Lithium orotate
 | |
 | |
| Clinical data | |
|---|---|
| AHFS/Drugs.com | Monograph |
| Routes of administration | Oral |
| Legal status | |
| Legal status |
|
| Identifiers | |
| CAS Number | |
| PubChem CID | |
| ChemSpider | |
| UNII | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.023.711 |
| Chemical and physical data | |
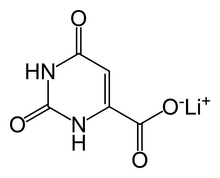
| Formula | C5H3LiN2O4 |
| Molar mass | 162.03 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Lithium orotate (C5H3LiN2O4) is a salt of orotic acid and lithium. It is available as the monohydrate, LiC5H3N2O4·H2O.[1] In this compound, lithium is non-covalently bound to an orotate ion, rather than to a carbonate or other ion, and like other salts, dissociates in solution to produce free lithium ions. It is marketed as a dietary supplement, though it has been researched minimally between 1973–1986 to treat certain medical conditions, such as alcoholism[2] and Alzheimer's disease.
While lithium orotate is capable of providing lithium to the body,[3] like lithium carbonate and other lithium salts, there are no systematic reviews supporting the efficacy of lithium orotate and it is not approved by the U.S. Food and Drug Administration (FDA) for the treatment of any medical condition.
Effectiveness[edit]
In 1973, Hans Nieper reported that lithium orotate contained 3.83 mg of elemental lithium per 100 mg and lithium carbonate contained 18.8 mg of elemental lithium per 100 mg.[4] Nieper went on to claim that lithium did not dissolve from the orotate carrier until it passed through the blood–brain barrier; however, a 1976 study documented that lithium concentrations within the brains of rats were not statistically different between equivalent dosages of lithium from lithium orotate, lithium carbonate, or lithium chloride.[5] However, another study in 1978 study showed that eight hours after intraperitoneal injections brain lithium concentrations of rats were significantly greater after lithium orotate than after lithium carbonate. While little serum lithium remained at 24 h after injection of 2·0 m equiv kg−1 lithium carbonate, two‐thirds of the 2 h serum lithium concentration was present 24 h after lithium orotate. Furthermore, the 24 h brain concentration of lithium after lithium orotate was approximately three times greater than that after lithium carbonate. These data suggest the possibility that lower doses of lithium orotate than lithium carbonate may achieve therapeutic brain lithium concentrations and relatively stable serum concentrations.[6] A year later, Smith and Schou repeated the experiment at a higher dose (2 mM Li+) and found that the higher concentrations in the brain could be possibly accounted for by decreased renal function in rats treated with lithium orotate.[7][8] The proponents of lithium orotate have since criticized the results by citing the fact that the dose of lithium orotate used in the study was in the toxic range. In 2022, Pacholko redid the experiment and showed lithium orotate to have a safer kidney profile than lithium carbonate, it also showed that both had an increased TSH only in females, but the increase was lower in the orotate group.[9]
The pharmacokinetics of lithium orotate in human brains is poorly documented, and there is no known mechanism by which orotate ions could alter the pharmacokinetics of dissociated lithium ions, however, lithium intake appears to be effective even at low doses, and this may account for lithium orotate's claimed effectiveness.[10][11][12]
The reason why lithium orotate is poorly studied as a medication compared to lithium carbonate is concerns raised in 1979 regarding the potential amplified renal toxicity of lithium orotate in comparison to lithium carbonate.[13] These concerns were likely based on the results of the use of excessively high concentrations of lithium orotate in the studies. As a result of these concerns, the clinical application and research of lithium orotate were halted for decades since the 1980s.[13] Still, interest in lithium orotate has been rekindled, and research into its use as medication was resumed in the 2010s.[13] The renewed interest is largely due to its purported ability to achieve higher serum and brain lithium-ion (Li+) concentrations than those observed from equivalent doses of lithium carbonate, a claim first made in 1978.[13]
Safety[edit]
Preliminary studies seem to indicate that lithium orotate is safe if taken at lower dosages; a 6 month alcoholism cessation study led to only minor adverse effects in 8 out of 42 patients.[2] However, the lack of safety studies and the OTC status raises concerns.[8] Attention to it was specially raised in medical literature after a case report of an 18 year old woman with mild, acute lithium toxicity after taking an overdose (2.16 grams) of a lithium orotate supplement. She was discharged after treatment.[14][15][16] Lithium blood levels 90 minutes after ingestion reached 0.31 mEq/L, and an hour later after treatment, 0.40 mEq/L, levels below the serum toxicity level of 1.5 milliequivalents per liter (mEq/L).[15][17]
Orotic acid can be mutagenic in very high doses of 50 mg/kg in mammalian somatic cells.[18] It is also mutagenic for bacteria and yeast.[19] Although lithium orotate was not shown to be genotoxic,[20] short and long term studies must be performed to guarantee public safety.[8]
See also[edit]
References[edit]
- ^ Bach I, Kumberger O, Schmidbaur H (1990). "Orotate complexes. Synthesis and crystal structure of lithium orotate( - I) monohydrate and magnesium bis[ orotate( - I)] octahydrate". Chemische Berichte. 123 (12): 2267–2271. doi:10.1002/cber.19901231207.
- ^ a b Sartori HE (1986). "Lithium orotate in the treatment of alcoholism and related conditions". Alcohol. 3 (2): 97–100. doi:10.1016/0741-8329(86)90018-2. PMID 3718672.
- ^ Pacholko AG, Bekar LK (2021). "Lithium orotate: A superior option for lithium therapy?". Brain and Behavior. 11 (4): e2262. doi:10.1002/brb3.2262. PMC 8413749. PMID 34196467.
- ^ Nieper HA (1973). "The clinical applications of lithium orotate. A two years study". Agressologie. 14 (6): 407–11. PMID 4607169.
- ^ Smith DF (April 1976). "Lithium orotate, carbonate and chloride: pharmacokinetics, polyuria in rats". British Journal of Pharmacology. 56 (4): 399–402. doi:10.1111/j.1476-5381.1976.tb07449.x. PMC 1666891. PMID 1260219.
- ^ Kling MA, Manowitz P, Pollack IW (June 1978). "Rat brain and serum lithium concentrations after acute injections of lithium carbonate and orotate". The Journal of Pharmacy and Pharmacology. 30 (6): 368–370. doi:10.1111/j.2042-7158.1978.tb13258.x. PMID 26768. S2CID 28459663.
- ^ Smith DF, Schou M (March 1979). "Kidney function and lithium concentrations of rats given an injection of lithium orotate or lithium carbonate". The Journal of Pharmacy and Pharmacology. 31 (3): 161–163. doi:10.1111/j.2042-7158.1979.tb13461.x. PMID 34690. S2CID 8691638.
- ^ a b c Pacholko AG, Bekar LK (August 2021). "Lithium orotate: A superior option for lithium therapy?". Brain and Behavior. 11 (8): e2262. doi:10.1002/brb3.2262. PMC 8413749. PMID 34196467. S2CID 235697871.
- ^ Pacholko AG, Bekar LK (2022-05-01). "Lithium orotate is more potent, effective, and less toxic than lithium carbonate in a mouse model of mania". bioRxiv 10.1101/2022.05.01.490227.
- ^ Alevizos B, Alevizos E, Leonardou A, Zervas I (2012). "Low dosage lithium augmentation in venlafaxine resistant depression: an open-label study". Psychiatrike = Psychiatriki. 23 (2): 143–148. PMID 22796912.
- ^ Nunes MA, Viel TA, Buck HS (January 2013). "Microdose lithium treatment stabilized cognitive impairment in patients with Alzheimer's disease". Current Alzheimer Research. 10 (1): 104–107. doi:10.2174/156720513804871354. PMID 22746245.
- ^ Berger GE, Wood SJ, Ross M, Hamer CA, Wellard RM, Pell G, et al. (2012). "Neuroprotective effects of low-dose lithium in individuals at ultra-high risk for psychosis. A longitudinal MRI/MRS study". Current Pharmaceutical Design. 18 (4): 570–575. doi:10.2174/138161212799316163. PMID 22239590.
- ^ a b c d Pacholko AG, Bekar LK (August 2021). "Lithium orotate: A superior option for lithium therapy?". Brain Behav. 11 (8): e2262. doi:10.1002/brb3.2262. PMC 8413749. PMID 34196467.
- ^ Balon R (February 2013). "Possible dangers of a "nutritional supplement" lithium orotate". Annals of Clinical Psychiatry. 25 (1): 71. PMID 23376874.
- ^ a b Pauzé DK, Brooks DE (June 2007). "Lithium toxicity from an Internet dietary supplement". Journal of Medical Toxicology. 3 (2): 61–62. doi:10.1007/bf03160910. PMC 3550087. PMID 18072162.
- ^ Kwan D, Beyene J, Shah PS (1 November 2009). "Adverse Consequences of Internet Purchase of Pharmacologic Agents or Dietary Supplements". Journal of Pharmacy Technology. 25 (6): 355–360. doi:10.1177/875512250902500602. S2CID 70625804.
- ^ Hedya SA, Avula A, Swoboda HD (2023). "Lithium Toxicity". StatPearls. Treasure Island (FL): StatPearls Publishing. PMID 29763168. Retrieved 2023-04-13.
- ^ "Vitamin B13 cannot be proven safe, says EFSA". nutraingredients.com. Retrieved 2018-10-08.
- ^ "Material Safety Data Sheet - Orotic Acid, anhydrous MSDS". ScienceLab.com. 2013-05-21. Archived from the original on 2018-01-20.
- ^ Murbach TS, Glávits R, Endres JR, Hirka G, Vértesi A, Béres E, Szakonyiné IP (August 2021). "A toxicological evaluation of lithium orotate". Regulatory Toxicology and Pharmacology. 124: 104973. doi:10.1016/j.yrtph.2021.104973. PMID 34146638. S2CID 235485928.
External links[edit]
- "Ask the Mental Health Expert". Ron Pies, M.D., clinical professor of psychiatry at Tufts University. December 2002. Archived from the original on 2018-07-24.
