Myocardial infarction
| Myocardial infarction | |
|---|---|
| Specialty | Cardiology |
Myocardial infarction (MI or AMI for acute myocardial infarction), commonly known as a heart attack, occurs when the blood supply to part of the heart is interrupted causing some heart cells to die. This is most commonly due to occlusion (blockage) of a coronary artery following the rupture of a vulnerable atherosclerotic plaque, which is an unstable collection of lipids (like cholesterol) and white blood cells (especially macrophages) in the wall of an artery. The resulting ischemia (restriction in blood supply) and oxygen shortage, if left untreated for a sufficient period, can cause damage and/or death (infarction) of heart muscle tissue (myocardium).
Classical symptoms of acute myocardial infarction include sudden chest pain (typically radiating to the left arm or left side of the neck), shortness of breath, nausea, vomiting, palpitations, sweating, and anxiety (often described as a sense of impending doom). Women may experience fewer typical symptoms than men, most commonly shortness of breath, weakness, a feeling of indigestion, and fatigue.[1] Approximately one quarter of all myocardial infarctions are silent, without chest pain or other symptoms. A heart attack is a medical emergency, and people experiencing chest pain are advised to alert their emergency medical services, because prompt treatment is beneficial.
Heart attacks are the leading cause of death for both men and women all over the world.[2] Important risk factors are previous cardiovascular disease (such as angina, a previous heart attack or stroke), older age (especially men over 40 and women over 50), tobacco smoking, high blood levels of certain lipids (triglycerides, low-density lipoprotein or "bad cholesterol") and low levels of high density lipoprotein (HDL, "good cholesterol"), diabetes, high blood pressure, obesity, chronic kidney disease, heart failure, excessive alcohol consumption, the abuse of certain drugs (such as cocaine and methamphetamine), and chronic high stress levels.[3][4]
Immediate treatment for suspected acute myocardial infarction includes oxygen, aspirin, and sublingual glyceryl trinitrate (colloquially referred to as nitroglycerin and abbreviated as NTG or GTN). Pain relief is also often given, classically morphine sulfate.[5] A 2009 review of high flow oxygen in myocardial infarction however found increased mortality and infarct size calling into question the recommendation about its routine use.[6]
The patient will receive a number of diagnostic tests, such as an electrocardiogram (ECG, EKG), a chest X-ray and blood tests to detect elevations in cardiac markers (blood tests to detect heart muscle damage). The most often used markers are the creatine kinase-MB (CK-MB) fraction and the troponin I (TnI) or troponin T (TnT) levels. On the basis of the ECG, a distinction is made between ST elevation MI (STEMI) or non-ST elevation MI (NSTEMI). Most cases of STEMI are treated with thrombolysis or if possible with percutaneous coronary intervention (PCI, angioplasty and stent insertion), provided the hospital has facilities for coronary angiography. NSTEMI is managed with medication, although PCI is often performed during hospital admission. In patients who have multiple blockages and who are relatively stable, or in a few extraordinary emergency cases, bypass surgery of the blocked coronary artery is an option.
The phrase "heart attack" is sometimes used incorrectly to describe sudden cardiac death, which may or may not be the result of acute myocardial infarction. A heart attack is different from, but can be the cause of cardiac arrest, which is the stopping of the heartbeat, and cardiac arrhythmia, an abnormal heartbeat. It is also distinct from heart failure, in which the pumping action of the heart is impaired; severe myocardial infarction may lead to heart failure, but not necessarily.
Classification
Myocardial infarction is further subclassified into ST elevation MI verses non ST elevation MI based on the ECG.
Signs and symptoms
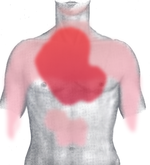

The onset of symptoms in myocardial infarction (MI) is usually gradual, over several minutes, and rarely instantaneous.[7] Chest pain is the most common symptom of acute myocardial infarction and is often described as a sensation of tightness, pressure, or squeezing. Chest pain due to ischemia (a lack of blood and hence oxygen supply) of the heart muscle is termed angina pectoris. Pain radiates most often to the left arm, but may also radiate to the lower jaw, neck, right arm, back, and epigastrium, where it may mimic heartburn. Levine's sign, in which the patient localizes the chest pain by clenching their fist over the sternum, has classically been thought to be predictive of cardiac chest pain, although a prospective observational study showed that it had a poor positive predictive value.[8]
Shortness of breath (dyspnea) occurs when the damage to the heart limits the output of the left ventricle, causing left ventricular failure and consequent pulmonary edema. Other symptoms include diaphoresis (an excessive form of sweating), weakness, light-headedness, nausea, vomiting, and palpitations. These symptoms are likely induced by a massive surge of catecholamines from the sympathetic nervous system[9] which occurs in response to pain and the hemodynamic abnormalities that result from cardiac dysfunction. Loss of consciousness (due to inadequate cerebral perfusion and cardiogenic shock) and even sudden death (frequently due to the development of ventricular fibrillation) can occur in myocardial infarctions.
Women and older patients experience atypical symptoms more frequently than their male and younger counterparts.[10] Women also have more symptoms compared to men (2.6 on average vs 1.8 symptoms in men).[10] The most common symptoms of MI in women include dyspnea, weakness, and fatigue. Fatigue, sleep disturbances, and dyspnea have been reported as frequently occurring symptoms which may manifest as long as one month before the actual clinically manifested ischemic event. In women, chest pain may be less predictive of coronary ischemia than in men.[11]
Approximately half of all MI patients have experienced warning symptoms such as chest pain prior to the infarction.[12]
Approximately one fourth of all myocardial infarctions are silent, without chest pain or other symptoms.[13] These cases can be discovered later on electrocardiograms or at autopsy without a prior history of related complaints. A silent course is more common in the elderly, in patients with diabetes mellitus[14] and after heart transplantation, probably because the donor heart is not connected to nerves of the host.[15] In diabetics, differences in pain threshold, autonomic neuropathy, and psychological factors have been cited as possible explanations for the lack of symptoms.[14]
Any group of symptoms compatible with a sudden interruption of the blood flow to the heart are called an acute coronary syndrome.[16]
The differential diagnosis includes other catastrophic causes of chest pain, such as pulmonary embolism, aortic dissection, pericardial effusion causing cardiac tamponade, tension pneumothorax, and esophageal rupture.[17]
Causes and risk factors
Heart attack rates are higher in association with intense exertion, be it psychological stress or physical exertion, especially if the exertion is more intense than the individual usually performs.[18] Quantitatively, the period of intense exercise and subsequent recovery is associated with about a 6-fold higher myocardial infarction rate (compared with other more relaxed time frames) for people who are physically very fit.[18] For those in poor physical condition, the rate differential is over 35-fold higher.[18] One observed mechanism for this phenomenon is the increased arterial pulse pressure stretching and relaxation of arteries with each heart beat which, as has been observed with intravascular ultrasound, increases mechanical "shear stress" on atheromas and the likelihood of plaque rupture.[18]
Acute severe infection, such as pneumonia, can trigger myocardial infarction. A more controversial link is that between Chlamydophila pneumoniae infection and atherosclerosis.[19] While this intracellular organism has been demonstrated in atherosclerotic plaques, evidence is inconclusive as to whether it can be considered a causative factor.[19] Treatment with antibiotics in patients with proven atherosclerosis has not demonstrated a decreased risk of heart attacks or other coronary vascular diseases.[20]
There is an association of an increased incidence of a heart attack in the morning hours, more specifically around 9 a.m. [21][22][23]. Some investigators have noticed that the ability of platelets to aggregate varies according to a circadian rhythm, although they have not proven causation.[24] Some investigators theorize that this increased incidence may be related to the circadian variation in cortisol production affecting the concentrations of various cytokines and other mediators of inflammation.[25]
Risk factors
Risk factors for atherosclerosis are generally risk factors for myocardial infarction:
- Older age
- Male sex[18]
- Tobacco smoking
- Hypercholesterolemia (more accurately hyperlipoproteinemia, especially high low density lipoprotein and low high density lipoprotein)
- Hyperhomocysteinemia (high homocysteine, a toxic blood amino acid that is elevated when intakes of vitamins B2, B6, B12 and folic acid are insufficient)
- Diabetes (with or without insulin resistance)
- High blood pressure
- Obesity[26] (defined by a body mass index of more than 30 kg/m², or alternatively by waist circumference or waist-hip ratio).
- Stress Occupations with high stress index are known to have susceptibility for atherosclerosis.
Many of these risk factors are modifiable, so many heart attacks can be prevented by maintaining a healthier lifestyle. Physical activity, for example, is associated with a lower risk profile.[27] Non-modifiable risk factors include age, sex, and family history of an early heart attack (before the age of 60), which is thought of as reflecting a genetic predisposition.[18]
Socioeconomic factors such as a shorter education and lower income (particularly in women), and unmarried cohabitation may also contribute to the risk of MI.[28] To understand epidemiological study results, it's important to note that many factors associated with MI mediate their risk via other factors. For example, the effect of education is partially based on its effect on income and marital status.[28]
Women who use combined oral contraceptive pills have a modestly increased risk of myocardial infarction, especially in the presence of other risk factors, such as smoking.[29]
Inflammation is known to be an important step in the process of atherosclerotic plaque formation.[30] C-reactive protein (CRP) is a sensitive but non-specific marker for inflammation. Elevated CRP blood levels, especially measured with high sensitivity assays, can predict the risk of MI, as well as stroke and development of diabetes.[30] Moreover, some drugs for MI might also reduce CRP levels.[30] The use of high sensitivity CRP assays as a means of screening the general population is advised against, but it may be used optionally at the physician's discretion, in patients who already present with other risk factors or known coronary artery disease.[31] Whether CRP plays a direct role in atherosclerosis remains uncertain.[30]
Inflammation in periodontal disease may be linked coronary heart disease, and since periodontitis is very common, this could have great consequences for public health.[32] Serological studies measuring antibody levels against typical periodontitis-causing bacteria found that such antibodies were more present in subjects with coronary heart disease.[33] Periodontitis tends to increase blood levels of CRP, fibrinogen and cytokines;[34] thus, periodontitis may mediate its effect on MI risk via other risk factors.[35] Preclinical research suggests that periodontal bacteria can promote aggregation of platelets and promote the formation of foam cells.[36][37] A role for specific periodontal bacteria has been suggested but remains to be established.[38]
Baldness, hair greying, a diagonal earlobe crease (Frank's sign[39]) and possibly other skin features have been suggested as independent risk factors for MI.[40] Their role remains controversial; a common denominator of these signs and the risk of MI is supposed, possibly genetic. [41]
Calcium deposition is another part of atherosclerotic plaque formation. Calcium deposits in the coronary arteries can be detected with CT scans. Several studies have shown that coronary calcium can provide predictive information beyond that of classical risk factors.[42][43][44]
Pathophysiology

Acute myocardial infarction refers to two subtypes of acute coronary syndrome, namely non-ST-elevated myocardial infarction and ST-elevated myocardial infarction, which are most frequently (but not always) a manifestation of coronary artery disease. The most common triggering event is the disruption of an atherosclerotic plaque in an epicardial coronary artery, which leads to a clotting cascade, sometimes resulting in total occlusion of the artery. Atherosclerosis is the gradual buildup of cholesterol and fibrous tissue in plaques in the wall of arteries (in this case, the coronary arteries), typically over decades. Blood stream column irregularities visible on angiography reflect artery lumen narrowing as a result of decades of advancing atherosclerosis. Plaques can become unstable, rupture, and additionally promote a thrombus (blood clot) that occludes the artery; this can occur in minutes. When a severe enough plaque rupture occurs in the coronary vasculature, it leads to myocardial infarction (necrosis of downstream myocardium).
If impaired blood flow to the heart lasts long enough, it triggers a process called the ischemic cascade; the heart cells in the territory of the occluded coronary artery die (chiefly through necrosis) and do not grow back. A collagen scar forms in its place.The necrosis is largely complete within 6 hours.[45] Recent studies indicate that another form of cell death called apoptosis also plays a role in the process of tissue damage subsequent to myocardial infarction.[46] As a result, the patient's heart will be permanently damaged. This scar tissue also puts the patient at risk for potentially life threatening arrhythmias, and may result in the formation of a ventricular aneurysm that can rupture with catastrophic consequences.
Injured heart tissue conducts electrical impulses more slowly than normal heart tissue. The difference in conduction velocity between injured and uninjured tissue can trigger re-entry or a feedback loop that is believed to be the cause of many lethal arrhythmias. The most serious of these arrhythmias is ventricular fibrillation (V-Fib/VF), an extremely fast and chaotic heart rhythm that is the leading cause of sudden cardiac death. Another life threatening arrhythmia is ventricular tachycardia (V-Tach/VT), which may or may not cause sudden cardiac death. However, ventricular tachycardia usually results in rapid heart rates that prevent the heart from pumping blood effectively. Cardiac output and blood pressure may fall to dangerous levels, which can lead to further coronary ischemia and extension of the infarct.
The cardiac defibrillator is a device that was specifically designed to terminate these potentially fatal arrhythmias. The device works by delivering an electrical shock to the patient in order to depolarize a critical mass of the heart muscle, in effect "rebooting" the heart. This therapy is time dependent, and the odds of successful defibrillation decline rapidly after the onset of cardiopulmonary arrest.
Diagnosis
The diagnosis of myocardial infarction is made by integrating the history of the presenting illness and physical examination with electrocardiogram findings and cardiac markers (blood tests for heart muscle cell damage).[47] A coronary angiogram allows visualization of narrowings or obstructions on the heart vessels, and therapeutic measures can follow immediately. At autopsy, a pathologist can diagnose a myocardial infarction based on anatomopathological findings.
A chest radiograph and routine blood tests may indicate complications or precipitating causes and are often performed upon arrival to an emergency department. New regional wall motion abnormalities on an echocardiogram are also suggestive of a myocardial infarction. Echo may be performed in equivocal cases by the on-call cardiologist.[48] In stable patients whose symptoms have resolved by the time of evaluation, technetium-99m 2-methoxyisobutylisonitrile (Tc99m MIBI) or thallium-201 chloride can be used in nuclear medicine to visualize areas of reduced blood flow in conjunction with physiologic or pharmocologic stress.[48][49] Thallium may also be used to determine viability of tissue, distinguishing whether non-functional myocardium is actually dead or merely in a state of hibernation or of being stunned.[50]
Diagnostic criteria
WHO criteria[51] have classically been used to diagnose MI; a patient is diagnosed with myocardial infarction if two (probable) or three (definite) of the following criteria are satisfied:
- Clinical history of ischaemic type chest pain lasting for more than 20 minutes
- Changes in serial ECG tracings
- Rise and fall of serum cardiac biomarkers such as creatine kinase-MB fraction and troponin
The WHO criteria were refined in 2000 to give more prominence to cardiac biomarkers.[52] According to the new guidelines, a cardiac troponin rise accompanied by either typical symptoms, pathological Q waves, ST elevation or depression or coronary intervention are diagnostic of MI.
Physical examination
The general appearance of patients may vary according to the experienced symptoms; the patient may be comfortable, or restless and in severe distress with an increased respiratory rate. A cool and pale skin is common and points to vasoconstriction. Some patients have low-grade fever (38–39 °C). Blood pressure may be elevated or decreased, and the pulse can be become irregular.[53][54]
If heart failure ensues, elevated jugular venous pressure and hepatojugular reflux, or swelling of the legs due to peripheral edema may be found on inspection. Rarely, a cardiac bulge with a pace different from the pulse rhythm can be felt on precordial examination. Various abnormalities can be found on auscultation, such as a third and fourth heart sound, systolic murmurs, paradoxical splitting of the second heart sound, a pericardial friction rub and rales over the lung.[53][55]
Electrocardiogram
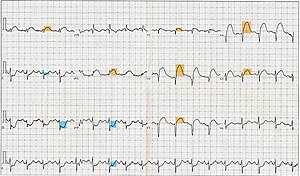
The primary purpose of the electrocardiogram is to detect ischemia or acute coronary injury in broad, symptomatic emergency department populations. However, the standard 12 lead ECG has several limitations. An ECG represents a brief sample in time. Because unstable ischemic syndromes have rapidly changing supply versus demand characteristics, a single ECG may not accurately represent the entire picture.[56] It is therefore desirable to obtain serial 12 lead ECGs, particularly if the first ECG is obtained during a pain-free episode. Alternatively, many emergency departments and chest pain centers use computers capable of continuous ST segment monitoring.[57] The standard 12 lead ECG also does not directly examine the right ventricle, and is relatively poor at examining the posterior basal and lateral walls of the left ventricle. In particular, acute myocardial infarction in the distribution of the circumflex artery is likely to produce a nondiagnostic ECG.[56] The use of additional ECG leads like right-sided leads V3R and V4R and posterior leads V7, V8, and V9 may improve sensitivity for right ventricular and posterior myocardial infarction. In spite of these limitations, the 12 lead ECG stands at the center of risk stratification for the patient with suspected acute myocardial infarction. Mistakes in interpretation are relatively common, and the failure to identify high risk features has a negative effect on the quality of patient care.[58]
The 12 lead ECG is used to classify patients into one of three groups:[59]
- those with ST segment elevation or new bundle branch block (suspicious for acute injury and a possible candidate for acute reperfusion therapy with thrombolytics or primary PCI),
- those with ST segment depression or T wave inversion (suspicious for ischemia), and
- those with a so-called non-diagnostic or normal ECG.
A normal ECG does not rule out acute myocardial infarction. Sometimes the earliest presentation of acute myocardial infarction is the hyperacute T wave, which is treated the same as ST segment elevation.[60] In practice this is rarely seen, because it only exists for 2-30 minutes after the onset of infarction.[61] Hyperacute T waves need to be distinguished from the peaked T waves associated with hyperkalemia.[62] The current guidelines for the ECG diagnosis of acute myocardial infarction require at least 1 mm (0.1 mV) of ST segment elevation in the limb leads, and at least 2 mm elevation in the precordial leads. These elevations must be present in anatomically contiguous leads.[59] (I, aVL, V5, V6 correspond to the lateral wall; V1-V4 correspond to the anterior wall; II, III, aVF correspond to the inferior wall.) This criterion is problematic, however, as acute myocardial infarction is not the most common cause of ST segment elevation in chest pain patients.[63] Over 90% of healthy men have at least 1 mm (0.1 mV) of ST segment elevation in at least one precordial lead.[64] The clinician must therefore be well versed in recognizing the so-called ECG mimics of acute myocardial infarction, which include left ventricular hypertrophy, left bundle branch block, paced rhythm, early repolarization, pericarditis, hyperkalemia, and ventricular aneurysm.[65][66][64]
Cardiac markers
Cardiac markers or cardiac enzymes are proteins that leak out of injured myocardial cells through their damaged cell membranes into the bloodstream. Until the 1980s, the enzymes SGOT and LDH were used to assess cardiac injury. Now, the markers most widely used in detection of MI are MB subtype of the enzyme creatine kinase and cardiac troponins T and I as they are more specific for myocardial injury. The cardiac troponins T and I which are released within 4-6 hours of an attack of MI and remain elevated for up to 2 weeks, have nearly complete tissue specificity and are now the preferred markers for asssessing myocardial damage.[67] Elevated troponins in the setting of chest pain may accurately predict a high likelihood of a myocardial infarction in the near future.[68] New markers such as glycogen phosphorylase isoenzyme BB are under investigation.[69]
The diagnosis of myocardial infarction requires two out of three components (history, ECG, and enzymes). When damage to the heart occurs, levels of cardiac markers rise over time, which is why blood tests for them are taken over a 24-hour period. Because these enzyme levels are not elevated immediately following a heart attack, patients presenting with chest pain are generally treated with the assumption that a myocardial infarction has occurred and then evaluated for a more precise diagnosis.[70]
Angiography

In difficult cases or in situations where intervention to restore blood flow is appropriate, coronary angiography can be performed. A catheter is inserted into an artery (usually the femoral artery) and pushed to the vessels supplying the heart. A radio-opaque dye is administered through the catheter and a sequence of x-rays (fluoroscopy) is performed. Obstructed or narrowed arteries can be identified, and angioplasty applied as a therapeutic measure (see below). Angioplasty requires extensive skill, especially in emergency settings. It is performed by a physician trained in interventional cardiology.
Histopathology

Histopathological examination of the heart may reveal infarction at autopsy. Under the microscope, myocardial infarction presents as a circumscribed area of ischemic, coagulative necrosis (cell death). On gross examination, the infarct is not identifiable within the first 12 hours.[71]
Although earlier changes can be discerned using electron microscopy, one of the earliest changes under a normal microscope are so-called wavy fibers.[72] Subsequently, the myocyte cytoplasm becomes more eosinophilic (pink) and the cells lose their transversal striations, with typical changes and eventually loss of the cell nucleus.[73] The interstitium at the margin of the infarcted area is initially infiltrated with neutrophils, then with lymphocytes and macrophages, who phagocytose ("eat") the myocyte debris. The necrotic area is surrounded and progressively invaded by granulation tissue, which will replace the infarct with a fibrous (collagenous) scar (which are typical steps in wound healing). The interstitial space (the space between cells outside of blood vessels) may be infiltrated with red blood cells.[71]
These features can be recognized in cases where the perfusion was not restored; reperfused infarcts can have other hallmarks, such as contraction band necrosis.[74]
Management
A heart attack is a medical urgency which demands both immediate attention and activation of the emergency medical services. The ultimate goal of the management in the acute phase of the disease is to salvage as much myocardium as possible and prevent further complications. As time passes, the risk of damage to the heart muscle increases; hence the phrase that in myocardial infarction, "time is muscle," and time wasted is muscle lost.[75]
Oxygen, aspirin, glyceryl trinitrate (nitroglycerin) and analgesia (usually morphine, although experts often argue this point), hence the popular mnemonic MONA, morphine, oxygen, nitro, aspirin) are usually administered as soon as possible. In many areas, first responders are trained to administer these prior to arrival at the hospital. Morphine is classically used if nitrogylcerin is not effective due to its ability to dilate blood vessels, which may aids in blood flow to the heart as well as the pain relief it provides. Morphine may also cause hypotension (usually in the setting of hypovolemia), and should be avoided in the case of right ventricular infarction. Moreover, the CRUSADE trial demonstrated an increase in mortality with administering morphine in the setting of NSTEMI.[76] A 2009 review of high flow oxygen in myocardial infarction found increased mortality and infarct size, calling into question the recommendation about its routine use.[77]
Of the first line agents, only aspirin has been proven to decrease mortality.[78]
Once the diagnosis of myocardial infarction is confirmed, other pharmacologic agents are often given. These include beta blockers,[79][80] anticoagulation (typically with heparin),[81] and possibly additional antiplatelet agents such as clopidogrel.[81] While these agents can decrease mortality in the setting of an acute myocardial infarction, they can lead to complications and potentially death if used in the wrong setting.[citation needed]
Cocaine associated myocardial infarction should be managed in a manner similar to other patients with acute coronary syndrome except beta blockers should not be used and benzodiazepines should be administered early.[82]
The treatment itself may have complications. If attempts to restore the blood flow are initiated after a critical period of only a few hours, the result may be a reperfusion injury instead of amelioration.[83]
First aid
As myocardial infarction is a common medical emergency, the signs are often part of first aid courses. The emergency action principles also apply in the case of myocardial infarction.
When symptoms of myocardial infarction occur, people wait an average of three hours, instead of doing what is recommended: calling for help immediately.[84][85] Acting immediately by calling the emergency services can prevent sustained damage to the heart ("time is muscle").[75]
Certain positions allow the patient to rest in a position which minimizes breathing difficulties. A half-sitting position with knees bent is often recommended. Access to more oxygen can be given by opening the window and widening the collar for easier breathing.
Aspirin can be given quickly (if the patient is not allergic to aspirin); but taking aspirin before calling the emergency medical services may be associated with unwanted delay.[86] Aspirin has an antiplatelet effect which inhibits formation of further thrombi (blood clots) that clog arteries. Chewing is the preferred method of administration, so that the Aspirin can be absorbed quickly. Dissolved soluble preparations or sublingual administration can also be used. U.S. guidelines recommend a dose of 162–325 mg.[81] Australian guidelines recommend a dose of 150–300 mg.[87]
Glyceryl trinitrate (nitroglycerin) sublingually (under the tongue) can be given if available.
If an automated external defibrillator (AED) is available the rescuer should immediately bring the AED to the patient's side and be prepared to follow its instructions, especially should the victim lose consciousness.
If possible the rescuer should obtain basic information from the victim, in case the patient is unable to answer questions once emergency medical technicians arrive. The victim's name and any information regarding the nature of the victim's pain will be useful to health care providers. The exact time that these symptoms started may be critical for determining what interventions can be safely attempted once the victim reaches the medical center. Other useful pieces of information include what the patient was doing at the onset of symptoms, and anything else that might give clues to the pathology of the chest pain. It is also very important to relay any actions that have been taken, such as the number or dose of aspirin or nitroglycerin given, to the EMS personnel.
Other general first aid principles include monitoring pulse, breathing, level of consciousness and, if possible, the blood pressure of the patient. In case of cardiac arrest, cardiopulmonary resuscitation (CPR) can be administered.
Automatic external defibrillation (AED)
Since the publication of data showing that the availability of automated external defibrillators (AEDs) in public places may significantly increase chances of survival, many of these have been installed in public buildings, public transport facilities, and in non-ambulance emergency vehicles (e.g. police cars and fire engines). AEDs analyze the heart's rhythm and determine whether the rhythm is amenable to defibrillation ("shockable"), as in ventricular tachycardia and ventricular fibrillation.
Emergency services
Emergency Medical Services (EMS) Systems vary considerably in their ability to evaluate and treat patients with suspected acute myocardial infarction. Some provide as little as first aid and early defibrillation. Others employ highly trained paramedics with sophisticated technology and advanced protocols.[88] Early access to EMS is promoted by a 9-1-1 system currently available to 90% of the population in the United States.[88] Most are capable of providing oxygen, IV access, sublingual nitroglycerine, morphine, and aspirin. Some are capable of providing thrombolytic therapy in the prehospital setting.[89][90]
With primary PCI emerging as the preferred therapy for ST segment elevation myocardial infarction, EMS can play a key role in reducing door to balloon intervals (the time from presentation to a hospital ER to the restoration of coronary artery blood flow) by performing a 12 lead ECG in the field and using this information to triage the patient to the most appropriate medical facility.[91][92][93][94] In addition, the 12 lead ECG can be transmitted to the receiving hospital, which enables time saving decisions to be made prior to the patient's arrival. This may include a "cardiac alert" or "STEMI alert" that calls in off duty personnel in areas where the cardiac cath lab is not staffed 24 hours a day.[95] Even in the absence of a formal alerting program, prehospital 12 lead ECGs are independently associated with reduced door to treatment intervals in the emergency department.[96]
Reperfusion
The concept of reperfusion has become so central to the modern treatment of acute myocardial infarction, that we are said to be in the reperfusion era.[97][98] Patients who present with suspected acute myocardial infarction and ST segment elevation (STEMI) or new bundle branch block on the 12 lead ECG are presumed to have an occlusive thrombosis in an epicardial coronary artery. They are therefore candidates for immediate reperfusion, either with thrombolytic therapy, percutaneous coronary intervention (PCI) or when these therapies are unsuccessful, bypass surgery.
Individuals without ST segment elevation are presumed to be experiencing either unstable angina (UA) or non-ST segment elevation myocardial infarction (NSTEMI). They receive many of the same initial therapies and are often stabilized with antiplatelet drugs and anticoagulated. If their condition remains (hemodynamically) stable, they can be offered either late coronary angiography with subsequent restoration of blood flow (revascularization), or non-invasive stress testing to determine if there is significant ischemia that would benefit from revascularization. If hemodynamic instability develops in individuals with NSTEMIs, they may undergo urgent coronary angiography and subsequent revascularization. The use of thrombolytic agents is contraindicated in this patient subset, however.[99]
The basis for this distinction in treatment regimens is that ST segment elevations on an ECG are typically due to complete occlusion of a coronary artery. On the other hand, in NSTEMIs there is typically a sudden narrowing of a coronary artery with preserved (but diminished) flow to the distal myocardium. Anticoagulation and antiplatelet agents are given to prevent the narrowed artery from occluding.
At least 10% of patients with STEMI don't develop myocardial necrosis (as evidenced by a rise in cardiac markers) and subsequent Q waves on EKG after reperfusion therapy. Such a successful restoration of flow to the infarct-related artery during an acute myocardial infarction is known as "aborting" the myocardial infarction. If treated within the hour, about 25% of STEMIs can be aborted.[100]
Thrombolytic therapy
Thrombolytic therapy is indicated for the treatment of STEMI if the drug can be administered within 12 hours of the onset of symptoms, the patient is eligible based on exclusion criteria, and primary PCI is not immediately available.[81] The effectiveness of thrombolytic therapy is highest in the first 2 hours. After 12 hours, the risk associated with thrombolytic therapy outweighs any benefit.[99][101] Because irreversible injury occurs within 2–4 hours of the infarction, there is a limited window of time available for reperfusion to work.
Thrombolytic drugs are contraindicated for the treatment of unstable angina and NSTEMI[99][102] and for the treatment of individuals with evidence of cardiogenic shock.[103]
Although no perfect thrombolytic agent exists, an ideal thrombolytic drug would lead to rapid reperfusion, have a high sustained patency rate, be specific for recent thrombi, be easily and rapidly administered, create a low risk for intra-cerebral and systemic bleeding, have no antigenicity, adverse hemodynamic effects, or clinically significant drug interactions, and be cost effective.[104] Currently available thrombolytic agents include streptokinase, urokinase, and alteplase (recombinant tissue plasminogen activator, rtPA). More recently, thrombolytic agents similar in structure to rtPA such as reteplase and tenecteplase have been used. These newer agents boast efficacy at least as good as rtPA with significantly easier administration. The thrombolytic agent used in a particular individual is based on institution preference and the age of the patient.
Depending on the thrombolytic agent being used, adjuvant anticoagulation with heparin or low molecular weight heparin may be of benefit.[105][106] With TPa and related agents (reteplase and tenecteplase), heparin is needed to maintain coronary artery patency. Because of the anticoagulant effect of fibrinogen depletion with streptokinase[107] and urokinase[108][109][110] treatment, it is less necessary there.[105]
Intracranial bleeding (ICB) and subsequent cerebrovascular accident (CVA) is a serious side effect of thrombolytic use. The risk of ICB is dependent on a number of factors, including a previous episode of intracranial bleed, age of the individual, and the thrombolytic regimen that is being used. In general, the risk of ICB due to thrombolytic use for the treatment of an acute myocardial infarction is between 0.5 and 1 percent.[105]
Thrombolytic therapy to abort a myocardial infarction is not always effective. The degree of effectiveness of a thrombolytic agent is dependent on the time since the myocardial infarction began, with the best results occurring if the thrombolytic agent is used within two hours of the onset of symptoms.[111][90] If the individual presents more than 12 hours after symptoms commenced, the risk of intracranial bleed are considered higher than the benefits of the thrombolytic agent.[112] Failure rates of thrombolytics can be as high as 20% or higher.[113] In cases of failure of the thrombolytic agent to open the infarct-related coronary artery, the patient is then either treated conservatively with anticoagulants and allowed to "complete the infarction" or percutaneous coronary intervention (PCI, see below) is then performed. Percutaneous coronary intervention in this setting is known as "rescue PCI" or "salvage PCI". Complications, particularly bleeding, are significantly higher with rescue PCI than with primary PCI due to the action of the thrombolytic agent.
Percutaneous coronary intervention

The benefit of prompt, expertly performed primary percutaneous coronary intervention over thrombolytic therapy for acute ST elevation myocardial infarction is now well established.[114][115][116] When performed rapidly by an experienced team, primary PCI restores flow in the culprit artery in more than 95% of patients compared with the spontaneous recanalization rate of about 65%.[114] Logistic and economic obstacles seem to hinder a more widespread application of percutaneous coronary intervention (PCI) via cardiac catheterization,[117] although the feasibility of regionalized PCI for STEMI is currently being explored in the United States.[118] The use of percutaneous coronary intervention as a therapy to abort a myocardial infarction is known as primary PCI. The goal of primary PCI is to open the artery as soon as possible, and preferably within 90 minutes of the patient presenting to the emergency room. This time is referred to as the door-to-balloon time. Few hospitals can provide PCI within the 90 minute interval,[119] which prompted the American College of Cardiology (ACC) to launch a national Door to Balloon (D2B) Initiative in November 2006. Over 800 hospitals have joined the D2B Alliance as of March 16, 2007.[120]
One particularly successful implementation of a primary PCI protocol is in the Calgary Health Region under the auspices of the Libin Cardiovascular Institute of Alberta. Under this model, EMS teams responding to an emergency electronically transmit the ECG directly to a digital archiving system that allows emergency room physicians and/or cardiologists to immediately confirm the diagnosis. This in turn allows for redirection of the EMS teams to facilities prepped to conduct time-critical angioplasty, based on the ECG analysis. In an article published in the Canadian Medical Association Journal in June 2007, the Calgary implementation resulted in a median time to treatment of 62 minutes.[121]
The current guidelines in the United States restrict primary PCI to hospitals with available emergency bypass surgery as a backup,[81] but this is not the case in other parts of the world.[122]
Primary PCI involves performing a coronary angiogram to determine the anatomical location of the infarcting vessel, followed by balloon angioplasty (and frequently deployment of an intracoronary stent) of the thrombosed arterial segment. In some settings, an extraction catheter may be used to attempt to aspirate (remove) the thrombus prior to balloon angioplasty. While the use of intracoronary stents do not improve the short term outcomes in primary PCI, the use of stents is widespread because of the decreased rates of procedures to treat restenosis compared to balloon angioplasty.[123]
Adjuvant therapy during primary PCI include intravenous heparin, aspirin, and clopidogrel. The use of glycoprotein IIb/IIIa inhibitors are often used in the setting of primary PCI to reduce the risk of ischemic complications during the procedure.[124][125] Due to the number of antiplatelet agents and anticoagulants used during primary PCI, the risk of bleeding associated with the procedure are higher than during an elective PCI.[126]
Coronary artery bypass surgery
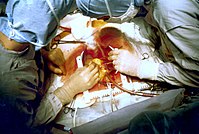
Despite the guidelines, emergency bypass surgery for the treatment of an acute myocardial infarction (MI) is less common than PCI or medical management. In an analysis of patients in the U.S. National Registry of Myocardial Infarction (NRMI) from January 1995 to May 2004, the percentage of patients with cardiogenic shock treated with primary PCI rose from 27.4% to 54.4%, while the increase in CABG treatment was only from 2.1% to 3.2%.[127]
Emergency coronary artery bypass graft surgery (CABG) is usually undertaken to simultaneously treat a mechanical complication, such as a ruptured papillary muscle, or a ventricular septal defect, with ensueing cardiogenic shock.[128] In uncomplicated MI, the mortality rate can be high when the surgery is performed immediately following the infarction.[129] If this option is entertained, the patient should be stabilized prior to surgery, with supportive interventions such as the use of an intra-aortic balloon pump.[130] In patients developing cardiogenic shock after a myocardial infarction, both PCI and CABG are satisfactory treatment options, with similar survival rates.[131][132]
Coronary artery bypass surgery involves an artery or vein from the patient being implanted to bypass narrowings or occlusions on the coronary arteries. Several arteries and veins can be used, however internal mammary artery grafts have demonstrated significantly better long-term patency rates than great saphenous vein grafts.[133] In patients with two or more coronary arteries affected, bypass surgery is associated with higher long-term survival rates compared to percutaneous interventions.[134] In patients with single vessel disease, surgery is comparably safe and effective, and may be a treatment option in selected cases.[135] Bypass surgery has higher costs initially, but becomes cost-effective in the long term.[136] A surgical bypass graft is more invasive initially but bears less risk of recurrent procedures (but these may be again minimally invasive).[135]
Monitoring for arrhythmias
Additional objectives are to prevent life-threatening arrhythmias or conduction disturbances. This requires monitoring in a coronary care unit and protocolised administration of antiarrhythmic agents. Antiarrhythmic agents are typically only given to individuals with life-threatening arrhythmias after a myocardial infarction and not to suppress the ventricular ectopy that is often seen after a myocardial infarction.[137][138][139]
Austere environments
- Wilderness first aid
In wilderness first aid, a possible heart attack justifies evacuation by the fastest available means, including MEDEVAC, even in the earliest or precursor stages. The patient will rapidly be incapable of further exertion and have to be carried out.
- Air travel
Certified personnel traveling by commercial aircraft may be able to assist an MI patient by using the on-board first aid kit, which may contain some cardiac drugs (such as glyceryl trinitrate spray, aspirin, or opioid painkillers), an AED,[140] and oxygen. Pilots may divert the flight to land at a nearby airport. Cardiac monitors are being introduced by some airlines, and they can be used by both on-board and ground-based physicians.[141]
Rehabilitation
Cardiac rehabilitation aims to optimize function and quality of life in those afflicted with a heart disease. This can be with the help of a physician, or in the form of a cardiac rehabilitation program.[142]
Physical exercise is an important part of rehabilitation after a myocardial infarction, with beneficial effects on cholesterol levels, blood pressure, weight, stress and mood.[142] Some patients become afraid of exercising because it might trigger another infarct.[143] Patients are stimulated to exercise, and should only avoid certain exerting activities such as shovelling. Local authorities may place limitations on driving motorised vehicles.[144] Some people are afraid to have sex after a heart attack. Most people can resume sexual activities after 3 to 4 weeks. The amount of activity needs to be dosed to the patient's possibilities.[145]
New therapies under investigation
Patients who receive stem cell treatment by coronary artery injections of stem cells derived from their own bone marrow after a myocardial infarction (MI) show improvements in left ventricular ejection fraction and end-diastolic volume not seen with placebo. The larger the initial infarct size, the greater the effect of the infusion. Clinical trials of progenitor cell infusion as a treatment approach to ST elevation MI are proceeding.[146]
There are currently 3 biomaterial and tissue engineering approaches for the treatment of MI, but these are in an even earlier stage of medical research, so many questions and issues need to be addressed before they can be applied to patients. The first involves polymeric left ventricular restraints in the prevention of heart failure. The second utilizes in vitro engineered cardiac tissue, which is subsequently implanted in vivo. The final approach entails injecting cells and/or a scaffold into the myocardium to create in situ engineered cardiac tissue.[147]
Complications
Complications may occur immediately following the heart attack (in the acute phase), or may need time to develop (a chronic problem). After an infarction, an obvious complication is a second infarction, which may occur in the domain of another atherosclerotic coronary artery, or in the same zone if there are any live cells left in the infarct.
Congestive heart failure
A myocardial infarction may compromise the function of the heart as a pump for the circulation, a state called heart failure. There are different types of heart failure; left- or right-sided (or bilateral) heart failure may occur depending on the affected part of the heart, and it is a low-output type of failure. If one of the heart valves is affected, this may cause dysfunction, such as mitral regurgitation in the case of left-sided coronary occlusion that disrupts the blood supply of the papillary muscles. The incidence of heart failure is particularly high in patients with diabetes and requires special management strategies.[148]
Myocardial rupture
Myocardial rupture is most common three to five days after myocardial infarction, commonly of small degree, but may occur one day to three weeks later. In the modern era of early revascularization and intensive pharmacotherapy as treatment for MI, the incidence of myocardial rupture is about 1% of all MIs.[149] This may occur in the free walls of the ventricles, the septum between them, the papillary muscles, or less commonly the atria. Rupture occurs because of increased pressure against the weakened walls of the heart chambers due to heart muscle that cannot pump blood out effectively. The weakness may also lead to ventricular aneurysm, a localized dilation or ballooning of the heart chamber.
Risk factors for myocardial rupture include completion of infarction (no revascularization performed), female sex, advanced age, and a lack of a previous history of myocardial infarction.[149] In addition, the risk of rupture is higher in individuals who are revascularized with a thrombolytic agent than with PCI.[150][151] The shear stress between the infarcted segment and the surrounding normal myocardium (which may be hypercontractile in the post-infarction period) makes it a nidus for rupture.[152]
Rupture is usually a catastrophic event that may result a life-threatening process known as cardiac tamponade, in which blood accumulates within the pericardium or heart sac, and compresses the heart to the point where it cannot pump effectively. Rupture of the intraventricular septum (the muscle separating the left and right ventricles) causes a ventricular septal defect with shunting of blood through the defect from the left side of the heart to the right side of the heart, which can lead to right ventricular failure as well as pulmonary overcirculation. Rupture of the papillary muscle may also lead to acute mitral regurgitation and subsequent pulmonary edema and possibly even cardiogenic shock.
Life-threatening arrhythmia

Since the electrical characteristics of the infarcted tissue change (see pathophysiology section), arrhythmias are a frequent complication.[153] The re-entry phenomenon may cause rapid heart rates (ventricular tachycardia and even ventricular fibrillation), and ischemia in the electrical conduction system of the heart may cause a complete heart block (when the impulse from the sinoatrial node, the normal cardiac pacemaker, does not reach the heart chambers).[154][155]
Pericarditis
As a reaction to the damage of the heart muscle, inflammatory cells are attracted. The inflammation may reach out and affect the heart sac. This is called pericarditis. In Dressler's syndrome, this occurs several weeks after the initial event.
Cardiogenic shock
A complication that may occur in the acute setting soon after a myocardial infarction or in the weeks following it is cardiogenic shock. Cardiogenic shock is defined as a hemodynamic state in which the heart cannot produce enough of a cardiac output to supply an adequate amount of oxygenated blood to the tissues of the body.
While the data on performing interventions on individuals with cardiogenic shock is sparse, trial data suggests a long-term mortality benefit in undergoing revascularization if the individual is less than 75 years old and if the onset of the acute myocardial infarction is less than 36 hours and the onset of cardiogenic shock is less than 18 hours.[103] If the patient with cardiogenic shock is not going to be revascularized, aggressive hemodynamic support is warranted, with insertion of an intra-aortic balloon pump if not contraindicated.[103] If diagnostic coronary angiography does not reveal a culprit blockage that is the cause of the cardiogenic shock, the prognosis is poor.[103]
Prevention
The risk of a recurrent myocardial infarction decreases with strict blood pressure management and lifestyle changes, chiefly smoking cessation, regular exercise, a sensible diet for patients with heart disease, and limitation of alcohol intake.
Patients are usually commenced on several long-term medications post-MI, with the aim of preventing secondary cardiovascular events such as further myocardial infarctions, congestive heart failure or cerebrovascular accident (CVA). Unless contraindicated, such medications may include:[156][87]
- Antiplatelet drug therapy such as aspirin and/or clopidogrel should be continued to reduce the risk of plaque rupture and recurrent myocardial infarction. Aspirin is first-line, owing to its low cost and comparable efficacy, with clopidogrel reserved for patients intolerant of aspirin. The combination of clopidogrel and aspirin may further reduce risk of cardiovascular events, however the risk of hemorrhage is increased.[157]
- Beta blocker therapy such as metoprolol or carvedilol should be commenced.[158] These have been particularly beneficial in high-risk patients such as those with left ventricular dysfunction and/or continuing cardiac ischaemia.[159] β-Blockers decrease mortality and morbidity. They also improve symptoms of cardiac ischemia in NSTEMI.
- ACE inhibitor therapy should be commenced 24–48 hours post-MI in hemodynamically-stable patients, particularly in patients with a history of MI, diabetes mellitus, hypertension, anterior location of infarct (as assessed by ECG), and/or evidence of left ventricular dysfunction. ACE inhibitors reduce mortality, the development of heart failure, and decrease ventricular remodelling post-MI.[160]
- Statin therapy has been shown to reduce mortality and morbidity post-MI.[161][162] The effects of statins may be more than their LDL lowering effects. The general consensus is that statins have plaque stabilization and multiple other ("pleiotropic") effects that may prevent myocardial infarction in addition to their effects on blood lipids.[163]
- The aldosterone antagonist agent eplerenone has been shown to further reduce risk of cardiovascular death post-MI in patients with heart failure and left ventricular dysfunction, when used in conjunction with standard therapies above.[164]
- Omega-3 fatty acids, commonly found in fish, have been shown to reduce mortality post-MI.[165] While the mechanism by which these fatty acids decrease mortality is unknown, it has been postulated that the survival benefit is due to electrical stabilization and the prevention of ventricular fibrillation.[166] However, further studies in a high-risk subset have not shown a clear-cut decrease in potentially fatal arrhythmias due to omega-3 fatty acids.[167][168]
Prognosis
The prognosis for patients with myocardial infarction varies greatly, depending on the patient, the condition itself and the given treatment. Using simple variables which are immediately available in the emergency room, patients with a higher risk of adverse outcome can be identified. For example, one study found that 0.4% of patients with a low risk profile had died after 90 days, whereas the mortality rate in high risk patients was 21.1%.[169]
Although studies differ in the identified variables, some of the more reproduced risk stratifiers include age, hemodynamic parameters (such as heart failure, cardiac arrest on admission, systolic blood pressure, or Killip class of two or greater), ST-segment deviation, diabetes, serum creatinine concentration, peripheral vascular disease and elevation of cardiac markers.[169][170][171]
Assessment of left ventricular ejection fraction may increase the predictive power of some risk stratification models.[172] The prognostic importance of Q-waves is debated.[173] Prognosis is significantly worsened if a mechanical complication (papillary muscle rupture, myocardial free wall rupture, and so on) were to occur.[150]
There is evidence that case fatality of myocardial infarction has been improving over the years in all ethnicities.[174]
Epidemiology
The examples and perspective in this article may not represent a worldwide view of the subject. |
Myocardial infarction is a common presentation of ischemic heart disease. The WHO estimated that in 2002, 12.6 percent of deaths worldwide were from ischemic heart disease.[2] Ischemic heart disease is the leading cause of death in developed countries, but third to AIDS and lower respiratory infections in developing countries.[175]
In the United States, diseases of the heart are the leading cause of death, causing a higher mortality than cancer (malignant neoplasms).[176] Coronary heart disease is responsible for 1 in 5 deaths in the U.S.. Some 7,200,000 men and 6,000,000 women are living with some form of coronary heart disease. 1,200,000 people suffer a (new or recurrent) coronary attack every year, and about 40% of them die as a result of the attack.[177] This means that roughly every 65 seconds, an American dies of a coronary event.
In India, cardiovascular disease (CVD) is the leading cause of death.[178] The deaths due to CVD in India were 32% of all deaths in 2007 and are expected to rise from 1.17 million in 1990 and 1.59 million in 2000 to 2.03 million in 2010.[179] Although a relatively new epidemic in India, it has quickly become a major health issue with deaths due to CVD expected to double during 1985-2015.[180][181] Mortality estimates due to CVD vary widely by state, ranging from 10% in Meghalaya to 49% in Punjab (percentage of all deaths). Punjab (49%), Goa (42%), Tamil Nadu (36%) and Andhra Pradesh (31%) have the highest CVD related mortality estimates.[182] State-wise differences are correlated with prevalence of specific dietary risk factors in the states. Moderate physical exercise is associated with reduced incidence of CVD in India (those who exercise have less than half the risk of those who don't).[180] CVD also affects Indians at a younger age (in their 30s and 40s) than is typical in other countries.
Legal implications
At common law, a myocardial infarction is generally a disease, but may sometimes be an injury. This has implications for no-fault insurance schemes such as workers' compensation. A heart attack is generally not covered;[183] however, it may be a work-related injury if it results, for example, from unusual emotional stress or unusual exertion.[184] Additionally, in some jurisdictions, heart attacks suffered by persons in particular occupations such as police officers may be classified as line-of-duty injuries by statute or policy. In some countries or states, a person who has suffered from a myocardial infarction may be prevented from participating in activity that puts other people's lives at risk, for example driving a car, taxi or airplane.[144]
References
- ^ Kosuge, M (March 2006). "Differences between men and women in terms of clinical features of ST-segment elevation acute myocardial infarction". Circulation Journal. 70 (3): 222–226. doi:10.1253/circj.70.222. PMID 16501283. Retrieved 2008-05-31.
{{cite journal}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ a b The World Health Report 2004 - Changing History (PDF). World Health Organization. 2004. pp. 120–4. ISBN 92-4-156265-X.
{{cite book}}: External link in|authorlink= - ^ Bax L, Algra A, Mali WP, Edlinger M, Beutler JJ, van der Graaf Y (2008). "Renal function as a risk indicator for cardiovascular events in 3216 patients with manifest arterial disease". Atherosclerosis. 200: 184. doi:10.1016/j.atherosclerosis.2007.12.006. PMID 18241872.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pearte CA, Furberg CD, O'Meara ES; et al. (2006). "Characteristics and baseline clinical predictors of future fatal versus nonfatal coronary heart disease events in older adults: the Cardiovascular Health Study". Circulation. 113 (18): 2177–85. doi:10.1161/CIRCULATIONAHA.105.610352. PMID 16651468.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Erhardt L, Herlitz J, Bossaert L; et al. (2002). "Task force on the management of chest pain" (PDF). Eur. Heart J. 23 (15): 1153–76. doi:10.1053/euhj.2002.3194. PMID 12206127.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ "Routine use of oxygen in the treatment of myocardial infarction: systematic review -- Wijesinghe et al. 95 (3): 198 -- Heart".
- ^ National Heart, Lung and Blood Institute. Heart Attack Warning Signs. Retrieved November 22, 2006.
- ^ Marcus GM, Cohen J, Varosy PD; et al. (2007). "The utility of gestures in patients with chest discomfort". Am. J. Med. 120 (1): 83–9. doi:10.1016/j.amjmed.2006.05.045. PMID 17208083.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Little RA, Frayn KN, Randall PE; et al. (1986). "Plasma catecholamines in the acute phase of the response to myocardial infarction". Arch Emerg Med. 3 (1): 20–7. PMID 3524599.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ a b Canto JG, Goldberg RJ, Hand MM; et al. (2007). "Symptom presentation of women with acute coronary syndromes: myth vs reality". Arch. Intern. Med. 167 (22): 2405–13. doi:10.1001/archinte.167.22.2405. PMID 18071161.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ McSweeney JC, Cody M, O'Sullivan P, Elberson K, Moser DK, Garvin BJ (2003). "Women's early warning symptoms of acute myocardial infarction". Circulation. 108 (21): 2619–23. doi:10.1161/01.CIR.0000097116.29625.7C. PMID 14597589.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ D Lee, D Kulick, J Marks. Heart Attack (Myocardial Infarction) by MedicineNet.com . Retrieved November 28, 2006.
- ^ Kannel WB. (1986). "Silent myocardial ischemia and infarction: insights from the Framingham Study". Cardiol Clin. 4 (4): 583–91. PMID 3779719.
- ^ a b Davis TM, Fortun P, Mulder J, Davis WA, Bruce DG (2004). "Silent myocardial infarction and its prognosis in a community-based cohort of Type 2 diabetic patients: the Fremantle Diabetes Study". Diabetologia. 47 (3): 395–9. doi:10.1007/s00125-004-1344-4. PMID 14963648.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Rubin's Pathology - Clinicopathological Foundations of Medicine. Maryland: Lippincott Williams & Wilkins. 2001. p. 549. ISBN 0-7817-4733-3.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Acute Coronary Syndrome. American Heart Association. Retrieved November 25, 2006.
- ^ Boie ET (2005). "Initial evaluation of chest pain". Emerg. Med. Clin. North Am. 23 (4): 937–57. doi:10.1016/j.emc.2005.07.007. PMID 16199332.
- ^ a b c d e f Wilson PW, D'Agostino RB, Levy D, Belanger AM, Silbershatz H, Kannel WB. "Prediction of coronary heart disease using risk factor categories". Circulation 1998; 97(18): 1837-47. PMID 9603539 Cite error: The named reference "Framingham1998" was defined multiple times with different content (see the help page).
- ^ a b Saikku P, Leinonen M, Tenkanen L, Linnanmaki E, Ekman MR, Manninen V, Manttari M, Frick MH, Huttunen JK. (1992). "Chronic Chlamydia pneumoniae infection as a risk factor for coronary heart disease in the Helsinki Heart Study". Ann Intern Med. 116 (4): 273–8. PMID 1733381.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Andraws R, Berger JS, Brown DL. (2005). "Effects of antibiotic therapy on outcomes of patients with coronary artery disease: a meta-analysis of randomized controlled trials". JAMA. 293 (21): 2641–7. doi:10.1001/jama.293.21.2641. PMID 15928286.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Muller JE, Stone PH, Turi ZG; et al. (1985). "Circadian variation in the frequency of onset of acute myocardial infarction". N. Engl. J. Med. 313 (21): 1315–22. PMID 2865677.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Beamer AD, Lee TH, Cook EF; et al. (1987). "Diagnostic implications for myocardial ischemia of the circadian variation of the onset of chest pain". Am. J. Cardiol. 60 (13): 998–1002. doi:10.1016/0002-9149(87)90340-7. PMID 3673917.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Cannon CP, McCabe CH, Stone PH; et al. (1997). "Circadian variation in the onset of unstable angina and non-Q-wave acute myocardial infarction (the TIMI III Registry and TIMI IIIB)". Am. J. Cardiol. 79 (3): 253–8. doi:10.1016/S0002-9149(97)00743-1. PMID 9036740.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Tofler GH, Brezinski D, Schafer AI; et al. (1987). "Concurrent morning increase in platelet aggregability and the risk of myocardial infarction and sudden cardiac death". N. Engl. J. Med. 316 (24): 1514–8. PMID 3587281.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Fantidis P, Perez De Prada T, Fernandez-Ortiz A; et al. (2002). "Morning cortisol production in coronary heart disease patients". Eur. J. Clin. Invest. 32 (5): 304–8. doi:10.1046/j.1365-2362.2002.00988.x. PMID 12027868.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Yusuf S, Hawken S, Ounpuu S, Bautista L, Franzosi MG, Commerford P, Lang CC, Rumboldt Z, Onen CL, Lisheng L, Tanomsup S, Wangai P Jr, Razak F, Sharma AM, Anand SS; INTERHEART Study Investigators. (2005). "Obesity and the risk of myocardial infarction in 27,000 participants from 52 countries: a case-control study". Lancet. 366 (9497): 1640–9. doi:10.1016/S0140-6736(05)67663-5. PMID 16271645.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Jensen G, Nyboe J, Appleyard M, Schnohr P. (1991). "Risk factors for acute myocardial infarction in Copenhagen, II: Smoking, alcohol intake, physical activity, obesity, oral contraception, diabetes, lipids, and blood pressure". Eur Heart J. 12 (3): 298–308. PMID 2040311.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Nyboe J, Jensen G, Appleyard M, Schnohr P. (1989). "Risk factors for acute myocardial infarction in Copenhagen. I: Hereditary, educational and socioeconomic factors. Copenhagen City Heart Study". Eur Heart J. 10 (10): 910–6. PMID 2598948.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Khader YS, Rice J, John L, Abueita O. (2003). "Oral contraceptives use and the risk of myocardial infarction: a meta-analysis". Contraception. 68 (1): 11–7. doi:10.1016/S0010-7824(03)00073-8. PMID 12878281.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b c d Wilson AM, Ryan MC, Boyle AJ. (2006). "The novel role of C-reactive protein in cardiovascular disease: risk marker or pathogen". Int J Cardiol. 106 (3): 291–7. doi:10.1016/j.ijcard.2005.01.068. PMID 16337036.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pearson TA, Mensah GA, Alexander RW, Anderson JL, Cannon RO 3rd, Criqui M, Fadl YY, Fortmann SP, Hong Y, Myers GL, Rifai N, Smith SC Jr, Taubert K, Tracy RP, Vinicor F; Centers for Disease Control and Prevention; American Heart Association. (2003). "Markers of inflammation and cardiovascular disease: application to clinical and public health practice: A statement for healthcare professionals from the Centers for Disease Control and Prevention and the American Heart Association" (PDF). Circulation. 107 (3): 499–511. doi:10.1161/01.CIR.0000052939.59093.45. PMID 12551878.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: numeric names: authors list (link) - ^ Janket SJ, Baird AE, Chuang SK, Jones JA. (2003). "Meta-analysis of periodontal disease and risk of coronary heart disease and stroke". Oral Surg Oral Med Oral Pathol Oral Radiol Endod. 95 (5): 559–69. doi:10.1038/sj.ebd.6400272. PMID 12738947.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Pihlstrom BL, Michalowicz BS, Johnson NW. (2005). "Periodontal diseases". Lancet. 366 (9499): 1809–20. doi:10.1016/S0140-6736(05)67728-8. PMID 16298220.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Scannapieco FA, Bush RB, Paju S. (2003). "Associations between periodontal disease and risk for atherosclerosis, cardiovascular disease, and stroke. A systematic review". Ann Periodontol. 8 (1): 38–53. doi:10.1902/annals.2003.8.1.38. PMID 14971247.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ D'Aiuto F, Parkar M, Nibali L, Suvan J, Lessem J, Tonetti MS. (2006). "Periodontal infections cause changes in traditional and novel cardiovascular risk factors: results from a randomized controlled clinical trial". Am Heart J. 151 (5): 977–84. doi:10.1016/j.ahj.2005.06.018. PMID 16644317.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Lourbakos A, Yuan YP, Jenkins AL, Travis J, Andrade-Gordon P, Santulli R, Potempa J, Pike RN. (2001). "Activation of protease-activated receptors by gingipains from Porphyromonas gingivalis leads to platelet aggregation: a new trait in microbial pathogenicity" (PDF). Blood. 97 (12): 3790–7. doi:10.1182/blood.V97.12.3790. PMID 11389018.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Qi M, Miyakawa H, Kuramitsu HK. (2003). "Porphyromonas gingivalis induces murine macrophage foam cell formation". Microb Pathog. 35 (6): 259–67. doi:10.1016/j.micpath.2003.07.002. PMID 14580389.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Spahr A, Klein E, Khuseyinova N, Boeckh C, Muche R, Kunze M, Rothenbacher D, Pezeshki G, Hoffmeister A, Koenig W. (2006). "Periodontal infections and coronary heart disease: role of periodontal bacteria and importance of total pathogen burden in the Coronary Event and Periodontal Disease (CORODONT) study". Arch Intern Med. 166 (5): 554–9. doi:10.1001/archinte.166.5.554. PMID 16534043.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Davis TM, Balme M, Jackson D, Stuccio G, Bruce DG (2000). "The diagonal ear lobe crease (Frank's sign) is not associated with coronary artery disease or retinopathy in type 2 diabetes: the Fremantle Diabetes Study". Aust N Z J Med. 30 (5): 573–7. PMID 11108067.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Lichstein E, Chadda KD, Naik D, Gupta PK. (1974). "Diagonal ear-lobe crease: prevalence and implications as a coronary risk factor". N Engl J Med. 290 (11): 615–6. PMID 4812503.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Miric D, Fabijanic D, Giunio L, Eterovic D, Culic V, Bozic I, Hozo I. (1998). "Dermatological indicators of coronary risk: a case-control study". Int J Cardiol. 67 (3): 251–5. doi:10.1016/S0167-5273(98)00313-1. PMID 9894707.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Greenland P, LaBree L, Azen SP, Doherty TM, Detrano RC (2004). "Coronary artery calcium score combined with Framingham score for risk prediction in asymptomatic individuals". JAMA. 291 (2): 210–5. doi:10.1001/jama.291.2.210. PMID 14722147.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Detrano R, Guerci AD, Carr JJ; et al. (2008). "Coronary calcium as a predictor of coronary events in four racial or ethnic groups". N. Engl. J. Med. 358 (13): 1336–45. doi:10.1056/NEJMoa072100. PMID 18367736.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Arad Y, Goodman KJ, Roth M, Newstein D, Guerci AD (2005). "Coronary calcification, coronary disease risk factors, C-reactive protein, and atherosclerotic cardiovascular disease events: the St. Francis Heart Study". J. Am. Coll. Cardiol. 46 (1): 158–65. doi:10.1016/j.jacc.2005.02.088. PMID 15992651.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ [1]Acute MI Powerpoint, Powerpoint on acute myocardial infarction
- ^ Krijnen PA, Nijmeijer R, Meijer CJ, Visser CA, Hack CE, Niessen HW. (2002). "Apoptosis in myocardial ischaemia and infarction". J Clin Pathol. 55 (11): 801–11. doi:10.1136/jcp.55.11.801. PMID 12401816.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Myocardial infarction: diagnosis and investigations - GPnotebook, retrieved November 27, 2006.
- ^ a b DE Fenton et al. Myocardial infarction - eMedicine, retrieved November 27, 2006.
- ^ HEART SCAN - Patient information from University College London. Retrieved November 27, 2006.
- ^ Skoufis E, McGhie AI (1998). "Radionuclide techniques for the assessment of myocardial viability". Tex Heart Inst J. 25 (4): 272–9. PMID 9885104.
- ^ Gillum RF, Fortmann SP, Prineas RJ, Kottke TE. International diagnostic criteria for acute myocardial infarction and acute stroke. Am Heart J 1984;108:150-8. PMID 6731265
- ^ Alpert JS, Thygesen K, Antman E, Bassand JP. (2000). "Myocardial infarction redefined--a consensus document of The Joint European Society of Cardiology/American College of Cardiology Committee for the redefinition of myocardial infarction". J Am Coll Cardiol. 36 (3): 959–69. doi:10.1016/S0735-1097(00)00804-4. PMID 10987628.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b S. Garas et al.. Myocardial Infarction. eMedicine. Retrieved November 22, 2006.
- ^ Kasper DL, Braunwald E, Fauci AS, Hauser SL, Longo DL, Jameson JL. Harrison's Principles of Internal Medicine. p. 1444. New York: McGraw-Hill, 2005. ISBN 0-07-139140-1.
- ^ Kasper DL, et al. Harrison's Principles of Internal Medicine. p. 1450.
- ^ a b Cannon CP at al. Management of Acute Coronary Syndromes. p. 175. New Jersey: Humana Press, 1999. ISBN 0-89603-552-2.
- ^ Selker HP, Zalenski RJ, Antman EM; et al. (1997). "An evaluation of technologies for identifying acute cardiac ischemia in the emergency department: executive summary of a National Heart Attack Alert Program Working Group Report". Ann Emerg Med. 29 (1): 1–12. doi:10.1016/S0196-0644(97)70297-X. PMID 8998085.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Masoudi FA, Magid DJ, Vinson DR; et al. (2006). "Implications of the failure to identify high-risk electrocardiogram findings for the quality of care of patients with acute myocardial infarction: results of the Emergency Department Quality in Myocardial Infarction (EDQMI) study". Circulation. 114 (15): 1565–71. doi:10.1161/CIRCULATIONAHA.106.623652. PMID 17015790.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b "2005 American Heart Association Guidelines for Cardiopulmonary Resuscitation and Emergency Cardiovascular Care - Part 8: Stabilization of the Patient With Acute Coronary Syndromes". Circulation. 112: IV–89–IV–110. 2005. doi:10.1161/CIRCULATIONAHA.105.166561.
- ^ Somers MP, Brady WJ, Perron AD, Mattu A (2002). "The prominant T wave: electrocardiographic differential diagnosis". Am J Emerg Med. 20 (3): 243–51. doi:10.1053/ajem.2002.32630. PMID 11992348.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Smith SW, Whitwam W (2006). "Acute coronary syndromes". Emerg. Med. Clin. North Am. 24 (1): 53–89, vi. doi:10.1016/j.emc.2005.08.008. PMID 16308113.
{{cite journal}}: Unknown parameter|month=ignored (help) - ^ Van Mieghem C, Sabbe M, Knockaert D (2004). "The clinical value of the ECG in noncardiac conditions". Chest. 125 (4): 1561–76. doi:10.1378/chest.125.4.1561. PMID 15078775.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Brady WJ, Perron AD, Martin ML, Beagle C, Aufderheide TP (2001). "Cause of ST segment abnormality in ED chest pain patients". Am J Emerg Med. 19 (1): 25–8. doi:10.1053/ajem.2001.18029. PMID 11146012.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b Wang K, Asinger RW, Marriott HJ (2003). "ST-segment elevation in conditions other than acute myocardial infarction". N. Engl. J. Med. 349 (22): 2128–35. doi:10.1056/NEJMra022580. PMID 14645641.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Brady WJ, Chan TC, Pollack M (2000). "Electrocardiographic manifestations: patterns that confound the EKG diagnosis of acute myocardial infarction-left bundle branch block, ventricular paced rhythm, and left ventricular hypertrophy". J Emerg Med. 18 (1): 71–8. doi:10.1016/S0736-4679(99)00178-X. PMID 10645842.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Brady WJ, Perron AD, Chan T (2001). "Electrocardiographic ST-segment elevation: correct identification of acute myocardial infarction (AMI) and non-AMI syndromes by emergency physicians". Acad Emerg Med. 8 (4): 349–60. doi:10.1111/j.1553-2712.2001.tb02113.x. PMID 11282670.
{{cite journal}}: Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Eisenman A (2006). "Troponin assays for the diagnosis of myocardial infarction and acute coronary syndrome: where do we stand?". Expert Rev Cardiovasc Ther. 4 (4): 509–14. doi:10.1586/14779072.4.4.509. PMID 16918269.
- ^ Aviles RJ, Askari AT, Lindahl B, Wallentin L, Jia G, Ohman EM, Mahaffey KW, Newby LK, Califf RM, Simoons ML, Topol EJ, Berger P, Lauer MS (2002). "Troponin T levels in patients with acute coronary syndromes, with or without renal dysfunction". N Engl J Med. 346 (26): 2047–52. doi:10.1056/NEJMoa013456. PMID 12087140.
{{cite journal}}: CS1 maint: multiple names: authors list (link). Summary for laymen - ^ Apple FS, Wu AH, Mair J; et al. (2005). "Future biomarkers for detection of ischemia and risk stratification in acute coronary syndrome". Clin. Chem. 51 (5): 810–24. doi:10.1373/clinchem.2004.046292. PMID 15774573.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Braunwald E, Antman EM, Beasley JW, Califf RM, Cheitlin MD, Hochman JS, Jones RH, Kereiakes D, Kupersmith J, Levin TN, Pepine CJ, Schaeffer JW, Smith EE III, Steward DE, Théroux P. (2002). "ACC/AHA 2002 guideline update for the management of patients with unstable angina and non–ST-segment elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee on the Management of Patients With Unstable Angina)" (PDF). J Am Coll Cardiol. 40: 1366–74. PMID 12383588.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Rubin's Pathology - Clinicopathological Foundations of Medicine. Maryland: Lippincott Williams & Wilkins. 2001. p. 546. ISBN 0-7817-4733-3.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Eichbaum FW. "'Wavy' myocardial fibers in spontaneous and experimental adrenergic cardiopathies" Cardiology 1975; 60(6): 358–65. PMID 782705
- ^ S Roy. Myocardial infarction. Retrieved November 28, 2006.
- ^ Fishbein MC. (1990). "Reperfusion injury". Clin Cardiol. 13 (3): 213–7. doi:10.1152/ajpheart.00270.2002. PMID 2182247.
{{cite journal}}: Unknown parameter|doi_brokendate=ignored (|doi-broken-date=suggested) (help) - ^ a b TIME IS MUSCLE TIME WASTED IS MUSCLE LOST. Early Heart Attack Care, St. Agnes Healthcare. Retrieved November 29, 2006.
- ^ Meine TJ, Roe MT, Chen AY; et al. (2005). "Association of intravenous morphine use and outcomes in acute coronary syndromes: results from the CRUSADE Quality Improvement Initiative". Am Heart J. 149 (6): 1043–9. doi:10.1016/j.ahj.2005.02.010. PMID 15976786.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ "Routine use of oxygen in the treatment of myocardial infarction: systematic review -- Wijesinghe et al. 95 (3): 198 -- Heart".
- ^ ISIS-2 Collaborative group (1988). "Randomized trial of intravenous streptokinase, oral aspirin, both, or neither among 17,187 cases of suspected acute myocardial infarction: ISIS-2". Lancet. 2 (8607): 349–60. PMID 2899772.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ^ ISIS-1 Collaborative Group (1986). "Randomised trial of intravenous atenolol among 16 027 cases of suspected acute myocardial infarction: ISIS-1". Lancet. 2 (8498): 57–66. PMID 2873379.
{{cite journal}}: CS1 maint: numeric names: authors list (link) - ^ The TIMI Study Group (1989). "Comparison of invasive and conservative strategies after treatment with intravenous tissue plasminogen activator in acute myocardial infarction". N Engl J Med. 320 (10): 618–27. PMID 2563896.
- ^ a b c d e Antman EM, Anbe DT, Armstrong PW, Bates ER, Green LA, Hand M, Hochman JS, Krumholz HM, Kushner FG, Lamas GA, Mullany CJ, Ornato JP, Pearle DL, Sloan MA, Smith SC Jr (2004). "ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction: a report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Committee to Revise the 1999 Guidelines for the Management of Patients With Acute Myocardial Infarction)". J Am Coll Cardiol. 44: 671–719. PMID 15358045.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ McCord J, Jneid H, Hollander JE; et al. (2008). "Management of cocaine-associated chest pain and myocardial infarction: a scientific statement from the American Heart Association Acute Cardiac Care Committee of the Council on Clinical Cardiology". Circulation. 117 (14): 1897–907. doi:10.1161/CIRCULATIONAHA.107.188950. PMID 18347214.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Faxon DP. "Coronary interventions and their impact on post myocardial infarction survival." Clin Cardiol 2005; 28(11 Suppl 1):I38-44. PMID 16450811
- ^ Heart attack first aid. MedlinePlus. Retrieved December 3, 2006.
- ^ Act In Time to Heart Attack Signs - NHLBI. Retrieved December 13, 2006.
- ^ Brown AL, Mann NC, Daya M, Goldberg R, Meischke H, Taylor J, Smith K, Osganian S, Cooper L. (2000). "Demographic, belief, and situational factors influencing the decision to utilize emergency medical services among chest pain patients. Rapid Early Action for Coronary Treatment (REACT) study". Circulation. 102 (2): 173–8. PMID 10889127.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Rossi S, editor. Australian Medicines Handbook 2006. Adelaide: Australian Medicines Handbook; 2006. ISBN 0-9757919-2-3.
- ^ a b Antman EM, Anbe DT, Armstrong PW; et al. (2004). "ACC/AHA guidelines for the management of patients with ST-elevation myocardial infarction--executive summary. A report of the American College of Cardiology/American Heart Association Task Force on Practice Guidelines (Writing Committee to revise the 1999 guidelines for the management of patients with acute myocardial infarction)". J. Am. Coll. Cardiol. 44 (3): 671–719. doi:10.1016/j.jacc.2004.07.002. PMID 15358045.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Morrow DA, Antman EM, Sayah A; et al. (2002). "Evaluation of the time saved by prehospital initiation of reteplase for ST-elevation myocardial infarction: results of The Early Retavase-Thrombolysis in Myocardial Infarction (ER-TIMI) 19 trial". J. Am. Coll. Cardiol. 40 (1): 71–7. doi:10.1016/S0735-1097(02)01936-8. PMID 12103258.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ a b Morrison LJ, Verbeek PR, McDonald AC, Sawadsky BV, Cook DJ. (2000). "Mortality and prehospital thrombolysis for acute myocardial infarction: A meta-analysis" (PDF). JAMA. 283 (20): 2686–92. doi:10.1001/jama.283.20.2686. PMID 10819952.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Rokos IC, Larson DM, Henry TD; et al. (2006). "Rationale for establishing regional ST-elevation myocardial infarction receiving center (SRC) networks". Am. Heart J. 152 (4): 661–7. doi:10.1016/j.ahj.2006.06.001. PMID 16996830.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Moyer P, Feldman J, Levine J; et al. (2004). "Implications of the Mechanical (PCI) vs Thrombolytic Controversy for ST Segment Elevation Myocardial Infarction on the Organization of Emergency Medical Services: The Boston EMS Experience". Crit Pathw Cardiol. 3 (2): 53–61. doi:10.1097/01.hpc.0000128714.35330.6d. PMID 18340140.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Terkelsen CJ, Lassen JF, Nørgaard BL; et al. (2005). "Reduction of treatment delay in patients with ST-elevation myocardial infarction: impact of pre-hospital diagnosis and direct referral to primary percutanous coronary intervention". Eur. Heart J. 26 (8): 770–7. doi:10.1093/eurheartj/ehi100. PMID 15684279.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link)T - ^ Henry TD, Atkins JM, Cunningham MS; et al. (2006). "ST-segment elevation myocardial infarction: recommendations on triage of patients to heart attack centers: is it time for a national policy for the treatment of ST-segment elevation myocardial infarction?". J. Am. Coll. Cardiol. 47 (7): 1339–45. doi:10.1016/j.jacc.2005.05.101. PMID 16580518.
{{cite journal}}: Explicit use of et al. in:|author=(help); Unknown parameter|month=ignored (help)CS1 maint: multiple names: authors list (link) - ^ Rokos I. and Bouthillet T., "The emergency medical systems-to-balloon (E2B) challenge: building on the foundations of the D2B Alliance," STEMI Systems, Issue Two, May 2007. Accessed June 16, 2007.
- ^ Cannon, Christopher (1999). Management of acute coronary syndromes. Totowa, NJ: Humana Press. ISBN 0-89603-552-2.
- ^ Lee KL, Woodlief LH, Topol EJ, et al. "Predictors of 30-Day Mortality in the Era of Reperfusion for Acute Myocardial Infarction." Circulation 1995; 91: 1659-1668. PMID 7882472
- ^ Stone GW, Grines CL, Browne KF, et al. "Predictors of in-hospital and 6-month outcome after acute myocardial infarction in the reperfusion era: the Primary Angioplasty in Myocardial Infarction (PAMI) trail." J Am Coll Cardiol 1995; 25: 370-377. PMID 14645641
- ^ a b c "Indications for fibrinolytic therapy in suspected acute myocardial infarction: collaborative overview of early mortality and major morbidity results from all randomised trials of more than 1000 patients. Fibrinolytic Therapy Trialists' (FTT) Collaborative Group." Lancet 1994; 343(8893): 311-22. PMID 7905143
- ^ Verheugt FW, Gersh BJ, Armstrong PW. "Aborted myocardial infarction: a new target for reperfusion therapy." Eur Heart J 2006; 27(8): 901-4. PMID 16543251
- ^ Boersma E, Maas AC, Deckers JW, Simoons ML. "Early thrombolytic treatment in acute myocardial infarction: reappraisal of the golden hour." Lancet 1996; 348 (9030): 771-5. PMID 8813982
- ^ "Effects of tissue plasminogen activator and a comparison of early invasive and conservative strategies in unstable angina and non-Q-wave myocardial infarction. Results of the TIMI IIIB Trial. Thrombolysis in Myocardial Ischemia." Circulation 1994; 89 (4): 1545-56. PMID 8149520
- ^ a b c d Hochman JS, Sleeper LA, Webb JG, Sanborn TA, White HD, Talley JD, Buller CE, Jacobs AK, Slater JN, Col J, McKinlay SM, LeJemtel TH. (1999). "Early revascularization in acute myocardial infarction complicated by cardiogenic shock. SHOCK Investigators. Should We Emergently Revascularize Occluded Coronaries for Cardiogenic Shock". N Engl J Med. 341 (9): 625–34. doi:10.1056/NEJM199908263410901. PMID 10460813.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ White HD, Van de Werf FJ. "Thrombolysis for acute myocardial infarction.." Circulation 1998; 97 (16): 1632-46. PMID 9593569
- ^ a b c The GUSTO investigators (1993). "An international randomized trial comparing four thrombolytic strategies for acute myocardial infarction. The GUSTO investigators". N Engl J Med. 329 (10): 673–82. doi:10.1056/NEJM199309023291001. PMID 8204123.
- ^ Sabatine MS, Morrow DA, Montalescot G, Dellborg M, Leiva-Pons JL, Keltai M, Murphy SA, McCabe CH, Gibson CM, Cannon CP, Antman EM, Braunwald E; Clopidogrel as Adjunctive Reperfusion Therapy (CLARITY)-Thrombolysis in Myocardial Infarction (TIMI) 28 Investigators. (2005). "Angiographic and clinical outcomes in patients receiving low-molecular-weight heparin versus unfractionated heparin in ST-elevation myocardial infarction treated with fibrinolytics in the CLARITY-TIMI 28 Trial". Circulation. 112 (25): 3846–54. doi:10.1161/CIRCULATIONAHA.105.595397. PMID 16291601.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: numeric names: authors list (link) - ^ Cowley MJ, Hastillo A, Vetrovec GW, Fisher LM, Garrett R, Hess ML. (1983). "Fibrinolytic effects of intracoronary streptokinase administration in patients with acute myocardial infarction and coronary insufficiency". Circulation. 67 (5): 1031–8. PMID 6831667.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Lourenco DM, Dosne AM, Kher A, Samama M. (1989). "Effect of standard heparin and a low molecular weight heparin on thrombolytic and fibrinolytic activity of single-chain urokinase plasminogen activator in vitro". Thromb Haemost. 62 (3): 923–6. PMID 2556812.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Van de Werf F, Vanhaecke J, de Geest H, Verstraete M, Collen D. (1986). "Coronary thrombolysis with recombinant single-chain urokinase-type plasminogen activator in patients with acute myocardial infarction". Circulation. 74 (5): 1066–70. PMID 2429783.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Bode C, Schoenermark S, Schuler G, Zimmermann R, Schwarz F, Kuebler W. (1988). "Efficacy of intravenous prourokinase and a combination of prourokinase and urokinase in acute myocardial infarction". Am J Cardiol. 61 (13): 971–4. doi:10.1016/0002-9149(88)90108-7. PMID 2452564.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Boersma E, Maas AC, Deckers JW, Simoons ML. (1996). "Early thrombolytic treatment in acute myocardial infarction: reappraisal of the golden hour". Lancet. 348 (9030): 771–5. doi:10.1016/S0140-6736(96)02514-7. PMID 8813982.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ LATE trial intestigatos. (1993). "Late Assessment of Thrombolytic Efficacy (LATE) study with alteplase 6-24 hours after onset of acute myocardial infarction". Lancet. 342 (8874): 759–66. doi:10.1016/0140-6736(93)91538-W. PMID 8103874.
- ^ Chesebro JH, Knatterud G, Roberts R, Borer J, Cohen LS, Dalen J, Dodge HT, Francis CK, Hillis D, Ludbrook P; et al. (1987). "Thrombolysis in Myocardial Infarction (TIMI) Trial, Phase I: A comparison between intravenous tissue plasminogen activator and intravenous streptokinase. Clinical findings through hospital discharge". Circulation. 76 (1): 142–54. PMID 3109764.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ a b Keeley EC, Boura JA, Grines CL. (2003). "Primary angioplasty versus intravenous thrombolytic therapy for acute myocardial infarction: a quantitative review of 23 randomised trials". Lancet. 361 (9351): 13–20. doi:10.1016/S0140-6736(03)12113-7. PMID 12517460.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Grines CL, Browne KF, Marco J, Rothbaum D, Stone GW, O'Keefe J, Overlie P, Donohue B, Chelliah N, Timmis GC; et al. (1993). "A comparison of immediate angioplasty with thrombolytic therapy for acute myocardial infarction. The Primary Angioplasty in Myocardial Infarction Study Group". N Engl J Med. 328 (10): 673–9. doi:10.1056/NEJM199303113281001. PMID 8433725.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ The Global Use of Strategies to Open Occluded Coronary Arteries in Acute Coronary Syndromes (GUSTO IIb) Angioplasty Substudy Investigators. (1997). "A clinical trial comparing primary coronary angioplasty with tissue plasminogen activator for acute myocardial infarction". N Engl J Med. 336 (23): 1621–8. doi:10.1056/NEJM199706053362301. PMID 9173270.
- ^ Boersma E; The Primary Coronary Angioplasty vs. Thrombolysis Group. "Does time matter? A pooled analysis of randomized clinical trials comparing primary percutaneous coronary intervention and in-hospital fibrinolysis in acute myocardial infarction patients." Eur Heart J 2006; 27(7):779-88. PMID 16513663
- ^ Rokos IC, Larson DM, Henry TD, et al.; "Rationale for establishing regional ST-elevation myocardial infarction receiving center (SRC) networks." Am Heart J 2006; 152(4):661-7. PMID 16996830
- ^ Bradley EH, Herrin J, Wang Y, Barton BA, Webster TR, Mattera JA, Roumanis SA, Curtis JP, Nallamothu BK, Magid DJ, McNamara RL, Parkosewich J, Loeb JM, Krumholz HM. "Strategies for reducing the door-to-balloon time in acute myocardial infarction." N Engl J Med 2006; 355(22): 2308-20. PMID 17101617
- ^ "D2B: An Alliance for Quality". American College of Cardiology. 2006. Retrieved April 15 2007.
{{cite web}}: Check date values in:|accessdate=(help); Unknown parameter|dateformat=ignored (help) - ^ De Villiers JS, Anderson T, McMeekin JD; et al. (2007). "Expedited transfer for primary percutaneous coronary intervention: a program evaluation". CMAJ. 176: 1833–8. PMID 17576980.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Aversano T; et al. (2002). "Thrombolytic therapy vs primary percutaneous coronary intervention for myocardial infarction in patients presenting to hospitals without on-site cardiac surgery: a randomized controlled trial". JAMA. 287 (15): 1943–51. doi:10.1001/jama.287.15.1943. PMID 11960536.
{{cite journal}}: Explicit use of et al. in:|author=(help) - ^ Grines CL, Cox DA, Stone GW, Garcia E, Mattos LA, Giambartolomei A, Brodie BR, Madonna O, Eijgelshoven M, Lansky AJ, O'Neill WW, Morice MC. (1999). "Coronary angioplasty with or without stent implantation for acute myocardial infarction. Stent Primary Angioplasty in Myocardial Infarction Study Group". N Engl J Med. 341 (26): 1949–56. doi:10.1056/NEJM199912233412601. PMID 10607811.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Brener SJ, Barr LA, Burchenal JE, Katz S, George BS, Jones AA, Cohen ED, Gainey PC, White HJ, Cheek HB, Moses JW, Moliterno DJ, Effron MB, Topol EJ. (1998). "Randomized, placebo-controlled trial of platelet glycoprotein IIb/IIIa blockade with primary angioplasty for acute myocardial infarction. ReoPro and Primary PTCA Organization and Randomized Trial (RAPPORT) Investigators". Circulation. 98 (8): 734–41. PMID 9727542.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Tcheng JE, Kandzari DE, Grines CL, Cox DA, Effron MB, Garcia E, Griffin JJ, Guagliumi G, Stuckey T, Turco M, Fahy M, Lansky AJ, Mehran R, Stone GW; CADILLAC Investigators. (2003). "Benefits and risks of abciximab use in primary angioplasty for acute myocardial infarction: the Controlled Abciximab and Device Investigation to Lower Late Angioplasty Complications (CADILLAC) trial". Circulation. 108 (11): 1316–23. doi:10.1161/01.CIR.0000087601.45803.86. PMID 12939213.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Mukherjee, Debabrata (2006). 900 Questions: An Interventional Cardiology Board Review. Lippincott Williams & Wilkins. ISBN 0781773490.
- ^ Babaev A, Frederick PD, Pasta DJ, Every N, Sichrovsky T, Hochman JS (2005). "Trends in management and outcomes of patients with acute myocardial infarction complicated by cardiogenic shock". JAMA. 294 (4): 448–54. doi:10.1001/jama.294.4.448. PMID 16046651.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Townsend, Courtney M. (2004). Sabiston Textbook of Surgery - The Biological Basis of Modern Surgical Practice. Philadelphia, Pennsylvania: Elsevier Saunders. p. 1871. ISBN 0-7216-0409-9.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Kaul TK, Fields BL, Riggins SL, Dacumos GC, Wyatt DA, Jones CR (1995). "Coronary artery bypass grafting within 30 days of an acute myocardial infarction". Ann. Thorac. Surg. 59 (5): 1169–76. doi:10.1016/0003-4975(95)00125-5. PMID 7733715.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Creswell LL, Moulton MJ, Cox JL, Rosenbloom M (1995). "Revascularization after acute myocardial infarction". Ann. Thorac. Surg. 60 (1): 19–26. PMID 7598589.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ White HD, Assmann SF, Sanborn TA; et al. (2005). "Comparison of percutaneous coronary intervention and coronary artery bypass grafting after acute myocardial infarction complicated by cardiogenic shock: results from the Should We Emergently Revascularize Occluded Coronaries for Cardiogenic Shock (SHOCK) trial". Circulation. 112 (13): 1992–2001. doi:10.1161/CIRCULATIONAHA.105.540948. PMID 16186436.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Hochman JS, Sleeper LA, Webb JG, Dzavik V, Buller CE, Aylward P, Col J, White HD; SHOCK Investigators. (2006). "Early revascularization and long-term survival in cardiogenic shock complicating acute myocardial infarction". JAMA. 295 (21): 2511–5. doi:10.1001/jama.295.21.2511. PMID 16757723.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Raja SG, Haider Z, Ahmad M, Zaman H (2004). "Saphenous vein grafts: to use or not to use?". Heart Lung Circ. 13 (4): 403–9. doi:10.1016/j.hlc.2004.04.004. PMID 16352226.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Hannan EL, Racz MJ, Walford G; et al. (2005). "Long-term outcomes of coronary-artery bypass grafting versus stent implantation". N. Engl. J. Med. 352 (21): 2174–83. doi:10.1056/NEJMoa040316. PMID 15917382.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ a b Bourassa MG (2000). "Clinical trials of coronary revascularization: coronary angioplasty vs. coronary bypass grafting". Curr. Opin. Cardiol. 15 (4): 281–6. doi:10.1097/00001573-200007000-00013. PMID 11139092.
- ^ Hlatky MA, Boothroyd DB, Melsop KA; et al. (2004). "Medical costs and quality of life 10 to 12 years after randomization to angioplasty or bypass surgery for multivessel coronary artery disease". Circulation. 110 (14): 1960–6. doi:10.1161/01.CIR.0000143379.26342.5C. PMID 15451795.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Echt DS, Liebson PR, Mitchell LB, Peters RW, Obias-Manno D, Barker AH, Arensberg D, Baker A, Friedman L, Greene HL; et al. (1991). "Mortality and morbidity in patients receiving encainide, flecainide, or placebo. The Cardiac Arrhythmia Suppression Trial". N Engl J Med. 324 (12): 781–8. PMID 1900101.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Waldo AL, Camm AJ, deRuyter H, Friedman PL, MacNeil DJ, Pauls JF, Pitt B, Pratt CM, Schwartz PJ, Veltri EP. (1996). "Effect of d-sotalol on mortality in patients with left ventricular dysfunction after recent and remote myocardial infarction. The SWORD Investigators. Survival With Oral d-Sotalol". Lancet. 348 (9019): 7–12. doi:10.1016/S0140-6736(96)02149-6. PMID 8691967.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Julian DG, Camm AJ, Frangin G, Janse MJ, Munoz A, Schwartz PJ, Simon P. (1997). "Randomised trial of effect of amiodarone on mortality in patients with left-ventricular dysfunction after recent myocardial infarction: EMIAT. European Myocardial Infarct Amiodarone Trial Investigators". Lancet. 349 (9053): 667–74. doi:10.1016/S0140-6736(96)09145-3. PMID 9078197.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Youngwith, Janice (2008-02-06). "Saving hearts in the air". Dailyherald.com. Retrieved 2008-06-12.
- ^ Dowdall N. "'Is there a doctor on the aircraft?' Top 10 in-flight medical emergencies." BMJ 2000; 321(7272):1336-7. PMID 11090520. PMC 1119071
- ^ a b Life after a Heart Attack. U.S. National Heart, Lung and Blood Institute. Retrieved December 2, 2006.
- ^ Trisha Macnair. Recovering after a heart attack. BBC, December 2005. Retrieved December 2, 2006.
- ^ a b "Classification of Drivers' Licenses Regulations". Nova Scotia Registry of Regulations. May 24, 2000. Retrieved April 22 2007.
{{cite web}}: Check date values in:|accessdate=(help); Unknown parameter|dateformat=ignored (help) - ^ "Heart Attack: Getting Back Into Your Life After a Heart Attack". American Academy of Family Physicians, updated March 2005. Retrieved December 4, 2006.
- ^ Schachinger V, Erbs S, Elsasser A, Haberbosch W, Hambrecht R, Holschermann H, Yu J, Corti R, Mathey DG, Hamm CW, Suselbeck T, Assmus B, Tonn T, Dimmeler S, Zeiher AM; REPAIR-AMI Investigators (2006). "Intracoronary bone marrow-derived progenitor cells in acute myocardial infarction". N Engl J Med. 355 (12): 1210–21. doi:10.1056/NEJMoa060186. PMID 16990384.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Christman KL, Lee RJ. "Biomaterials for the Treatment of Myocardial Infarction". J Am Coll Cardiol 2006; 48(5): 907-13. PMID 16949479
- ^ Canto JG, Shlipak MG, Rogers WJ, Malmgren JA, Frederick PD, Lambrew CT, Ornato JP, Barron HV, Kiefe CI. (2000). "Prevalence, clinical characteristics, and mortality among patients with myocardial infarction presenting without chest pain". JAMA. 283 (24): 3223–9. doi:10.1001/jama.283.24.3223. PMID 10866870.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Yip HK, Wu CJ, Chang HW, Wang CP, Cheng CI, Chua S, Chen MC. (2003). "Cardiac rupture complicating acute myocardial infarction in the direct percutaneous coronary intervention reperfusion era" (PDF). Chest. 124 (2): 565–71. doi:10.1378/chest.124.2.565. PMID 12907544.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Becker RC, Gore JM, Lambrew C, Weaver WD, Rubison RM, French WJ, Tiefenbrunn AJ, Bowlby LJ, Rogers WJ. (1996). "A composite view of cardiac rupture in the United States National Registry of Myocardial Infarction". J Am Coll Cardiol. 27 (6): 1321–6. doi:10.1016/0735-1097(96)00008-3. PMID 8626938.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Moreno R, Lopez-Sendon J, Garcia E, Perez de Isla L, Lopez de Sa E, Ortega A, Moreno M, Rubio R, Soriano J, Abeytua M, Garcia-Fernandez MA. (2002). "Primary angioplasty reduces the risk of left ventricular free wall rupture compared with thrombolysis in patients with acute myocardial infarction". J Am Coll Cardiol. 39 (4): 598–603. doi:10.1016/S0735-1097(01)01796-X. PMID 11849857.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Shin P, Sakurai M, Minamino T, Onishi S, Kitamura H. (1983). "Postinfarction cardiac rupture. A pathogenetic consideration in eight cases". Acta Pathol Jpn. 33 (5): 881–93. PMID 6650169.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Podrid, Philip J. (2001). Cardiac Arrhythmia: Mechanisms, Diagnosis, and Management. Lippincott Williams & Wilkins. ISBN 0781724864.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Sung, Ruey J. (2000). Fundamental Approaches to the Management of Cardiac Arrhythmias. Springer. ISBN 0792365593.
{{cite book}}: Unknown parameter|coauthors=ignored (|author=suggested) (help) - ^ Josephson, Mark E. (2002). Clinical Cardiac Electrophysiology: Techniques and Interpretations. Lippincott Williams & Wilkins. ISBN 0683306936.
- ^ Smith A, Aylward P, Campbell T, et al. Therapeutic Guidelines: Cardiovascular, 4th edition. North Melbourne: Therapeutic Guidelines; 2003. ISSN 1327-9513
- ^ Peters RJ, Mehta SR, Fox KA, Zhao F, Lewis BS, Kopecky SL, Diaz R, Commerford PJ, Valentin V, Yusuf S; Clopidogrel in Unstable angina to prevent Recurrent Events (CURE) Trial Investigators. (2003). "Effects of aspirin dose when used alone or in combination with clopidogrel in patients with acute coronary syndromes: observations from the Clopidogrel in Unstable angina to prevent Recurrent Events (CURE) study". Circulation. 108 (14): 1682–7. doi:10.1161/01.CIR.0000091201.39590.CB. PMID 14504182.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Yusuf S, Peto R, Lewis J, Collins R, Sleight P (1985). "Beta blockade during and after myocardial infarction: an overview of the randomized trials". Prog Cardiovasc Dis. 27 (5): 335–71. doi:10.1016/S0033-0620(85)80003-7. PMID 2858114.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Dargie HJ. (2001). "Effect of carvedilol on outcome after myocardial infarction in patients with left-ventricular dysfunction: the CAPRICORN randomised trial". Lancet. 357 (9266): 1385–90. doi:10.1016/S0140-6736(00)04560-8. PMID 11356434.
- ^ Pfeffer MA, Braunwald E, Moye LA, Basta L, Brown EJ Jr, Cuddy TE, Davis BR, Geltman EM, Goldman S, Flaker GC; et al. (1992). "Effect of captopril on mortality and morbidity in patients with left ventricular dysfunction after myocardial infarction. Results of the survival and ventricular enlargement trial. The SAVE Investigators". N Engl J Med. 327 (10): 669–77. PMID 1386652.
{{cite journal}}: Explicit use of et al. in:|author=(help)CS1 maint: multiple names: authors list (link) - ^ Sacks FM, Pfeffer MA, Moye LA, Rouleau JL, Rutherford JD, Cole TG, Brown L, Warnica JW, Arnold JM, Wun CC, Davis BR, Braunwald E. (1996). "The effect of pravastatin on coronary events after myocardial infarction in patients with average cholesterol levels. Cholesterol and Recurrent Events Trial investigators". N Engl J Med. 335 (14): 1001–9. doi:10.1056/NEJM199610033351401. PMID 8801446.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Sacks FM, Moye LA, Davis BR, Cole TG, Rouleau JL, Nash DT, Pfeffer MA, Braunwald E. (1998). "Relationship between plasma LDL concentrations during treatment with pravastatin and recurrent coronary events in the Cholesterol and Recurrent Events trial". Circulation. 97 (15): 1446–52. PMID 9576424.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Ray KK, Cannon CP (2005). "The potential relevance of the multiple lipid-independent (pleiotropic) effects of statins in the management of acute coronary syndromes". J. Am. Coll. Cardiol. 46 (8): 1425–33. doi:10.1016/j.jacc.2005.05.086. PMID 16226165.
- ^ Keating G, Plosker G (2004). "Eplerenone: a review of its use in left ventricular systolic dysfunction and heart failure after acute myocardial infarction". Drugs. 64 (23): 2689–707. doi:10.1157/13089615. PMID 15537370.
- ^ "Dietary supplementation with n-3 polyunsaturated fatty acids and vitamin E after myocardial infarction: results of the GISSI-Prevenzione trial. Gruppo Italiano per lo Studio della Sopravvivenza nell'Infarto miocardico". Lancet. 354 (9177): 447–55. 2001. doi:10.1016/S0140-6736(99)07072-5. PMID 10465168.
- ^ Leaf A, Albert C, Josephson M, Steinhaus D, Kluger J, Kang J, Cox B, Zhang H, Schoenfeld D (2005). "Prevention of fatal arrhythmias in high-risk subjects by fish oil n-3 fatty acid intake". Circulation. 112 (18): 2762–8. doi:10.1161/CIRCULATIONAHA.105.549527. PMID 16267249.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Brouwer IA, Zock PL, Camm AJ, Bocker D, Hauer RN, Wever EF, Dullemeijer C, Ronden JE, Katan MB, Lubinski A, Buschler H, Schouten EG; SOFA Study Group. (2006). "Effect of fish oil on ventricular tachyarrhythmia and death in patients with implantable cardioverter defibrillators: the Study on Omega-3 Fatty Acids and Ventricular Arrhythmia (SOFA) randomized trial". JAMA. 295 (22): 2613–9. doi:10.1001/jama.295.22.2613. PMID 16772624.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Raitt MH, Connor WE, Morris C, Kron J, Halperin B, Chugh SS, McClelland J, Cook J, MacMurdy K, Swenson R, Connor SL, Gerhard G, Kraemer DF, Oseran D, Marchant C, Calhoun D, Shnider R, McAnulty J. (2005). "Fish oil supplementation and risk of ventricular tachycardia and ventricular fibrillation in patients with implantable defibrillators: a randomized controlled trial". JAMA. 293 (23): 2284–91. doi:10.1001/jama.293.23.2884. PMID 15956633.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ a b Lopez de Sa E, Lopez-Sendon J, Anguera I, Bethencourt A, Bosch X; Proyecto de Estudio del Pronostico de la Angina (PEPA) Investigators. "Prognostic value of clinical variables at presentation in patients with non-ST-segment elevation acute coronary syndromes: results of the Proyecto de Estudio del Pronostico de la Angina (PEPA)." Medicine (Baltimore) 2002; 81(6): 434-42. PMID 12441900
- ^ Fox KA, Dabbous OH, Goldberg RJ, Pieper KS, Eagle KA, Van de Werf F, Avezum A, Goodman SG, Flather MD, Anderson FA Jr, Granger CB. "Prediction of risk of death and myocardial infarction in the six months after presentation with acute coronary syndrome: prospective multinational observational study (GRACE)." BMJ 2006; 333(7578):1091. PMID 17032691
- ^ Weir RA, McMurray JJ, Velazquez EJ. (2006). "Epidemiology of heart failure and left ventricular systolic dysfunction after acute myocardial infarction: prevalence, clinical characteristics, and prognostic importance". Am J Cardiol. 97 (10A): 13F–25F. doi:10.1016/j.amjcard.2006.03.005. PMID 16698331.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Bosch X, Theroux P. (2005). "Left ventricular ejection fraction to predict early mortality in patients with non-ST-segment elevation acute coronary syndromes". Am Heart J. 150 (2): 215–20. doi:10.1016/j.ahj.2004.09.027. PMID 16086920.
- ^ Nicod P, Gilpin E, Dittrich H, Polikar R, Hjalmarson A, Blacky A, Henning H, Ross J (1989). "Short- and long-term clinical outcome after Q wave and non-Q wave myocardial infarction in a large patient population". Circulation. 79 (3): 528–36. PMID 2645061.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Liew R, Sulfi S, Ranjadayalan K, Cooper J, Timmis AD. (2006). "Declining case fatality rates for acute myocardial infarction in South Asian and white patients in the past 15 years". Heart. 92 (8): 1030–4. doi:10.1136/hrt.2005.078634. PMID 16387823.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ "Cause of Death - UC Atlas of Global Inequality". Center for Global, International and Regional Studies (CGIRS) at the University of California Santa Cruz. Retrieved December 7 2006.
{{cite web}}: Check date values in:|accessdate=(help); Unknown parameter|dateformat=ignored (help) - ^ "Deaths and percentage of total death for the 10 leading causes of death: United States, 2002-2003" (PDF). National Center of Health Statistics. Retrieved April 17 2007.
{{cite web}}: Check date values in:|accessdate=(help); Unknown parameter|dateformat=ignored (help) - ^ "Heart Attack and Angina Statistics". American Heart Association. 2003. Retrieved December 7 2006.
{{cite web}}: Check date values in:|accessdate=(help); Unknown parameter|dateformat=ignored (help) - ^ Mukherjee AK. (1995). "Prediction of coronary heart disease using risk factor categories". J Indian Med Assoc. PMID 8713248.
- ^ Ghaffar A, Reddy KS and Singhi M (2004). "Burden of non-communicable diseases in South Asia" (PDF). BMJ. 328: 807–810. doi:10.1136/bmj.328.7443.807. PMID 15070638.
- ^ a b Rastogi T, Vaz M, Spiegelman D, Reddy KS, Bharathi AV, Stampfer MJ, Willett WC and Ascherio1 A (2004). "Physical activity and risk of coronary heart disease in India" (PDF). Int. J. Epidemiol. 33: 1–9. doi:10.1093/ije/dyh042. PMID 15044412.
{{cite journal}}: CS1 maint: multiple names: authors list (link) CS1 maint: numeric names: authors list (link) - ^ Gupta R. (2007). "Escalating Coronary Heart Disease and Risk Factors in South Asians" (PDF). Indian Heart Journal: 214–17.
- ^ Gupta R, Misra A, Pais P, Rastogi P and Gupta VP. (2006). "Correlation of regional cardiovascular disease mortality in India with lifestyle and nutritional factors" (PDF). International Journal of Cardiology. 108 (3): 291–300. doi:10.1016/j.ijcard.2005.05.044.
{{cite journal}}: CS1 maint: multiple names: authors list (link) - ^ Workers' Compensation FAQ. Prairie View A&M University. Retrieved November 22, 2006.
- ^ SIGNIFICANT DECISIONS Subject Index. Board of Industrial Insurance Appeals. Retrieved November 22, 2006.
External links
- American Heart Association's Heart Attack web site - Information and resources for preventing, recognizing and treating heart attack.
- Heart Attack - overview of resources from MedlinePlus.
- Risk Assessment Tool for Estimating 10-year Risk of Having a Heart Attack - based on information of the Framingham Heart Study, from the United States National Heart, Lung and Blood Institute
