Angelman syndrome
| Angelman syndrome | |
|---|---|
| Other names | Angelman's syndrome[1][2] |
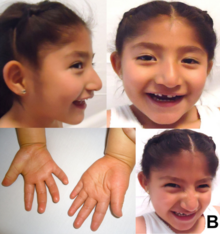 | |
| A five-year-old girl with Angelman syndrome. Features shown include telecanthus, bilateral epicanthic folds, small head, wide mouth, and an apparently happy demeanor; hands with tapered fingers, abnormal creases and broad thumbs. | |
| Pronunciation | |
| Specialty | Medical genetics |
| Symptoms | Delayed development, unusually happy, intellectual disability, limited to no functional speech, balance and movement problems, small head, seizures[6] |
| Usual onset | Noticeable by 6–12 months[6] |
| Causes | Genetic (new mutation)[6] |
| Diagnostic method | Based on symptoms, genetic testing[7] |
| Differential diagnosis | Cerebral palsy, autism, Rett syndrome, Prader–Willi syndrome[7][8] |
| Treatment | Supportive care[7] |
| Prognosis | Nearly normal life expectancy[6] |
| Frequency | 1 in 12,000 to 20,000 people[6] |
Angelman syndrome (AS) is a genetic disorder that mainly affects the nervous system.[6] Symptoms include a small head and a specific facial appearance, severe intellectual disability, developmental disability, limited to no functional speech, balance and movement problems, seizures, and sleep problems.[6] Children usually have a happy personality and have a particular interest in water.[6] The symptoms generally become noticeable by one year of age.[6]
Angelman syndrome is due to a lack of function of part of chromosome 15, typically due to a new mutation rather than one inherited.[6] Most often it is due to a deletion or mutation of the UBE3A gene on that chromosome.[6] Occasionally it is due to the inheritance of two copies of chromosome 15 from the father and none from the mother (paternal uniparental disomy).[6] As the father's versions are inactivated by a process known as genomic imprinting, no functional version of the gene remains.[6] Diagnosis is based on symptoms and possibly genetic testing.[7]
No cure is available.[7] Treatment is generally supportive in nature.[7] Anti-seizure medications are used in those with seizures.[7] Physical therapy and bracing may help with walking.[7] Those affected have a nearly normal life expectancy.[6]
AS affects 1 in 12,000 to 20,000 people.[6] Males and females are affected with equal frequency.[7] It is named after British pediatrician Harry Angelman, who first described the syndrome in 1965.[7][9] An older term, happy puppet syndrome, is generally considered pejorative.[10] Prader–Willi syndrome is a separate condition, caused by a similar loss of the father's chromosome 15.[11]
Signs and symptoms
[edit]Signs and symptoms of Angelman syndrome and their relative frequency in affected individuals are:[12]
Consistent (100%)
[edit]- Developmental delay, functionally severe
- Speech impairment, no or minimal use of words; receptive and non-verbal communication skills higher than verbal ones
- Movement or balance disorder, usually ataxia of gait and/or tremulous movement of limbs
- Behavioral characteristics of the following types: any combination of atypical frequent laughter/smiling; atypically happy demeanor; easily excitable personality, often with hand flapping movements; hypermotoric behavior; short attention span
Frequent (more than 80%)
[edit]- Delayed, disproportionate growth in head circumference, usually resulting in microcephaly (absolute or relative) by age 2
- Seizures, onset usually less than 3 years of age
- Abnormal EEG, characteristic pattern with large amplitude slow-spike waves
Associated (20–80%)
[edit]- Strabismus
- Hypopigmented skin and eyes
- Tongue thrusting; suck/swallowing disorders
- Hyperactive tendon reflexes
- Feeding problems during infancy
- Uplifted, flexed arms during walking
- Prominent mandible
- Increased sensitivity to heat
- Wide mouth, wide-spaced teeth
- Sleep disturbance
- Frequent drooling, protruding tongue
- Attraction to/fascination with water
- Excessive chewing/mouthing behaviors
- Flat back of head
- Smooth palms
- Gastroesophageal reflux disease (GERD)[13]
- Constipation[13]
Cause
[edit]Angelman syndrome is caused by the lack of expression of a gene known as UBE3A during development.[14] This gene is located within a region of chromosome 15 known as 15q11-q13 and is part of the ubiquitin pathway. In fact, UBE3A codes for a very selective E6-AP ubiquitin ligase for which MAPK1, PRMT5, CDK1, CDK4, β-catenin, and UBXD8 have been identified as ubiquitination targets[15]
Typically, a fetus inherits a maternal copy of UBE3A and a paternal copy of UBE3A. In certain areas of the developing brain, the paternal copy of UBE3A is inactivated through a process known as imprinting and the fetus relies on the functioning maternal copy of UBE3A in order to develop normally. In an individual with AS, however, the maternal UBE3A gene is absent or not functioning normally. This can be due to genetic errors such as the deletion or mutation of a segment of chromosome 15, uniparental disomy, or translocation. While Angelman syndrome can be caused by a single mutation in the UBE3A gene, the most common genetic defect leading to Angelman syndrome is a 5- to 7-Mb (megabase) maternal deletion in chromosomal region 15q11.2-q13.[16]
Specifically, the paternal copy of UBE3A is known to be imprinted within the hippocampus, cortex, thalamus, olfactory bulb, and cerebellum. Therefore, in these areas of the brain, a functioning maternal copy of UBE3A is essential for proper development.[17]
Region 15q11-13 is implicated in both Angelman syndrome and Prader–Willi syndrome (PWS). While AS results from mutation, loss or abnormal imprinting involving the UBE3A gene within this region on the maternal chromosome,[16] loss of a different cluster of genes within the same region on the paternal chromosome causes PWS.[18]
The methylation test that is performed for Angelman syndrome looks for methylation on the gene's neighbor SNRPN, which is silenced by methylation on the maternal copy of the gene.[19]
Neurophysiology
[edit]The electroencephalogram (EEG) in AS is usually abnormal, more so than clinically expected.[20] This EEG facilitates the differential diagnosis of AS, but is not pathognomonic.[20][21] Three distinct interictal patterns are seen in these patients.[22] The most common pattern is a very large amplitude 2–3 Hz rhythm most prominent in prefrontal leads. Next most common is a symmetrical 4–6 Hz high voltage rhythm. The third pattern, 3–6 Hz activity punctuated by spikes and sharp waves in occipital leads, is associated with eye closure. Paroxysms of laughter have no relation to the EEG, ruling out this feature as a gelastic phenomenon.[20]
EEG anomalies may be used as a quantitative biomarkers to "chart progression of AS and as clinical outcome measures".[23] Slow delta activity (~3 Hz) is greatly increased in AS relative to typically developing children, yet more pronounced in children with partial 15q deletions as opposed to those with etiologies principally affecting UBE3A.[24] Theta activity (~5 Hz) is much greater in children with partial 15q deletions.[24] Thus, delta activity appears to be chiefly reflective of UBE3A dysfunction with some modulation from other 15q genes, whereas theta activity may be an electrophysiological readout of genes beyond UBE3A such as GABRA5, GABRB3, and GABRG3.[24]
Diagnosis
[edit]The diagnosis of Angelman syndrome is based on:[citation needed]
- A history of delayed motor milestones and then later a delay in general development, especially of speech
- Unusual movements including fine tremors, jerky limb movements, hand flapping and a wide-based, stiff-legged gait.
- Characteristic facial appearance (but not in all cases).
- A history of epilepsy and an abnormal EEG tracing.
- A happy disposition with frequent laughter
- A deletion or inactivity on chromosome 15 by array comparative genomic hybridization (aCGH) or by BACs-on-Beads technology.
Diagnostic criteria for the disorder were initially established in 1995 in collaboration with the Angelman syndrome Foundation (US);[25] these criteria underwent revision in 2005.[26]
Seizures are a consequence, as is excessive laughter,[27] which is a major hindrance to early diagnosis.
Differential diagnosis
[edit]Other conditions that can appear similar include:[7][8]
- Autism spectrum
- Cerebral palsy
- Rett syndrome
- Mowat–Wilson syndrome
- Adenylosuccinate lyase deficiency
- Pitt–Hopkins syndrome
- Phelan–McDermid syndrome
- Prader–Willi syndrome
Treatment
[edit]
There is currently no approved cure available. The epilepsy can be controlled by the use of one or more types of anticonvulsant medications. However, there are difficulties in ascertaining the levels and types of anticonvulsant medications needed to establish control, because people with AS often have multiple types of seizures.[28] Many families use melatonin[13] to promote sleep in a condition which often affects sleep patterns. Mild laxatives are also used frequently to encourage regular bowel movements. Additionally, among a cohort of 163 individuals with AS, ranitidine was shown to be the most frequently prescribed medication for treating gastroesophageal reflux disease (GERD).[13] Early intervention with physiotherapy is sometimes used to encourage joint mobility and prevent stiffening of the joints.[citation needed]
Occupational therapists can contribute to the development and augmentation of non-verbal communication skills by addressing the foundational skills such as finger isolation, motor planning, hand-eye coordination, spatial awareness, and refining gestures.[29] This is important because individuals with Angelman Syndrome who already possess some form of non-verbal communication have a much harder time adapting to changes in a new or existing AAC device because they can communicate their needs much faster nonverbally.[29]
Occupational therapists can assist individuals with Angelman syndrome with many other skills as well.[30] Many individuals with Angelman syndrome also have difficulty processing sensory information and responding appropriately to sensory stimuli.[31] Occupational therapists can work together with these individuals to improve their visual perceptual skills and increase their sensory awareness.[31]
Expressive verbal communication is limited by AS, but many people with the disorder are able to learn non-verbal communication skills to express their needs. Deictic gesturing (i.e, pointing to an object) is the most commonly used form of non-symbolic communication in AS, followed by physically manipulating others (such as moving a caregiver's hand to a specific object or guiding a person to a new location) and non-speech vocalizations.[30] Some are able to use symbolic communication such as signing, though the prevalence of this ability is related to both genetic etiology and epilepsy status, with non-deletion etiologies without epilepsy showing the highest prevalence of symbolic communication skills.[32] People with AS tend to have much higher receptive language abilities than expressive; recent studies have shown that patients with AS have typical auditory brain region responses to speech but atypical memory responses, suggesting that word meaning recall is delayed or processed differently in AS.[33] This may be caused by the altered cortical morphology seen in AS [34] in the precuneus, a region of the brain involved in self-reflection and memory. Similarly, both adults and children with AS show a delay in processing speed in speech processing,[35] and this should be accounted for during communication.
An experimental treatment, currently in clinical development by Roche, targets the GABAA α5 receptor. Alogabat is a small molecule that acts as a positive allosteric modulator (PAMs) of the receptor. It does not involve replacing or activating the UBE3A gene, instead it is a mechanism aimed at improving various symptoms of AS such as learning, sleep, and seizure control.[36][37][38][39]
Prognosis
[edit]The severity of the symptoms associated with Angelman syndrome varies significantly across the population of those affected. Some speech and a greater degree of self-care are possible among the least profoundly affected. Walking and the use of simple sign language may be beyond the reach of the more profoundly affected. Early and continued participation in physical, occupational (related to the development of fine-motor control skills), and communication (speech) therapies are believed to significantly improve the prognosis (in the areas of cognition and communication) of individuals affected by AS. Further, the specific genetic mechanism underlying the condition is thought to correlate to the general prognosis of the affected person. On one end of the spectrum, a mutation to the UBE3A gene is thought to correlate to the least affected, whereas larger deletions on chromosome 15 are thought to correspond to the most affected.[citation needed]
The clinical features of Angelman syndrome alter with age. As adulthood approaches, hyperactivity and poor sleep patterns improve. The seizures decrease in frequency and often cease altogether and the EEG abnormalities are less obvious. Medication is typically advisable to those with seizure disorders. Often overlooked is the contribution of the poor sleep patterns to the frequency and/or severity of the seizures. Medication may be worthwhile to help deal with this issue and improve the prognosis with respect to seizures and sleep. Also noteworthy are the reports that the frequency and severity of seizures temporarily escalate in pubescent Angelman syndrome girls, but do not seem to affect long-term health.[citation needed]The facial features remain recognizable with age, but many adults with AS look remarkably youthful for their age.[citation needed]
Puberty and menstruation begin at around the average age. Sexual development is thought to be unaffected, as evidenced by a single reported case of a woman with Angelman syndrome conceiving a female child who also had Angelman syndrome.[40]
The majority of those with AS achieve continence by day and some by night. Angelman syndrome is not a degenerative syndrome, and thus people with AS may improve their living skills with support.[citation needed]
Dressing skills are variable and usually limited to items of clothing without buttons or zippers. Most adults can eat with a knife or spoon and fork, and can learn to perform simple household tasks. Particular problems which have arisen in adults are a tendency to obesity (more in females), and worsening of scoliosis[41] if it is present. The affectionate nature may also persist into adult life where it can pose a problem socially, but this problem is not insurmountable. People with Angelman syndrome appear to have a reduced but near-normal life expectancy, dying on average 10 to 15 years earlier than the general population.[42]
Epidemiology
[edit]Though the prevalence of Angelman syndrome is not precisely known, there are some estimates. The best data available are from studies of school age children, ages 6–13 years, living in Sweden and from Denmark where the diagnosis of AS children in medical clinics was compared to an 8-year period of about 45,000 births. The Swedish study showed an AS prevalence of about 1/20,000[43] and the Danish study showed a minimum AS prevalence of about 1/10,000.[44]
History
[edit]
Harry Angelman, a pediatrician working in Warrington, England, first reported three children with this condition in 1965.[9] Angelman later described his choice of the title "Puppet Children" to describe these cases as being related to an oil painting he had seen while vacationing in Italy:
The history of medicine is full of interesting stories about the discovery of illnesses. The saga of Angelman's syndrome is one such story. It was purely by chance that nearly thirty years ago (e.g. [sic], circa 1964) three handicapped children were admitted at various times to my children's ward in England. They had a variety of disabilities and although at first sight they seemed to be suffering from different conditions I felt that there was a common cause for their illness. The diagnosis was purely a clinical one because in spite of technical investigations which today are more refined I was unable to establish scientific proof that the three children all had the same handicap. In view of this I hesitated to write about them in the medical journals. However, when on holiday in Italy I happened to see an oil painting in the Castelvecchio Museum in Verona called ... a Boy with a Puppet. The boy's laughing face and the fact that my patients exhibited jerky movements gave me the idea of writing an article about the three children with a title of Puppet Children. It was not a name that pleased all parents but it served as a means of combining the three little patients into a single group. Later the name was changed to Angelman syndrome. This article was published in 1965 and after some initial interest lay almost forgotten until the early eighties.
— Angelman quoted by Charles Williams[46]
Case reports from the United States first began appearing in the medical literature in the early 1980s.[47][48] In 1987, it was first noted that around half of the children with AS have a small piece of chromosome 15 missing (chromosome 15q partial deletion).[49]
Society and culture
[edit]Many poems in Richard Price's poetry collections Hand Held (1997), Lucky Day (2005), and Small World (2012) reflect on his daughter, who has Angelman syndrome. In October 2007, actor Colin Farrell publicly announced that his son has Angelman syndrome.[50] In the 2011 Philippine drama series Budoy, the titular character and main protagonist Budoy Maniego (played by Filipino actor Gerald Anderson) is diagnosed with Angelman syndrome.
See also
[edit]References
[edit]- ^ a b "Angelman syndrome". Oxford English Dictionary. Archived from the original on June 17, 2020 – via Lexico.com.
- ^ a b c "Angelman syndrome". Merriam-Webster Medical Dictionary. Merriam-Webster. Retrieved April 15, 2022 – via Merriam-Webster.com.
- ^ "Angelman syndrome". McGraw-Hill Dictionary of Scientific & Technical Terms. The McGraw-Hill Companies, Inc. 2003. Retrieved April 15, 2022 – via thefreedictionary.com.
- ^ a b "Angelman syndrome". American Heritage Medical Dictionary (2007 ed.). Houghton Mifflin Company. 2004. Retrieved April 15, 2022 – via thefreedictionary.com.
- ^ Urraca N, Reiter L (2013). "Angelman syndrome". Comprehensive Developmental Neuroscience: Neural Circuit Development and Function in the Heathy and Diseased Brain. Elsevier Inc. Chapter 32. ISBN 9780128063415. Retrieved April 15, 2022 – via Google Books.
- ^ a b c d e f g h i j k l m n o "Angelman syndrome". ghr.nlm.nih.gov. Genetics Home Reference, US National Institutes of Health. May 2015. Archived from the original on 27 August 2016. Retrieved 28 April 2017.
- ^ a b c d e f g h i j k "Angelman Syndrome". rarediseases.org. National Organization for Rare Disorders (NORD). 2015. Archived from the original on 13 November 2016. Retrieved 28 April 2017.
- ^ a b "Common Misdiagnoses". Foundation For Angelman Syndrome Therapeutics. 2 August 2019. Retrieved 10 March 2024.
- ^ a b Angelman H (1965). "'Puppet' Children: A report of three cases". Developmental Medicine & Child Neurology. 7 (6): 681–688. doi:10.1111/j.1469-8749.1965.tb07844.x. S2CID 53730099.
- ^ Wilson GN, Cooley WC (2000). Preventive Management of Children with Congenital Anomalies and Syndromes. Cambridge University Press. p. 193. ISBN 9780521776738. Archived from the original on 2017-11-05.
- ^ Kumar V, Abbas AK, Aster JC (2013). "Genetic and Pediatric Diseases". Robbins Basic Pathology. Elsevier Health Sciences. p. 244. ISBN 978-1437717815. Archived from the original on 2017-11-05. Retrieved 2022-04-15 – via Google Books.
- ^ "Facts about Angelman syndrome" (PDF). US: Angelman Syndrome Foundation. Archived from the original (PDF) on 2013-05-27. Retrieved 2012-09-29.
- ^ a b c d Khan N (8 April 2019). "Healthcare burden among individuals with Angelman syndrome: Findings from the Angelman Syndrome Natural History Study". Molecular Genetics & Genomic Medicine. 7 (7): 3. doi:10.1002/mgg3.734. PMC 6625091. PMID 31090212.
- ^ Weeber E, Levenson J, Sweatt J (2002). "Molecular genetics of human cognition". Molecular Interventions. 2 (6): 376–91, 339. doi:10.1124/mi.2.6.376. PMID 14993414.
- ^ Wang, Yiyang, et al. (2017). "Identifying the ubiquitination targets of E6AP by orthogonal ubiquitin transfer". Nature Communications. 8 (1): 2232. Bibcode:2017NatCo...8.2232W. doi:10.1038/s41467-017-01974-7. PMC 5738348. PMID 29263404.
- ^ a b Dagli AI, Mathews J, Williams CA (22 April 2021). Adam MP, Everman DB, Mirzaa GM, Pagon RA, Wallace SE (eds.). "Angelman Syndrome". GeneReviews. Seattle: University of Washington, Seattle. PMID 20301323. Retrieved 28 August 2022.
- ^ Mabb AM, Judson MC, Zylka MJ, Philpot BD (May 2011). "Angelman Syndrome: Insights into Genomic Imprinting and Neurodevelopmental Phenotypes". Trends Neurosci. 34 (6): 293–303. doi:10.1016/j.tins.2011.04.001. PMC 3116240. PMID 21592595.
- ^ Cassidy SB, Dykens E (2000). "Prader–Willi and Angelman syndromes: sister imprinted disorders". Am J Med Genet. 97 (2): 136–146. doi:10.1002/1096-8628(200022)97:2<136::aid-ajmg5>3.0.co;2-v. PMID 11180221. S2CID 20832857.
- ^ White HE, Durston VJ, Harvey JF, Cross NC (2006). "Quantitative analysis of SNRPN(correction of SRNPN) gene methylation by pyrosequencing as a diagnostic test for Prader–Willi syndrome and Angelman syndrome". Clin. Chem. 52 (6): 1005–13. doi:10.1373/clinchem.2005.065086. PMID 16574761.
- ^ a b c Williams C (2005) "Neurological aspects of the Angelman syndrome" Brain & Development 27: 88–94
- ^ Laan LA, Vein AA (2005). "Angelman syndrome: is there a characteristic EEG?". Brain and Development. 27 (2): 80–87. doi:10.1016/j.braindev.2003.09.013. ISSN 0387-7604. PMID 15668045. S2CID 5912.
- ^ Dan, B., Angelman syndrome: Current understanding and research prospects. Epilepsia, 2009. 50(11): p. 2331–2339.
- ^ Sidorov MS, Deck GM, Dolatshahi M, Thibert RL, Bird LM, Chu CJ, Philpot BD (2017-05-08). "Delta rhythmicity is a reliable EEG biomarker in Angelman syndrome: a parallel mouse and human analysis". Journal of Neurodevelopmental Disorders. 9: 17. doi:10.1186/s11689-017-9195-8. ISSN 1866-1955. PMC 5422949. PMID 28503211.
- ^ a b c Frohlich, Joel, Meghan Miller, Lynne M. Bird, Pilar Garces, Hannah Purtell, Marius C. Hoener, Benjamin D. Philpot et al. "Electrophysiological phenotype in Angelman syndrome differs between genotypes." Biological Psychiatry (2019).
- ^ Williams CA, Angelman H, Clayton-Smith J, et al. (1995). "Angelman syndrome: consensus for diagnostic criteria. Angelman syndrome Foundation". Am. J. Med. Genet. 56 (2): 237–8. doi:10.1002/ajmg.1320560224. PMID 7625452.
- ^ Williams CA, Beaudet AL, Clayton-Smith J, et al. (2006). "Angelman syndrome 2005: updated consensus for diagnostic criteria". Am. J. Med. Genet. A. 140 (5): 413–8. doi:10.1002/ajmg.a.31074. PMID 16470747. S2CID 2449346.
- ^ Buntinx IM, Hennekam RC, Brouwer OF, et al. (March 1995). "Clinical profile of Angelman syndrome at different ages". American Journal of Medical Genetics. 56 (2): 176–83. doi:10.1002/ajmg.1320560213. PMID 7625442.
- ^ Leung HT, Ring H (January 2013). "Epilepsy in four genetically determined syndromes of intellectual disability". Journal of Intellectual Disability Research. 57 (1): 3–20. doi:10.1111/j.1365-2788.2011.01505.x. PMID 22142420.
- ^ a b Pearson E, Wilde L, Heald M, Royston R, Oliver C (November 2019). "Communication in Angelman syndrome: a scoping review". Developmental Medicine & Child Neurology. 61 (11): 1266–1274. doi:10.1111/dmcn.14257. ISSN 0012-1622. PMID 31074506. S2CID 149445749.
- ^ a b Walz NC, Baranek GT (2006-07-01). "Sensory Processing Patterns in Persons With Angelman Syndrome". American Journal of Occupational Therapy. 60 (4): 472–479. doi:10.5014/ajot.60.4.472. ISSN 0272-9490. PMID 16915878.
- ^ a b Heald M, Adams D, Oliver C (2020). "Profiles of atypical sensory processing in Angelman, Cornelia de Lange and Fragile X syndromes". Journal of Intellectual Disability Research. 64 (2): 117–130. doi:10.1111/jir.12702. hdl:10072/396330. ISSN 1365-2788. PMID 31828905. S2CID 209329797.
- ^ >Didden R, Sigafoos J, Korzilius H, Baas A, Lancioni GE, O'Reilly MF, Curfs LM (2009). "Form and Function of Communicative Behaviors in Individuals with Angelman Syndrome". Journal of Applied Research in Intellectual Disabilities. 22 (6): 526–537. doi:10.1111/j.1468-3148.2009.00520.x. hdl:2066/77094.
- ^ Key AP, Roth S, Jones D, Hunt-Hawkins H (2023). "Typical and atypical neural mechanisms support spoken word processing in Angelman syndrome". Brain and Language. 236. doi:10.1016/j.bandl.2022.105215. PMC 9839587. PMID 36502770.
- ^ Du X, Wei L, Yang B, Long S, Wang J, Sun A, Jiang Y, Qiao Z, Wang H, Wang Y (2023). "Cortical and subcortical morphological alteration in Angelman syndrome". Journal of Neurodevelopmental Disorders. 15 (1): 7. doi:10.1186/s11689-022-09469-3. ISSN 1866-1955. PMC 9930225. PMID 36788499.
- ^ Key AP, Roth S, Venker C (11 October 2022). "Spoken language comprehension in children and adults with Angelman Syndrome". Journal of Communication Disorders. 100. doi:10.1016/j.jcomdis.2022.106272. PMC 9994640. PMID 36244082.
- ^ WO2018104419A1, Buettelmann, Bernd; Cecere, Giuseppe & FASCHING, Bernhard et al., "New isoxazolyl ether derivatives as gaba a alpha5 pam", issued 2018-06-14
- ^ "Alogabat". www.glpbio.com (in German). Retrieved 2024-09-20.
- ^ "Recruitment Open for the Aldebaran Study of Alogabat – Angelman Syndrome Foundation". www.angelman.org. Retrieved 2024-09-20.
- ^ MS MW (2023-06-28). "Roche stopping testing of rugonersen to treat Angelman syndrome | Children in Phase 1 trial may move into monthslong extension study | Angelman Syndrome News". angelmansyndromenews.com. Retrieved 2024-09-20.
- ^ Lossie A, Driscoll D (1999). "Transmission of Angelman syndrome by an affected mother". Genet Med. 1 (6): 262–6. doi:10.1097/00125817-199909000-00004. PMID 11258627.
- ^ Laan LA, den Boer AT, Hennekam RC, Renier WO, Brouwer OF (1996). "Angelman syndrome in adulthood". Am. J. Med. Genet. 66 (3): 356–60. doi:10.1002/(SICI)1096-8628(19961218)66:3<356::AID-AJMG21>3.0.CO;2-K. hdl:2066/22929. PMID 9072912.
- ^ Coppus AM (2013). "People with intellectual disability: What do we know about adulthood and life expectancy?". Developmental Disabilities Research Reviews. 18 (1): 6–16. doi:10.1002/ddrr.1123. PMID 23949824.
- ^ Steffenburg S, Gillberg CL, Steffenburg U, Kyllerman M (1996). "Autism in Angelman syndrome: a population-based study". Pediatr. Neurol. 14 (2): 131–6. doi:10.1016/0887-8994(96)00011-2. PMID 8703225.
- ^ Petersen MB, Brøndum-Nielsen K, Hansen LK, Wulff K (1995). "Clinical, cytogenetic, and molecular diagnosis of Angelman syndrome: estimated prevalence rate in a Danish county; the disorder predominantly affects Anglo-Saxons". Am. J. Med. Genet. 60 (3): 261–2. doi:10.1002/ajmg.1320600317. PMID 7573182.
- ^ Galassi FM, Armocida E, Rühli FJ (September 2016). "Angelman Syndrome in the Portrait of a Child With a Drawing by Giovanni F. Caroto" (PDF). JAMA Pediatrics. 170 (9): 831. doi:10.1001/jamapediatrics.2016.0581. PMID 27380555.
- ^ Williams C. "Harry Angelman and the History of AS". Stay informed. US: Angelman Syndrome Foundation. Archived from the original on 2011-02-06. Retrieved 2011-07-01.
- ^ Dooley JM, Berg JM, Pakula Z, MacGregor DL. (1981). "The puppet-like syndrome of Angelman". Am J Dis Child. 135 (7): 621–4. doi:10.1001/archpedi.1981.02130310027010. PMID 7246489.
- ^ Williams CA, Frias JL (1982). "The Angelman ("happy puppet") syndrome". Am J Med Genet. 11 (4): 453–60. doi:10.1002/ajmg.1320110411. PMID 7091188.
- ^ Magenis RE, Brown MG, Lacy DA, Budden S, LaFranchi S. (1987). "Is Angelman syndrome an alternate result of del(15)(q11q13)?". Am J Med Genet. 28 (4): 829–38. doi:10.1002/ajmg.1320280407. PMID 3688021.
- ^ "Superstar Colin tells of 'blessed' life with special needs child". Irish Independent. 15 October 2007. Archived from the original on 17 October 2007. Retrieved 15 October 2007.
