Doravirine
 | |
| Clinical data | |
|---|---|
| Trade names | Pifeltro |
| Other names | MK-1439 |
| Routes of administration | Oral[1] |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.234.454 |
| Chemical and physical data | |
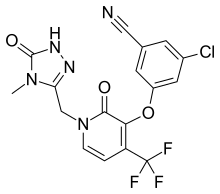
| Formula | C17H11ClF3N5O3 |
| Molar mass | 425.75 g/mol g·mol−1 |
| 3D model (JSmol) | |
| |
| |
Doravirine (MK-1439) is a non-nucleoside reverse transcriptase inhibitor developed by Merck & Co. for use in the treatment of HIV/AIDS. Doravirine demonstrated robust antiviral activity and good tolerability in a small clinical study of 7-day monotherapy reported at the 20th Conference on Retroviruses and Opportunistic Infections in March 2013. Doravirine appeared safe and generally well-tolerated with most adverse events being mild-to-moderate.[2][3] In August 2018, the FDA approved doravine under the product name Pifeltro, and in a combination tablet, doravirine/lamivudine/tenofovir disoproxil fumarate (Delstrigo).
medication side effects: It causes fatigue, headache, abnormal dreams, insomnia, nausea, diarrhea, abdominal pain, skin rash, increase in serum creatinine
References
- ^ Collins, Simon; Horn, Tim. "The Antiretroviral Pipeline" (PDF). Pipeline Report. p. 10. Archived from the original (PDF) on 11 March 2016. Retrieved 6 December 2015.
{{cite web}}: Unknown parameter|deadurl=ignored (|url-status=suggested) (help) - ^ Safety and Antiviral Activity of MK-1439, a Novel NNRTI, in Treatment-naïve HIV+ Patients. Gathe, Joseph et al. 20th Conference on Retroviruses and Opportunistic Infections. 3–6 March 2013. Abstract 100.
- ^ CROI 2013: MK-1439, a Novel HIV NNRTI, Shows Promise in Early Clinical Trials. Highleyman, Liz. HIVandHepatitis.com. 6 March 2013.
- ^ FDA Approves Merck’s DELSTRIGO™ (doravirine / lamivudine / tenofovir disoproxil fumarate), a Once-Daily Fixed-Dose Combination Tablet as a Complete Regimen and PIFELTRO™ (doravirine), an NNRTI, Both for the Treatment of HIV-1 in Appropriate Patients
