Avanafil
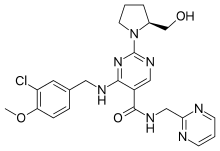 | |
 Avanafil is a PDE5 inhibitor | |
| Clinical data | |
|---|---|
| Trade names | Stendra |
| AHFS/Drugs.com | Monograph |
| MedlinePlus | a614010 |
| License data | |
| Routes of administration | By mouth |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| ChEMBL | |
| PDB ligand | |
| CompTox Dashboard (EPA) | |
| Chemical and physical data | |
| Formula | C23H26ClN7O3 |
| Molar mass | 483.96 g·mol−1 |
| 3D model (JSmol) | |
| |
| |
| | |
Avanafil is a PDE5 inhibitor approved for erectile dysfunction by the FDA on April 27, 2012[2] and by EMA on June 21, 2013.[3] Avanafil is sold under the brand names Stendra and Spedra. It was invented at Mitsubishi Tanabe Pharma, formerly known as Tanabe Seiyaku Co.,[4] and licensed to Vivus Inc., which partnered with Menarini Group to commercialise Spedra in over forty European countries, Australia, and New Zealand.[5] Metuchen Pharmaceuticals obtained exclusive rights within the United States.[6]
Avanafil acts by inhibiting a specific phosphodiesterase type 5 enzyme found in various body tissues, primarily in the corpus cavernosum penis.[7] Other similar drugs are sildenafil, tadalafil and vardenafil. The advantage of avanafil is that it has very fast onset of action compared with other PDE5 inhibitors. It is absorbed quickly, reaching a maximum serum concentration in about thirty to forty-five minutes.[8] About two-thirds of the participants were able to engage in sexual activity within fifteen minutes.[8]
Medical use[edit]
Avanafil is used to treat erectile dysfunction (ED).[9]
Adverse effects[edit]
Although avanafil is generally well tolerated, dose dependent adverse effects can occur.[8] The most common adverse effects include headache, flushing, nasopharyngitis, nasal congestion, and back pain.[8] While it is also uncommon, there is a potential for visual disturbances to occur in patients.[8]
Mechanism of action[edit]
Avanafil inhibits phosphodiesterase-5, preventing the degradation of cGMP.[10][11] The increased levels of cGMP causes vasodilation, resulting in an increased blood flow in the penis.[11] Avanafil's mechanism of action takes places once nitric oxide is released, in association with sexual stimulation.[11]
Synthesis[edit]
Avanafil can be synthesized from a benzylamine derivative and a pyrimidine derivative:[4]
References[edit]
- ^ "Prescription medicines: registration of new chemical entities in Australia, 2016". Therapeutic Goods Administration (TGA). 21 June 2022. Retrieved 10 April 2023.
- ^ "Stendra FDA Approval History". Drugs.com. Retrieved 6 March 2021.
- ^ "Spedra (avanafil)". European Medicines Agency. Retrieved 17 April 2014.
- ^ a b US 6797709, Yamada K, Matsuki K, Omori K Kikkawa K, "Aromatic nitrogen-containing 6-membered cyclic compounds", issued 11 December 2003, assigned to Tanabe Seiyaku Co
- ^ "VIVUS Announces Avanafil Partnership With Menarini". Vivus Inc. Archived from the original on 2015-12-08.
- ^ "VIVUS and Metuchen Pharmaceuticals Announce License Agreement for Commercial Rights to Stendra". Vivus Inc. 3 October 2016.
- ^ "avanafil, Spedra". Medicine Net. Retrieved 17 April 2014.
- ^ a b c d e Kyle JA, Brown DA, Hill JK (October 2013). "Avanafil for erectile dysfunction". The Annals of Pharmacotherapy. 47 (10). Sage Publishing: 1312–1320. doi:10.1177/1060028013501989. PMID 24259695. S2CID 6562049.
- ^ Burke RM, Evans JD (2012). "Avanafil for treatment of erectile dysfunction: review of its potential". Vascular Health and Risk Management. 8: 517–523. doi:10.2147/VHRM.S26712. PMC 3433322. PMID 22973106.
- ^ Kotera J, Mochida H, Inoue H, Noto T, Fujishige K, Sasaki T, et al. (August 2012). "Avanafil, a potent and highly selective phosphodiesterase-5 inhibitor for erectile dysfunction". The Journal of Urology. 188 (2): 668–674. doi:10.1016/j.juro.2012.03.115. PMID 22704456.
- ^ a b c Sanford M (October 2013). "Avanafil: A Review of Its Use in Patients with Erectile Dysfunction". Drugs & Aging. 30 (10): 853–62. doi:10.1007/s40266-013-0112-x. PMID 23955441. S2CID 23558269.
External links[edit]
- "Avanafil". Drug Information Portal. U.S. National Library of Medicine.

