 4-aminosalicylic acid is indicated for the treatment of tuberculosis in combination with other active agents.[6]
In the European Union, it is used in combination with other medicines to treat adults and children from 28 days of age who have multi-drug resistant tuberculosis when combinations without this medicine cannot be used, either because the disease is resistant to them or because of their side effects.[1]
Tuberculosis
[edit]Aminosalicylic acid was introduced to clinical use in 1944. It was the second antibiotic found to be effective in the treatment of tuberculosis, after streptomycin. PAS formed part of the standard treatment for tuberculosis prior to the introduction of rifampicin and pyrazinamide.[7]
Its potency is less than that of the current five first-line drugs (isoniazid, rifampicin, ethambutol, pyrazinamide, and streptomycin) for treating tuberculosis and its cost is higher, but it is still useful in the treatment of multidrug-resistant tuberculosis.[8] PAS is always used in combination with other anti-TB drugs.[citation needed]
The dose when treating tuberculosis is 150 mg/kg/day divided into two to four daily doses; the usual adult dose is therefore approximately 2 to 4 grams four times a day. It is sold in the US as "Paser" by Jacobus Pharmaceutical, which comes in the form of 4 g packets of delayed-release granules. The drug should be taken with acid food or drink (orange, apple or tomato juice).[9] PAS was once available in a combination formula with isoniazid called Pasinah[10] or Pycamisan 33.[11]
4-Aminosalicylic acid was approved for medical use in the United States in June 1994, and for medical use in the European Union in April 2014.[12][1]
Inflammatory bowel disease
[edit]4-Aminosalicylic acid has also been used in the treatment of inflammatory bowel disease (ulcerative colitis and Crohn's disease),[13] but has been superseded by other drugs such as sulfasalazine and mesalazine.
Others
[edit]4-Aminosalicylic acid has been investigated for the use in manganese chelation therapy, and a 17-year follow-up study shows that it might be superior to other chelation protocols such as EDTA.[14]
Side effects
[edit]Gastrointestinal side-effects (nausea, vomiting, diarrhoea) are common; the delayed-release formulation is meant to help overcome this problem.[15] It is also a cause of drug-induced hepatitis. Patients with glucose-6-phosphate dehydrogenase deficiency should avoid taking aminosalicylic acid as it causes haemolysis.[16] Thyroid goitre is also a side-effect because aminosalicylic acid inhibits the synthesis of thyroid hormones.[17]
Drug interactions include elevated phenytoin levels. When taken with rifampicin, the levels of rifampicin in the blood fall by about half.[18]
It is not known whether it will harm an unborn baby.[19]
Pharmacology
[edit]With heat, 4-aminosalicylic acid is decarboxylated to produce CO2 and 3-aminophenol.[20]
Mode of action
[edit]4-Aminosalicylic acid has been shown to be a pro-drug and it is incorporated into the folate pathway by dihydropteroate synthase (DHPS) and dihydrofolate synthase (DHFS) to generate a hydroxyl dihydrofolate (Hydroxy-H2Pte and Hydroxy-H2PteGlu) antimetabolite, which competes with dihydrofolate at the binding site of dihydrofolate reductase (DHFR). The binding of Hydroxy-H2PteGlu to dihydrofolate reductase will block the enzymatic activity.[21]
Mechanism of action
[edit]Some studies have shown that principal antitubercular action of PAS occurs via poisoning of folate metabolism.[22]
Resistance
[edit]It was initially thought that resistance of 4-aminosalicylic acid came from a mutation affecting dihydrofolate reductase (DHFR). However, it was discovered that it was caused by a mutation affecting the dihydrofolate synthesis (DHFS) enzyme activity. The mutations of isoleucine 43, arginine 49, serine 150, phenylalanine 152, glutamate 153, and alanine 183 were found to affect the binding pocket of the dihydrofolate synthase enzyme. This will reduce the ability for hydroxy-H2Pte to bind to dihydrofolate synthase and preventing 4-aminosalicylic acid from poisoning the folate metabolism.[23]
History
[edit]4-Aminosalicylic acid was first synthesized by Seidel and Bittner in 1902.[4] It was rediscovered by the Swedish chemist Jörgen Lehmann upon the report that the tuberculosis bacterium avidly metabolized salicylic acid.[24] Lehmann first tried PAS as an oral TB therapy late in 1944. The first patient made a dramatic recovery.[25] The drug proved better than streptomycin, which had nerve toxicity and to which TB could easily develop resistance. In the 1948, researchers at Britain's Medical Research Council demonstrated that combined treatment with streptomycin and PAS was superior to either drug alone, and established the principle of combination therapy for tuberculosis.[8][4]
Other names
[edit]4-Aminosalicylic acid has many names including para-aminosalicylic acid, p-aminosalicylic acid, 4-ASA, and simply P.[medical citation needed]
References
[edit]- ^ a b c d "Granupas (previously Para-aminosalicylic acid Lucane)". European Medicines Agency (EMA). 17 September 2018. Archived from the original on 15 August 2020. Retrieved 3 April 2020.
- ^ World Health Organization (2009). Stuart MC, Kouimtzi M, Hill SR (eds.). WHO Model Formulary 2008. World Health Organization. p. 140. hdl:10665/44053. ISBN 9789241547659.
- ^ a b c d e f g h "Aminosalicylic Acid". The American Society of Health-System Pharmacists. Archived from the original on 20 December 2016. Retrieved 8 December 2016.
- ^ a b c Donald PR, Diacon AH (September 2015). "Para-aminosalicylic acid: the return of an old friend". The Lancet. Infectious Diseases. 15 (9): 1091–1099. doi:10.1016/s1473-3099(15)00263-7. PMID 26277036.
- ^ World Health Organization (2023). The selection and use of essential medicines 2023: web annex A: World Health Organization model list of essential medicines: 23rd list (2023). Geneva: World Health Organization. hdl:10665/371090. WHO/MHP/HPS/EML/2023.02.
- ^ a b "Paser- aminosalicylic acid granule, delayed release". DailyMed. 1 May 2010. Archived from the original on 3 August 2020. Retrieved 3 April 2020.
- ^ Mitchison DA (September 2000). "Role of individual drugs in the chemotherapy of tuberculosis". The International Journal of Tuberculosis and Lung Disease. 4 (9): 796–806. PMID 10985648.
- ^ a b Fox W, Ellard GA, Mitchison DA (October 1999). "Studies on the treatment of tuberculosis undertaken by the British Medical Research Council tuberculosis units, 1946-1986, with relevant subsequent publications". The International Journal of Tuberculosis and Lung Disease. 3 (10 Suppl 2): S231–S279. PMID 10529902.
- ^ "Paser". RxList. Archived from the original on 13 September 2008. Retrieved 10 October 2008.
- ^ Smith NP, Ryan TJ, Sanderson KV, Sarkany I (March 1976). "Lichen scrofulosorum. A report of four cases". The British Journal of Dermatology. 94 (3): 319–325. doi:10.1111/j.1365-2133.1976.tb04391.x. PMID 1252363. S2CID 26281951.
- ^ Black JM, Sutherland IB (June 1961). "Two Incidents of Tuberculous Infection by Milk from Attested Herds". British Medical Journal. 1 (5241): 1732–1735. doi:10.1136/bmj.1.5241.1732. PMC 1954350. PMID 20789163.
- ^ "Paser: FDA-Approved Drugs". U.S. Food and Drug Administration (FDA). Archived from the original on 15 September 2020. Retrieved 3 April 2020.
- ^ Daniel F, Seksik P, Cacheux W, Jian R, Marteau P (May 2004). "Tolerance of 4-aminosalicylic acid enemas in patients with inflammatory bowel disease and 5-aminosalicylic-induced acute pancreatitis". Inflammatory Bowel Diseases. 10 (3): 258–260. doi:10.1097/00054725-200405000-00013. PMID 15290921.
- ^ Jiang YM, Mo XA, Du FQ, Fu X, Zhu XY, Gao HY, et al. (June 2006). "Effective treatment of manganese-induced occupational Parkinsonism with p-aminosalicylic acid: a case of 17-year follow-up study". Journal of Occupational and Environmental Medicine. 48 (6): 644–649. doi:10.1097/01.jom.0000204114.01893.3e. PMC 4180660. PMID 16766929.
- ^ Das KM, Eastwood MA, McManus JP, Sircus W (September 1973). "Adverse reactions during salicylazosulfapyridine therapy and the relation with drug metabolism and acetylator phenotype". The New England Journal of Medicine. 289 (10): 491–495. doi:10.1056/NEJM197309062891001. PMID 4146729.
- ^ Szeinberg A, Sheba C, Hirshorn N, Bodonyi E (July 1957). "Studies on erthrocytes in cases with past history of favism and drug-induced acute hemolytic anemia". Blood. 12 (7): 603–613. doi:10.1182/blood.V12.7.603.603. PMID 13436516.
- ^ Macgregor AG, Somner AR (November 1954). "The anti-thyroid action of para-aminosalicylic acid". Lancet. 267 (6845): 931–936. doi:10.1016/S0140-6736(54)92552-0. PMID 13213079.
- ^ Boman G (1974). "Serum concentration and half-life of rifampicin after simultaneous oral administration of aminosalicylic acid or isoniazid". European Journal of Clinical Pharmacology. 7 (3): 217–225. doi:10.1007/BF00560384. PMID 4854257. S2CID 24202603.
- ^ "Aminosalicylic acid (Paser) Use During Pregnancy". Drugs.com. 28 February 2020. Archived from the original on 15 September 2020. Retrieved 3 April 2020.
- ^ Vetuschi C, Ragno G, Mazzeo P (1988). "Determination of p-aminosalicylic acid and m-aminophenol by derivative UV-spectrophotometry". Journal of Pharmaceutical and Biomedical Analysis. 6 (4): 383–391. doi:10.1016/0731-7085(88)80003-7. PMID 16867404.
- ^ Zheng J, Rubin EJ, Bifani P, Mathys V, Lim V, Au M, et al. (August 2013). "para-Aminosalicylic acid is a prodrug targeting dihydrofolate reductase in Mycobacterium tuberculosis". The Journal of Biological Chemistry. 288 (32): 23447–23456. doi:10.1074/jbc.m113.475798. PMC 3789992. PMID 23779105.
- ^ Minato Y, Thiede JM, Kordus SL, McKlveen EJ, Turman BJ, Baughn AD (September 2015). "Mycobacterium tuberculosis folate metabolism and the mechanistic basis for para-aminosalicylic acid susceptibility and resistance". Antimicrobial Agents and Chemotherapy. 59 (9): 5097–5106. doi:10.1128/AAC.00647-15. PMC 4538520. PMID 26033719.
- ^ Zhao F, Wang XD, Erber LN, Luo M, Guo AZ, Yang SS, et al. (1 January 2014). "Binding pocket alterations in dihydrofolate synthase confer resistance to para-aminosalicylic acid in clinical isolates of Mycobacterium tuberculosis". Antimicrobial Agents and Chemotherapy. 58 (3): 1479–1487. doi:10.1128/aac.01775-13. PMC 3957869. PMID 24366731.
- ^ Lehmann J (December 1949). "The treatment of tuberculosis in Sweden with para-aminosalicylic acid; a review". Diseases of the Chest. 16 (6): 684–703, illust. doi:10.1378/chest.16.6.684. PMID 15396516.
- ^ Lehmann J (January 1946). "Para-aminosalicylic acid in the treatment of tuberculosis". Lancet. 1 (6384): 15–16. doi:10.1016/s0140-6736(46)91185-3. PMID 21008766.
Further reading
[edit]- "Para-aminosalicylic acid". Tuberculosis. 88 (2): 137–138. March 2008. doi:10.1016/S1472-9792(08)70019-2. PMC 1822430. PMID 18486053.
